Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Glutamate receptor 2
Ligand
BDBM50175229
Substrate
n/a
Meas. Tech.
ChEMBL_1704151 (CHEMBL4055384)
Ki
33000±n/a nM
Citation
 Espahbodinia, M; Ettari, R; Wen, W; Wu, A; Shen, YC; Niu, L; Grasso, S; Zappal�, M Development of novel N-3-bromoisoxazolin-5-yl substituted 2,3-benzodiazepines as noncompetitive AMPAR antagonists. Bioorg Med Chem 25:3631-3637 (2017) [PubMed] Article
Espahbodinia, M; Ettari, R; Wen, W; Wu, A; Shen, YC; Niu, L; Grasso, S; Zappal�, M Development of novel N-3-bromoisoxazolin-5-yl substituted 2,3-benzodiazepines as noncompetitive AMPAR antagonists. Bioorg Med Chem 25:3631-3637 (2017) [PubMed] Article More Info.:
Target
Name:
Glutamate receptor 2
Synonyms:
AMPA-selective glutamate receptor 2 | GLUR2 | GRIA2 | GRIA2_HUMAN | GluR-2 | GluR-B | GluR-K2 | Glutamate AMPA 2 | Glutamate receptor 2 | Glutamate receptor AMPA 1/2 | Glutamate receptor AMPA 2/3 | Glutamate receptor ionotropic AMPA | Glutamate receptor ionotropic, AMPA 2
Type:
Enzyme Catalytic Domain
Mol. Mass.:
98825.96
Organism:
Homo sapiens (Human)
Description:
Glutamate AMPA 2 GRIA2 HUMAN::P42262
Residue:
883
Sequence:
MQKIMHISVLLSPVLWGLIFGVSSNSIQIGGLFPRGADQEYSAFRVGMVQFSTSEFRLTPHIDNLEVANSFAVTNAFCSQFSRGVYAIFGFYDKKSVNTITSFCGTLHVSFITPSFPTDGTHPFVIQMRPDLKGALLSLIEYYQWDKFAYLYDSDRGLSTLQAVLDSAAEKKWQVTAINVGNINNDKKDEMYRSLFQDLELKKERRVILDCERDKVNDIVDQVITIGKHVKGYHYIIANLGFTDGDLLKIQFGGANVSGFQIVDYDDSLVSKFIERWSTLEEKEYPGAHTTTIKYTSALTYDAVQVMTEAFRNLRKQRIEISRRGNAGDCLANPAVPWGQGVEIERALKQVQVEGLSGNIKFDQNGKRINYTINIMELKTNGPRKIGYWSEVDKMVVTLTELPSGNDTSGLENKTVVVTTILESPYVMMKKNHEMLEGNERYEGYCVDLAAEIAKHCGFKYKLTIVGDGKYGARDADTKIWNGMVGELVYGKADIAIAPLTITLVREEVIDFSKPFMSLGISIMIKKPQKSKPGVFSFLDPLAYEIWMCIVFAYIGVSVVLFLVSRFSPYEWHTEEFEDGRETQSSESTNEFGIFNSLWFSLGAFMQQGCDISPRSLSGRIVGGVWWFFTLIIISSYTANLAAFLTVERMVSPIESAEDLSKQTEIAYGTLDSGSTKEFFRRSKIAVFDKMWTYMRSAEPSVFVRTTAEGVARVRKSKGKYAYLLESTMNEYIEQRKPCDTMKVGGNLDSKGYGIATPKGSSLRNAVNLAVLKLNEQGLLDKLKNKWWYDKGECGSGGGDSKEKTSALSLSNVAGVFYILVGGLGLAMLVALIEFCYKSRAEAKRMKVAKNAQNINPSSSQNSQNFATYKEGYNVYGIESVKI
Inhibitor
Name:
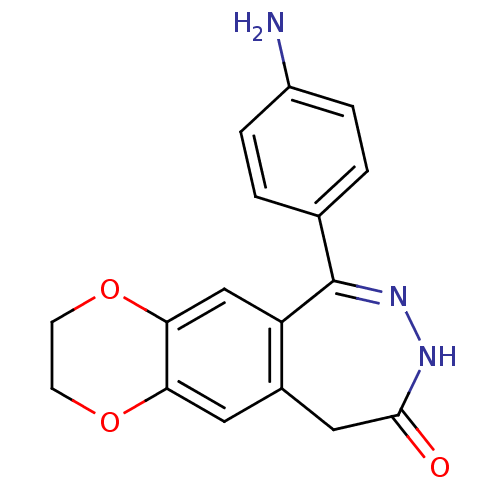
BDBM50175229
Synonyms:
6-(4-amino-phenyl)-2,3,8,10-tetrahydro-1,4-dioxa-7,8-diaza-cyclohepta[b]naphthalen-9-one | 6-(4-amino-phenyl)-2,3,8,10-tetrahyro-1,4-dioxa-7,8-diaza-cyclohepta[b]naphthalen-9-one | CHEMBL381673
Type:
Small organic molecule
Emp. Form.:
C17H15N3O3
Mol. Mass.:
309.3193
SMILES:
Nc1ccc(cc1)C1=NNC(=O)Cc2cc3OCCOc3cc12 |t:8|