Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Peroxisome proliferator-activated receptor alpha
Ligand
BDBM33287
Substrate
BDBM33283
Meas. Tech.
Scintillation Proximity Assay (IC50) and PPAR alpha Transactivation Assay (EC50)
pH
7.2±n/a
Temperature
288.15±n/a K
EC50
8500±n/a nM
Comments
Efficacy=48% (The maximum efficacy of GW9578 was defined as 100%).
Citation
 Casimiro-Garcia, A; Bigge, CF; Davis, JA; Padalino, T; Pulaski, J; Ohren, JF; McConnell, P; Kane, CD; Royer, LJ; Stevens, KA; Auerbach, B; Collard, W; McGregor, C; Song, K Synthesis and evaluation of novel alpha-heteroaryl-phenylpropanoic acid derivatives as PPARalpha/gamma dual agonists. Bioorg Med Chem 17:7113-25 (2009) [PubMed] Article
Casimiro-Garcia, A; Bigge, CF; Davis, JA; Padalino, T; Pulaski, J; Ohren, JF; McConnell, P; Kane, CD; Royer, LJ; Stevens, KA; Auerbach, B; Collard, W; McGregor, C; Song, K Synthesis and evaluation of novel alpha-heteroaryl-phenylpropanoic acid derivatives as PPARalpha/gamma dual agonists. Bioorg Med Chem 17:7113-25 (2009) [PubMed] Article More Info.:
Target
Name:
Peroxisome proliferator-activated receptor alpha
Synonyms:
NR1C1 | Nuclear receptor subfamily 1 group C member 1 | PPAR | PPAR alpha/gamma | PPAR-alpha | PPARA | PPARA_HUMAN | Peroxisome Proliferator-Activated Receptor alpha | Peroxisome proliferator-activated receptor | Peroxisome proliferator-activated receptor alpha (PPAR alpha)
Type:
Enzyme
Mol. Mass.:
52222.08
Organism:
Homo sapiens (Human)
Description:
Q07869
Residue:
468
Sequence:
MVDTESPLCPLSPLEAGDLESPLSEEFLQEMGNIQEISQSIGEDSSGSFGFTEYQYLGSCPGSDGSVITDTLSPASSPSSVTYPVVPGSVDESPSGALNIECRICGDKASGYHYGVHACEGCKGFFRRTIRLKLVYDKCDRSCKIQKKNRNKCQYCRFHKCLSVGMSHNAIRFGRMPRSEKAKLKAEILTCEHDIEDSETADLKSLAKRIYEAYLKNFNMNKVKARVILSGKASNNPPFVIHDMETLCMAEKTLVAKLVANGIQNKEAEVRIFHCCQCTSVETVTELTEFAKAIPGFANLDLNDQVTLLKYGVYEAIFAMLSSVMNKDGMLVAYGNGFITREFLKSLRKPFCDIMEPKFDFAMKFNALELDDSDISLFVAAIICCGDRPGLLNVGHIEKMQEGIVHVLRLHLQSNHPDDIFLFPKLLQKMADLRQLVTEHAQLVQIIKKTESDAALHPLLQEIYRDMY
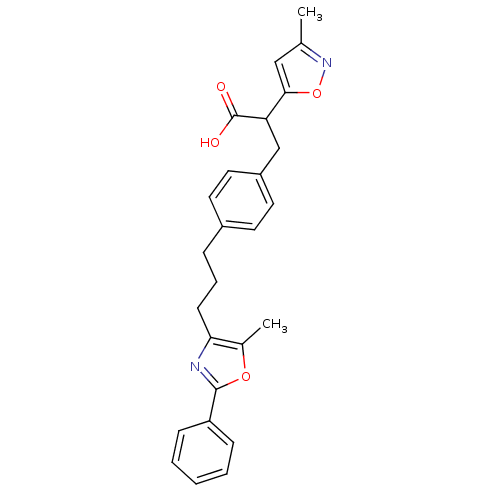
Inhibitor
Name:
BDBM33287
Synonyms:
phenylpropanoic acid derivative, 17c | racemic
Type:
Small organic molecule
Emp. Form.:
C26H26N2O4
Mol. Mass.:
430.4956
SMILES:
Cc1cc(on1)C(Cc1ccc(CCCc2nc(oc2C)-c2ccccc2)cc1)C(O)=O