Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
DNA topoisomerase 2-alpha
Ligand
BDBM21690
Substrate
n/a
Meas. Tech.
ChEMBL_1817315 (CHEMBL4316975)
IC50
153600±n/a nM
Citation
 Shi, C; Zhang, Y; Wang, T; Lu, W; Zhang, S; Guo, B; Chen, Q; Luo, C; Zhou, X; Yang, Y Design, Synthesis, and Biological Evaluation of Novel DNA Gyrase-Inhibiting Spiropyrimidinetriones as Potent Antibiotics for Treatment of Infections Caused by Multidrug-Resistant Gram-Positive Bacteria. J Med Chem 62:2950-2973 (2019) [PubMed] Article
Shi, C; Zhang, Y; Wang, T; Lu, W; Zhang, S; Guo, B; Chen, Q; Luo, C; Zhou, X; Yang, Y Design, Synthesis, and Biological Evaluation of Novel DNA Gyrase-Inhibiting Spiropyrimidinetriones as Potent Antibiotics for Treatment of Infections Caused by Multidrug-Resistant Gram-Positive Bacteria. J Med Chem 62:2950-2973 (2019) [PubMed] Article More Info.:
Target
Name:
DNA topoisomerase 2-alpha
Synonyms:
DNA topoisomerase 2-alpha | DNA topoisomerase II | DNA topoisomerase II (Topo II) | DNA topoisomerase II alpha | DNA topoisomerase II, alpha isozyme | TOP2 | TOP2A | TOP2A_HUMAN | Topoisomerase I/II | Topoisomerase II alpha (HuTopoIIα)
Type:
Protein
Mol. Mass.:
174415.30
Organism:
Homo sapiens (Human)
Description:
P11388
Residue:
1531
Sequence:
MEVSPLQPVNENMQVNKIKKNEDAKKRLSVERIYQKKTQLEHILLRPDTYIGSVELVTQQMWVYDEDVGINYREVTFVPGLYKIFDEILVNAADNKQRDPKMSCIRVTIDPENNLISIWNNGKGIPVVEHKVEKMYVPALIFGQLLTSSNYDDDEKKVTGGRNGYGAKLCNIFSTKFTVETASREYKKMFKQTWMDNMGRAGEMELKPFNGEDYTCITFQPDLSKFKMQSLDKDIVALMVRRAYDIAGSTKDVKVFLNGNKLPVKGFRSYVDMYLKDKLDETGNSLKVIHEQVNHRWEVCLTMSEKGFQQISFVNSIATSKGGRHVDYVADQIVTKLVDVVKKKNKGGVAVKAHQVKNHMWIFVNALIENPTFDSQTKENMTLQPKSFGSTCQLSEKFIKAAIGCGIVESILNWVKFKAQVQLNKKCSAVKHNRIKGIPKLDDANDAGGRNSTECTLILTEGDSAKTLAVSGLGVVGRDKYGVFPLRGKILNVREASHKQIMENAEINNIIKIVGLQYKKNYEDEDSLKTLRYGKIMIMTDQDQDGSHIKGLLINFIHHNWPSLLRHRFLEEFITPIVKVSKNKQEMAFYSLPEFEEWKSSTPNHKKWKVKYYKGLGTSTSKEAKEYFADMKRHRIQFKYSGPEDDAAISLAFSKKQIDDRKEWLTNFMEDRRQRKLLGLPEDYLYGQTTTYLTYNDFINKELILFSNSDNERSIPSMVDGLKPGQRKVLFTCFKRNDKREVKVAQLAGSVAEMSSYHHGEMSLMMTIINLAQNFVGSNNLNLLQPIGQFGTRLHGGKDSASPRYIFTMLSSLARLLFPPKDDHTLKFLYDDNQRVEPEWYIPIIPMVLINGAEGIGTGWSCKIPNFDVREIVNNIRRLMDGEEPLPMLPSYKNFKGTIEELAPNQYVISGEVAILNSTTIEISELPVRTWTQTYKEQVLEPMLNGTEKTPPLITDYREYHTDTTVKFVVKMTEEKLAEAERVGLHKVFKLQTSLTCNSMVLFDHVGCLKKYDTVLDILRDFFELRLKYYGLRKEWLLGMLGAESAKLNNQARFILEKIDGKIIIENKPKKELIKVLIQRGYDSDPVKAWKEAQQKVPDEEENEESDNEKETEKSDSVTDSGPTFNYLLDMPLWYLTKEKKDELCRLRNEKEQELDTLKRKSPSDLWKEDLATFIEELEAVEAKEKQDEQVGLPGKGGKAKGKKTQMAEVLPSPRGQRVIPRITIEMKAEAEKKNKKKIKNENTEGSPQEDGVELEGLKQRLEKKQKREPGTKTKKQTTLAFKPIKKGKKRNPWSDSESDRSSDESNFDVPPRETEPRRAATKTKFTMDLDSDEDFSDFDEKTDDEDFVPSDASPPKTKTSPKLSNKELKPQKSVVSDLEADDVKGSVPLSSSPPATHFPDETEITNPVPKKNVTVKKTAAKSQSSTSTTGAKKRAAPKGTKRDPALNSGVSQKPDPAKTKNRRKRKPSTSDDSDSNFEKIVSKAVTSKKSKGESDDFHMDFDSAVAPRAKSVRAKKPIKYLEESDEDDLF
Inhibitor
Name:
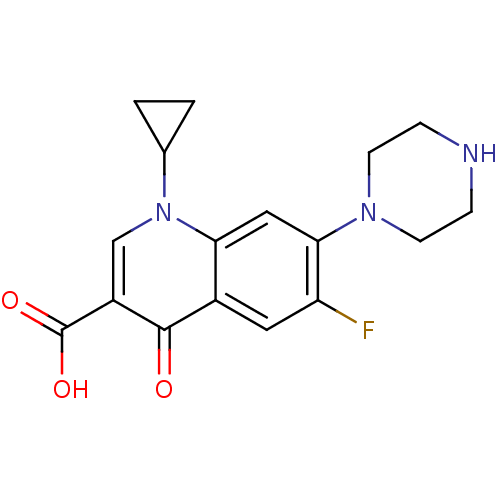
BDBM21690
Synonyms:
1-cyclopropyl-6-fluoro-4-oxo-7-(piperazin-1-yl)-1,4-dihydroquinoline-3-carboxylic acid | 1-cyclopropyl-6-fluoro-4-oxo-7-piperazin-1-yl-1,4-dihydroquinoline-3-carboxylic acid | Bay 09867 | CHEMBL8 | Ciprinol | Cipro | Ciprofloxacin | US11590142, Compound Ciprofloxacin | US9138393, Ciprofloxacin HCl | US9144538, Ciprofloxacin HCl
Type:
Small organic molecule
Emp. Form.:
C17H18FN3O3
Mol. Mass.:
331.3415
SMILES:
OC(=O)c1cn(C2CC2)c2cc(N3CCNCC3)c(F)cc2c1=O