Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Cytochrome P450 3A4
Ligand
BDBM50314501
Substrate
n/a
Meas. Tech.
ChEMBL_624608 (CHEMBL1111568)
Ki
70±n/a nM
Citation
 McNulty, J; Nair, JJ; Singh, M; Crankshaw, DJ; Holloway, AC Potent and selective inhibition of human cytochrome P450 3A4 by seco-pancratistatin structural analogs. Bioorg Med Chem Lett 20:2335-9 (2010) [PubMed] Article
McNulty, J; Nair, JJ; Singh, M; Crankshaw, DJ; Holloway, AC Potent and selective inhibition of human cytochrome P450 3A4 by seco-pancratistatin structural analogs. Bioorg Med Chem Lett 20:2335-9 (2010) [PubMed] Article More Info.:
Target
Name:
Cytochrome P450 3A4
Synonyms:
Albendazole monooxygenase | Albendazole sulfoxidase | CP3A4_HUMAN | CYP3A3 | CYP3A4 | CYPIIIA3 | CYPIIIA4 | Cytochrome P450 3A3 | Cytochrome P450 3A4 (CYP3A4) | Cytochrome P450 HLp | Nifedipine oxidase | Quinine 3-monooxygenase | Taurochenodeoxycholate 6-alpha-hydroxylase
Type:
Enzyme
Mol. Mass.:
57349.57
Organism:
Homo sapiens (Human)
Description:
n/a
Residue:
503
Sequence:
MALIPDLAMETWLLLAVSLVLLYLYGTHSHGLFKKLGIPGPTPLPFLGNILSYHKGFCMFDMECHKKYGKVWGFYDGQQPVLAITDPDMIKTVLVKECYSVFTNRRPFGPVGFMKSAISIAEDEEWKRLRSLLSPTFTSGKLKEMVPIIAQYGDVLVRNLRREAETGKPVTLKDVFGAYSMDVITSTSFGVNIDSLNNPQDPFVENTKKLLRFDFLDPFFLSITVFPFLIPILEVLNICVFPREVTNFLRKSVKRMKESRLEDTQKHRVDFLQLMIDSQNSKETESHKALSDLELVAQSIIFIFAGYETTSSVLSFIMYELATHPDVQQKLQEEIDAVLPNKAPPTYDTVLQMEYLDMVVNETLRLFPIAMRLERVCKKDVEINGMFIPKGVVVMIPSYALHRDPKYWTEPEKFLPERFSKKNKDNIDPYIYTPFGSGPRNCIGMRFALMNMKLALIRVLQNFSFKPCKETQIPLKLSLGGLLQPEKPVVLKVESRDGTVSGA
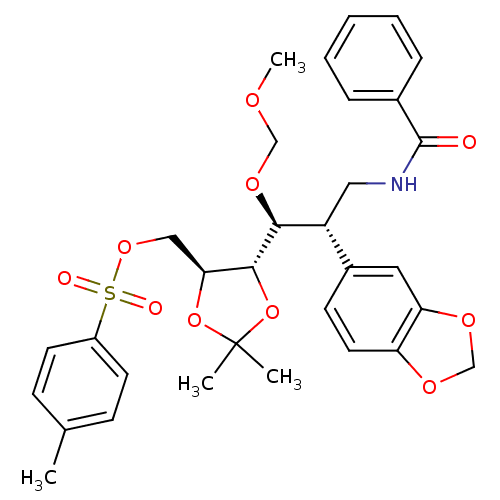
Inhibitor
Name:
BDBM50314501
Synonyms:
((4S,5S)-5-((1S,2S)-3-benzamido-2-(benzo[d][1,3]dioxol-5-yl)-1-(methoxymethoxy)propyl)-2,2-dimethyl-1,3-dioxolan-4-yl)methyl 4-methylbenzenesulfonate | CHEMBL1089957
Type:
Small organic molecule
Emp. Form.:
C32H37NO10S
Mol. Mass.:
627.702
SMILES:
COCO[C@@H]([C@H](CNC(=O)c1ccccc1)c1ccc2OCOc2c1)[C@@H]1OC(C)(C)O[C@H]1COS(=O)(=O)c1ccc(C)cc1 |r|