Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Muscarinic acetylcholine receptor M3
Ligand
BDBM50208052
Substrate
n/a
Meas. Tech.
ChEMBL_429188 (CHEMBL913822)
Ki
612±n/a nM
Citation
 Peretto, I; Fossati, C; Giardina, GA; Giardini, A; Guala, M; La Porta, E; Petrillo, P; Radaelli, S; Radice, L; Raveglia, LF; Santoro, E; Scudellaro, R; Scarpitta, F; Cerri, A; Menegon, S; Dondio, GM; Rizzi, A; Armani, E; Amari, G; Civelli, M; Villetti, G; Patacchini, R; Bergamaschi, M; Bassani, F; Delcanale, M; Imbimbo, BP Discovery of diaryl imidazolidin-2-one derivatives, a novel class of muscarinic M3 selective antagonists (Part 2). J Med Chem 50:1693-7 (2007) [PubMed] Article
Peretto, I; Fossati, C; Giardina, GA; Giardini, A; Guala, M; La Porta, E; Petrillo, P; Radaelli, S; Radice, L; Raveglia, LF; Santoro, E; Scudellaro, R; Scarpitta, F; Cerri, A; Menegon, S; Dondio, GM; Rizzi, A; Armani, E; Amari, G; Civelli, M; Villetti, G; Patacchini, R; Bergamaschi, M; Bassani, F; Delcanale, M; Imbimbo, BP Discovery of diaryl imidazolidin-2-one derivatives, a novel class of muscarinic M3 selective antagonists (Part 2). J Med Chem 50:1693-7 (2007) [PubMed] Article More Info.:
Target
Name:
Muscarinic acetylcholine receptor M3
Synonyms:
ACM3_HUMAN | CHRM3 | Cholinergic, muscarinic M3 | Muscarinic Receptors M3 | Muscarinic receptor M3 | RecName: Full=Muscarinic acetylcholine receptor M3
Type:
Enzyme
Mol. Mass.:
66151.03
Organism:
Homo sapiens (Human)
Description:
P20309
Residue:
590
Sequence:
MTLHNNSTTSPLFPNISSSWIHSPSDAGLPPGTVTHFGSYNVSRAAGNFSSPDGTTDDPLGGHTVWQVVFIAFLTGILALVTIIGNILVIVSFKVNKQLKTVNNYFLLSLACADLIIGVISMNLFTTYIIMNRWALGNLACDLWLAIDYVASNASVMNLLVISFDRYFSITRPLTYRAKRTTKRAGVMIGLAWVISFVLWAPAILFWQYFVGKRTVPPGECFIQFLSEPTITFGTAIAAFYMPVTIMTILYWRIYKETEKRTKELAGLQASGTEAETENFVHPTGSSRSCSSYELQQQSMKRSNRRKYGRCHFWFTTKSWKPSSEQMDQDHSSSDSWNNNDAAASLENSASSDEEDIGSETRAIYSIVLKLPGHSTILNSTKLPSSDNLQVPEEELGMVDLERKADKLQAQKSVDDGGSFPKSFSKLPIQLESAVDTAKTSDVNSSVGKSTATLPLSFKEATLAKRFALKTRSQITKRKRMSLVKEKKAAQTLSAILLAFIITWTPYNIMVLVNTFCDSCIPKTFWNLGYWLCYINSTVNPVCYALCNKTFRTTFKMLLLCQCDKKKRRKQQYQQRQSVIFHKRAPEQAL
Inhibitor
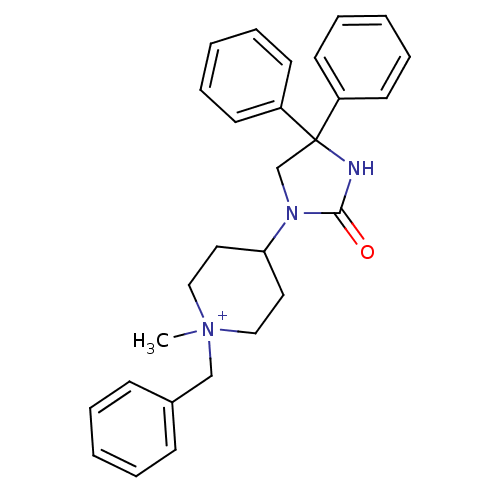
Name:
BDBM50208052
Synonyms:
1-benzyl-1-methyl-4-(2-oxo-4,4-diphenyl-imidazolidin-1-yl)-piperidinium | CHEMBL222425
Type:
Small organic molecule
Emp. Form.:
C28H32N3O
Mol. Mass.:
426.5726
SMILES:
C[N+]1(Cc2ccccc2)CCC(CC1)N1CC(NC1=O)(c1ccccc1)c1ccccc1 |(1.29,2.89,;.49,4.2,;1.57,5.3,;3.07,4.97,;3.54,3.51,;5.04,3.17,;6.08,4.31,;5.6,5.78,;4.1,6.11,;.16,2.7,;-1.3,2.23,;-2.45,3.26,;-2.12,4.77,;-.65,5.24,;-3.91,2.78,;-4.39,1.32,;-5.93,1.32,;-6.4,2.78,;-5.16,3.69,;-5.16,5.23,;-5.22,-.04,;-6.06,-1.34,;-5.35,-2.7,;-3.81,-2.77,;-2.98,-1.46,;-3.7,-.1,;-7.48,1.32,;-8.25,2.67,;-9.79,2.67,;-10.57,1.33,;-9.79,-.02,;-8.24,-.01,)|