Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Indoleamine 2,3-dioxygenase 1
Ligand
BDBM50559677
Substrate
n/a
Meas. Tech.
ChEMBL_2069931 (CHEMBL4725184)
IC50
14±n/a nM
Citation
 Zhang, L; Cherney, EC; Zhu, X; Lin, TA; Gullo-Brown, J; Maley, D; Johnston-Allegretto, K; Kopcho, L; Fereshteh, M; Huang, C; Li, X; Traeger, SC; Dhar, G; Anandam, A; Mahankali, S; Padmanabhan, S; Rajanna, P; Murali, V; Mariappan, T; Borzilleri, R; Vite, G; Hunt, JT; Balog, A Discovery of Imidazopyridines as Potent Inhibitors of Indoleamine 2,3-Dioxygenase 1 for Cancer Immunotherapy. ACS Med Chem Lett 12:494-501 (2021) [PubMed] Article
Zhang, L; Cherney, EC; Zhu, X; Lin, TA; Gullo-Brown, J; Maley, D; Johnston-Allegretto, K; Kopcho, L; Fereshteh, M; Huang, C; Li, X; Traeger, SC; Dhar, G; Anandam, A; Mahankali, S; Padmanabhan, S; Rajanna, P; Murali, V; Mariappan, T; Borzilleri, R; Vite, G; Hunt, JT; Balog, A Discovery of Imidazopyridines as Potent Inhibitors of Indoleamine 2,3-Dioxygenase 1 for Cancer Immunotherapy. ACS Med Chem Lett 12:494-501 (2021) [PubMed] Article More Info.:
Target
Name:
Indoleamine 2,3-dioxygenase 1
Synonyms:
I23O1_HUMAN | IDO | IDO-1 | IDO1 | INDO | Indoleamine 2,3-Dioxygenasae (IDO) | Indoleamine 2,3-dioxygenase | Indoleamine-pyrrole 2,3-dioxygenase
Type:
Enzyme
Mol. Mass.:
45330.80
Organism:
Homo sapiens (Human)
Description:
P14902
Residue:
403
Sequence:
MAHAMENSWTISKEYHIDEEVGFALPNPQENLPDFYNDWMFIAKHLPDLIESGQLRERVEKLNMLSIDHLTDHKSQRLARLVLGCITMAYVWGKGHGDVRKVLPRNIAVPYCQLSKKLELPPILVYADCVLANWKKKDPNKPLTYENMDVLFSFRDGDCSKGFFLVSLLVEIAAASAIKVIPTVFKAMQMQERDTLLKALLEIASCLEKALQVFHQIHDHVNPKAFFSVLRIYLSGWKGNPQLSDGLVYEGFWEDPKEFAGGSAGQSSVFQCFDVLLGIQQTAGGGHAAQFLQDMRRYMPPAHRNFLCSLESNPSVREFVLSKGDAGLREAYDACVKALVSLRSYHLQIVTKYILIPASQQPKENKTSEDPSKLEAKGTGGTDLMNFLKTVRSTTEKSLLKEG
Inhibitor
Name:
BDBM50559677
Synonyms:
CHEMBL4746203
Type:
Small organic molecule
Emp. Form.:
C27H28F2N4O2
Mol. Mass.:
478.5336
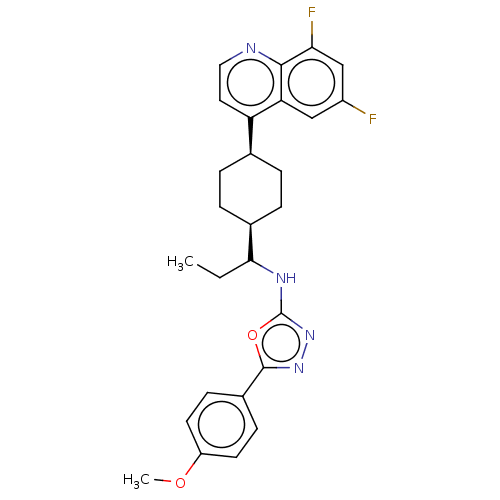
SMILES:
CCC(Nc1nnc(o1)-c1ccc(OC)cc1)[C@H]1CC[C@H](CC1)c1ccnc2c(F)cc(F)cc12 |r,wU:20.25,17.18,(3.44,-6.79,;4.77,-6.02,;6.11,-6.79,;7.45,-6.02,;8.78,-6.79,;9.11,-8.29,;10.65,-8.46,;11.27,-7.08,;10.11,-6.02,;12.76,-6.68,;13.16,-5.2,;14.65,-4.8,;15.74,-5.89,;17.22,-5.49,;17.62,-4,;15.34,-7.37,;13.86,-7.78,;6.11,-8.33,;7.45,-9.1,;7.45,-10.65,;6.11,-11.42,;4.77,-10.65,;4.77,-9.1,;6.11,-12.96,;4.78,-13.73,;4.78,-15.27,;6.12,-16.05,;7.44,-15.27,;8.78,-16.03,;8.79,-17.57,;10.1,-15.25,;10.1,-13.72,;11.43,-12.96,;8.76,-12.96,;7.45,-13.73,)|