Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Glycogen phosphorylase, liver form
Ligand
BDBM50158278
Substrate
n/a
Meas. Tech.
ChEMBL_304931 (CHEMBL826961)
IC50
12±n/a nM
Citation
 Wright, SW; Rath, VL; Genereux, PE; Hageman, DL; Levy, CB; McClure, LD; McCoid, SC; McPherson, RK; Schelhorn, TM; Wilder, DE; Zavadoski, WJ; Gibbs, EM; Treadway, JL 5-Chloroindoloyl glycine amide inhibitors of glycogen phosphorylase: synthesis, in vitro, in vivo, and X-ray crystallographic characterization. Bioorg Med Chem Lett 15:459-65 (2004) [PubMed] Article
Wright, SW; Rath, VL; Genereux, PE; Hageman, DL; Levy, CB; McClure, LD; McCoid, SC; McPherson, RK; Schelhorn, TM; Wilder, DE; Zavadoski, WJ; Gibbs, EM; Treadway, JL 5-Chloroindoloyl glycine amide inhibitors of glycogen phosphorylase: synthesis, in vitro, in vivo, and X-ray crystallographic characterization. Bioorg Med Chem Lett 15:459-65 (2004) [PubMed] Article More Info.:
Target
Name:
Glycogen phosphorylase, liver form
Synonyms:
Glycogen Phosphorylase (PYGL) | Glycogen Phosphorylase, liver form | Liver glycogen phosphorylase | PYGL | PYGL_HUMAN
Type:
Homodimer
Mol. Mass.:
97153.98
Organism:
Homo sapiens (Human)
Description:
Dimers associate into a tetramer to form the enzymatically active phosphorylase A.
Residue:
847
Sequence:
MAKPLTDQEKRRQISIRGIVGVENVAELKKSFNRHLHFTLVKDRNVATTRDYYFALAHTVRDHLVGRWIRTQQHYYDKCPKRVYYLSLEFYMGRTLQNTMINLGLQNACDEAIYQLGLDIEELEEIEEDAGLGNGGLGRLAACFLDSMATLGLAAYGYGIRYEYGIFNQKIRDGWQVEEADDWLRYGNPWEKSRPEFMLPVHFYGKVEHTNTGTKWIDTQVVLALPYDTPVPGYMNNTVNTMRLWSARAPNDFNLRDFNVGDYIQAVLDRNLAENISRVLYPNDNFFEGKELRLKQEYFVVAATLQDIIRRFKASKFGSTRGAGTVFDAFPDQVAIQLNDTHPALAIPELMRIFVDIEKLPWSKAWELTQKTFAYTNHTVLPEALERWPVDLVEKLLPRHLEIIYEINQKHLDRIVALFPKDVDRLRRMSLIEEEGSKRINMAHLCIVGSHAVNGVAKIHSDIVKTKVFKDFSELEPDKFQNKTNGITPRRWLLLCNPGLAELIAEKIGEDYVKDLSQLTKLHSFLGDDVFLRELAKVKQENKLKFSQFLETEYKVKINPSSMFDVQVKRIHEYKRQLLNCLHVITMYNRIKKDPKKLFVPRTVIIGGKAAPGYHMAKMIIKLITSVADVVNNDPMVGSKLKVIFLENYRVSLAEKVIPATDLSEQISTAGTEASGTGNMKFMLNGALTIGTMDGANVEMAEEAGEENLFIFGMRIDDVAALDKKGYEAKEYYEALPELKLVIDQIDNGFFSPKQPDLFKDIINMLFYHDRFKVFADYEAYVKCQDKVSQLYMNPKAWNTMVLKNIAASGKFSSDRTIKEYAQNIWNVEPSDLKISLSNESNKVNGN
Inhibitor
Name:
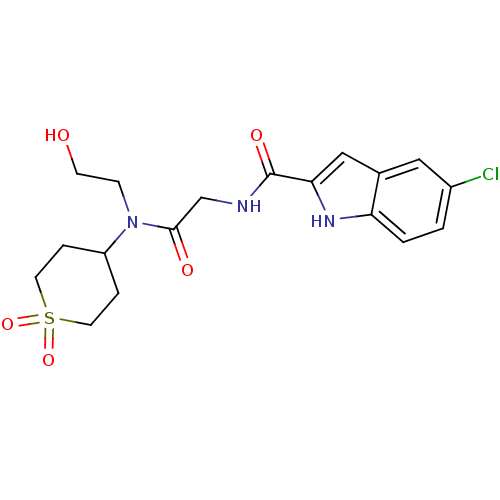
BDBM50158278
Synonyms:
5-Chloro-1H-indole-2-carboxylic acid {[(1,1-dioxo-hexahydro-1lambda*6*-thiopyran-4-yl)-(2-hydroxy-ethyl)-carbamoyl]-methyl}-amide | CHEMBL179485
Type:
Small organic molecule
Emp. Form.:
C18H22ClN3O5S
Mol. Mass.:
427.902
SMILES:
OCCN(C1CCS(=O)(=O)CC1)C(=O)CNC(=O)c1cc2cc(Cl)ccc2[nH]1