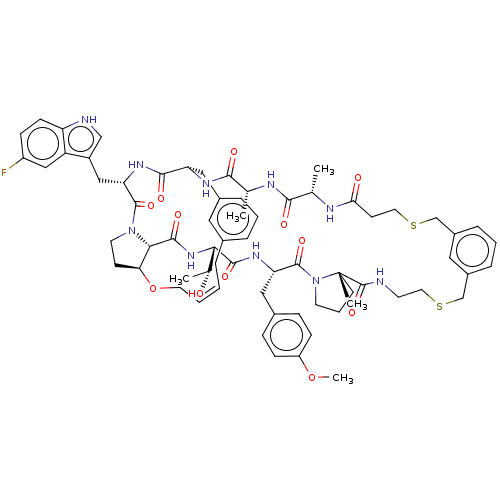
Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Proprotein convertase subtilisin/kexin type 9
Ligand
BDBM50581538
Substrate
n/a
Meas. Tech.
ChEMBL_2151332 (CHEMBL5035794)
Ki
4.7±n/a nM
Citation
 Tucker, TJ; Embrey, MW; Alleyne, C; Amin, RP; Bass, A; Bhatt, B; Bianchi, E; Branca, D; Bueters, T; Buist, N; Ha, SN; Hafey, M; He, H; Higgins, J; Johns, DG; Kerekes, AD; Koeplinger, KA; Kuethe, JT; Li, N; Murphy, B; Orth, P; Salowe, S; Shahripour, A; Tracy, R; Wang, W; Wu, C; Xiong, Y; Zokian, HJ; Wood, HB; Walji, A A Series of Novel, Highly Potent, and Orally Bioavailable Next-Generation Tricyclic Peptide PCSK9 Inhibitors. J Med Chem 64:16770-16800 (2021) [PubMed] Article
Tucker, TJ; Embrey, MW; Alleyne, C; Amin, RP; Bass, A; Bhatt, B; Bianchi, E; Branca, D; Bueters, T; Buist, N; Ha, SN; Hafey, M; He, H; Higgins, J; Johns, DG; Kerekes, AD; Koeplinger, KA; Kuethe, JT; Li, N; Murphy, B; Orth, P; Salowe, S; Shahripour, A; Tracy, R; Wang, W; Wu, C; Xiong, Y; Zokian, HJ; Wood, HB; Walji, A A Series of Novel, Highly Potent, and Orally Bioavailable Next-Generation Tricyclic Peptide PCSK9 Inhibitors. J Med Chem 64:16770-16800 (2021) [PubMed] Article More Info.:
Target
Name:
Proprotein convertase subtilisin/kexin type 9
Synonyms:
NARC-1 | NARC1 | Neural apoptosis-regulated convertase 1 | PC9 | PCSK9 | PCSK9_HUMAN | Proprotein convertase 9 | Proprotein convertase subtilisin/kexin type 9 | Proprotein convertase subtilisin/kexin type 9 (PCSK9) | Subtilisin/kexin type 9 | Subtilisin/kexin-like protease PC9
Type:
Enzyme
Mol. Mass.:
74286.93
Organism:
Homo sapiens (Human)
Description:
Q8NBP7
Residue:
692
Sequence:
MGTVSSRRSWWPLPLLLLLLLLLGPAGARAQEDEDGDYEELVLALRSEEDGLAEAPEHGTTATFHRCAKDPWRLPGTYVVVLKEETHLSQSERTARRLQAQAARRGYLTKILHVFHGLLPGFLVKMSGDLLELALKLPHVDYIEEDSSVFAQSIPWNLERITPPRYRADEYQPPDGGSLVEVYLLDTSIQSDHREIEGRVMVTDFENVPEEDGTRFHRQASKCDSHGTHLAGVVSGRDAGVAKGASMRSLRVLNCQGKGTVSGTLIGLEFIRKSQLVQPVGPLVVLLPLAGGYSRVLNAACQRLARAGVVLVTAAGNFRDDACLYSPASAPEVITVGATNAQDQPVTLGTLGTNFGRCVDLFAPGEDIIGASSDCSTCFVSQSGTSQAAAHVAGIAAMMLSAEPELTLAELRQRLIHFSAKDVINEAWFPEDQRVLTPNLVAALPPSTHGAGWQLFCRTVWSAHSGPTRMATAVARCAPDEELLSCSSFSRSGKRRGERMEAQGGKLVCRAHNAFGGEGVYAIARCCLLPQANCSVHTAPPAEASMGTRVHCHQQGHVLTGCSSHWEVEDLGTHKPPVLRPRGQPNQCVGHREASIHASCCHAPGLECKVKEHGIPAPQEQVTVACEEGWTLTGCSALPGTSHVLGAYAVDNTCVVRSRDVSTTGSTSEGAVTAVAICCRSRHLAQASQELQ
Inhibitor
Name:
BDBM50581538
Synonyms:
CHEMBL5081995
Type:
Small organic molecule
Emp. Form.:
C68H83FN10O12S2
Mol. Mass.:
1315.575
SMILES:
COc1ccc(C[C@@H]2NC(=O)[C@@H](NC(=O)[C@@H]3[C@@H]4CCN3C(=O)[C@H](Cc3c[nH]c5ccc(F)cc35)NC(=O)[C@H](Cc3cccc(C\C=C/CO4)c3)NC(=O)[C@@H](C)NC(=O)[C@H](C)NC(=O)CCSCc3cccc(CSCCNC(=O)[C@]4(C)CCCN4C2=O)c3)[C@@H](C)O)cc1 |r,c:48|