Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Mitogen-activated protein kinase 14
Ligand
BDBM13534
Substrate
n/a
Meas. Tech.
ChEMBL_586347 (CHEMBL1061091)
Kd
>10000±n/a nM
Citation
 Karaman, MW; Herrgard, S; Treiber, DK; Gallant, P; Atteridge, CE; Campbell, BT; Chan, KW; Ciceri, P; Davis, MI; Edeen, PT; Faraoni, R; Floyd, M; Hunt, JP; Lockhart, DJ; Milanov, ZV; Morrison, MJ; Pallares, G; Patel, HK; Pritchard, S; Wodicka, LM; Zarrinkar, PP A quantitative analysis of kinase inhibitor selectivity. Nat Biotechnol 26:127-32 (2008) [PubMed] Article
Karaman, MW; Herrgard, S; Treiber, DK; Gallant, P; Atteridge, CE; Campbell, BT; Chan, KW; Ciceri, P; Davis, MI; Edeen, PT; Faraoni, R; Floyd, M; Hunt, JP; Lockhart, DJ; Milanov, ZV; Morrison, MJ; Pallares, G; Patel, HK; Pritchard, S; Wodicka, LM; Zarrinkar, PP A quantitative analysis of kinase inhibitor selectivity. Nat Biotechnol 26:127-32 (2008) [PubMed] Article More Info.:
Target
Name:
Mitogen-activated protein kinase 14
Synonyms:
CSAID-binding protein | CSBP | CSBP1 | CSBP2 | CSPB1 | Cytokine suppressive anti-inflammatory drug-binding protein | MAP kinase 14 | MAP kinase MXI2 | MAP kinase p38 alpha | MAPK 14 | MAPK14 | MAX-interacting protein 2 | MK14_HUMAN | MXI2 | Mitogen-activated protein kinase p38 alpha | SAPK2A | Stress-activated protein kinase 2a | p38 MAP kinase alpha/beta
Type:
Serine/threonine-protein kinase
Mol. Mass.:
41286.76
Organism:
Homo sapiens (Human)
Description:
Q16539
Residue:
360
Sequence:
MSQERPTFYRQELNKTIWEVPERYQNLSPVGSGAYGSVCAAFDTKTGLRVAVKKLSRPFQSIIHAKRTYRELRLLKHMKHENVIGLLDVFTPARSLEEFNDVYLVTHLMGADLNNIVKCQKLTDDHVQFLIYQILRGLKYIHSADIIHRDLKPSNLAVNEDCELKILDFGLARHTDDEMTGYVATRWYRAPEIMLNWMHYNQTVDIWSVGCIMAELLTGRTLFPGTDHIDQLKLILRLVGTPGAELLKKISSESARNYIQSLTQMPKMNFANVFIGANPLAVDLLEKMLVLDSDKRITAAQALAHAYFAQYHDPDDEPVADPYDQSFESRDLLIDEWKSLTYDEVISFVPPPLDQEEMES
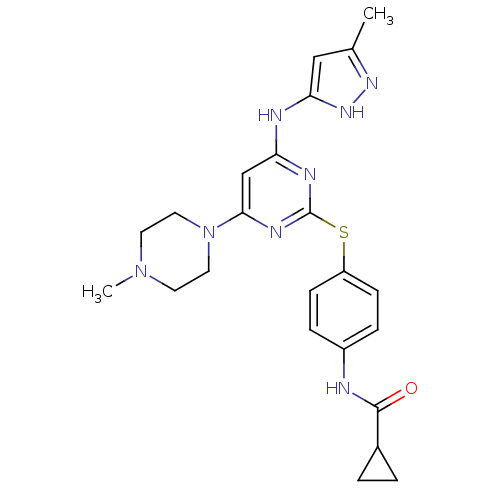
Inhibitor
Name:
BDBM13534
Synonyms:
CHEMBL572878 | N-[4-({4-[(3-methyl-1H-pyrazol-5-yl)amino]-6-(4-methylpiperazin-1-yl)pyrimidin-2-yl}sulfanyl)phenyl]cyclopropanecarboxamide | N-[4-[[4-(4-methylpiperazino)-6-[(5-methyl-1H-pyrazol-3-yl)amino]pyrimidin-2-yl]thio]phenyl]cyclopropanecarboxamide | VX-680 | VX680 | cyclopropane carboxylic acid {4-[4-(4-methyl-piperazin-1-yl)-6-(5-methyl-2H-pyrazol-3-ylamino)-pyrimidin-2ylsulphanyl]-phenyl}-amide
Type:
Small organic molecule
Emp. Form.:
C23H28N8OS
Mol. Mass.:
464.586
SMILES:
CN1CCN(CC1)c1cc(Nc2cc(C)n[nH]2)nc(Sc2ccc(NC(=O)C3CC3)cc2)n1