Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Prothrombin
Ligand
BDBM50071575
Substrate
n/a
Meas. Tech.
ChEBML_208893
Ki
0.200000±n/a nM
Citation
 Ogbu, CO; Qabar, MN; Boatman, PD; Urban, J; Meara, JP; Ferguson, MD; Tulinsky, J; Lum, C; Babu, S; Blaskovich, MA; Nakanishi, H; Ruan, F; Cao, B; Minarik, R; Little, T; Nelson, S; Nguyen, M; Gall, A; Kahn, M Highly efficient and versatile synthesis of libraries of constrained beta-strand mimetics. Bioorg Med Chem Lett 8:2321-6 (1999) [PubMed] Article
Ogbu, CO; Qabar, MN; Boatman, PD; Urban, J; Meara, JP; Ferguson, MD; Tulinsky, J; Lum, C; Babu, S; Blaskovich, MA; Nakanishi, H; Ruan, F; Cao, B; Minarik, R; Little, T; Nelson, S; Nguyen, M; Gall, A; Kahn, M Highly efficient and versatile synthesis of libraries of constrained beta-strand mimetics. Bioorg Med Chem Lett 8:2321-6 (1999) [PubMed] Article More Info.:
Target
Name:
Prothrombin
Synonyms:
Activation peptide fragment 1 | Activation peptide fragment 2 | Coagulation factor II | F2 | Prothrombin precursor | THRB_HUMAN | Thrombin heavy chain | Thrombin light chain
Type:
Protein
Mol. Mass.:
70029.57
Organism:
Homo sapiens (Human)
Description:
P00734
Residue:
622
Sequence:
MAHVRGLQLPGCLALAALCSLVHSQHVFLAPQQARSLLQRVRRANTFLEEVRKGNLERECVEETCSYEEAFEALESSTATDVFWAKYTACETARTPRDKLAACLEGNCAEGLGTNYRGHVNITRSGIECQLWRSRYPHKPEINSTTHPGADLQENFCRNPDSSTTGPWCYTTDPTVRRQECSIPVCGQDQVTVAMTPRSEGSSVNLSPPLEQCVPDRGQQYQGRLAVTTHGLPCLAWASAQAKALSKHQDFNSAVQLVENFCRNPDGDEEGVWCYVAGKPGDFGYCDLNYCEEAVEEETGDGLDEDSDRAIEGRTATSEYQTFFNPRTFGSGEADCGLRPLFEKKSLEDKTERELLESYIDGRIVEGSDAEIGMSPWQVMLFRKSPQELLCGASLISDRWVLTAAHCLLYPPWDKNFTENDLLVRIGKHSRTRYERNIEKISMLEKIYIHPRYNWRENLDRDIALMKLKKPVAFSDYIHPVCLPDRETAASLLQAGYKGRVTGWGNLKETWTANVGKGQPSVLQVVNLPIVERPVCKDSTRIRITDNMFCAGYKPDEGKRGDACEGDSGGPFVMKSPFNNRWYQMGIVSWGEGCDRDGKYGFYTHVFRLKKWIQKVIDQFGE
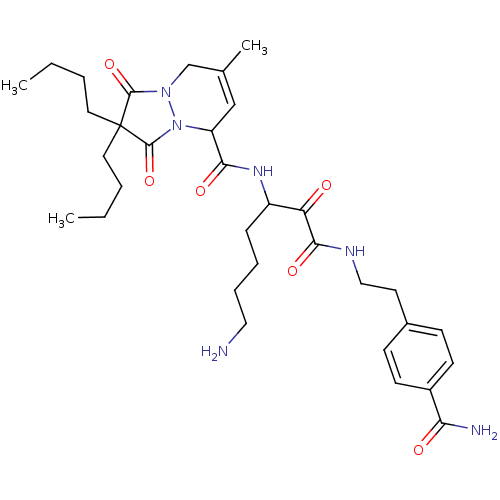
Inhibitor
Name:
BDBM50071575
Synonyms:
2,2-Dibutyl-7-methyl-1,3-dioxo-2,3,5,8-tetrahydro-1H-pyrazolo[1,2-a]pyridazine-5-carboxylic acid {5-amino-1-[2-(4-carbamoyl-phenyl)-ethylaminooxalyl]-pentyl}-amide | CHEMBL307587
Type:
Small organic molecule
Emp. Form.:
C33H48N6O6
Mol. Mass.:
624.7708
SMILES:
CCCCC1(CCCC)C(=O)N2CC(C)=CC(N2C1=O)C(=O)NC(CCCCN)C(=O)C(=O)NCCc1ccc(cc1)C(N)=O |c:14|