Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Potassium voltage-gated channel subfamily H member 2
Ligand
BDBM50326136
Substrate
n/a
Meas. Tech.
ChEMBL_657972 (CHEMBL1248651)
Ki
2511.89±n/a nM
Citation
 Bromidge, SM; Arban, R; Bertani, B; Bison, S; Borriello, M; Cavanni, P; Dal Forno, G; Di-Fabio, R; Donati, D; Fontana, S; Gianotti, M; Gordon, LJ; Granci, E; Leslie, CP; Moccia, L; Pasquarello, A; Sartori, I; Sava, A; Watson, JM; Worby, A; Zonzini, L; Zucchelli, V Design and synthesis of novel tricyclic benzoxazines as potent 5-HT(1A/B/D) receptor antagonists leading to the discovery of 6-{2-[4-(2-methyl-5-quinolinyl)-1-piperazinyl]ethyl}-4H-imidazo[5,1-c][1,4]benzoxazine-3-carboxamide (GSK588045). J Med Chem 53:5827-43 (2010) [PubMed] Article
Bromidge, SM; Arban, R; Bertani, B; Bison, S; Borriello, M; Cavanni, P; Dal Forno, G; Di-Fabio, R; Donati, D; Fontana, S; Gianotti, M; Gordon, LJ; Granci, E; Leslie, CP; Moccia, L; Pasquarello, A; Sartori, I; Sava, A; Watson, JM; Worby, A; Zonzini, L; Zucchelli, V Design and synthesis of novel tricyclic benzoxazines as potent 5-HT(1A/B/D) receptor antagonists leading to the discovery of 6-{2-[4-(2-methyl-5-quinolinyl)-1-piperazinyl]ethyl}-4H-imidazo[5,1-c][1,4]benzoxazine-3-carboxamide (GSK588045). J Med Chem 53:5827-43 (2010) [PubMed] Article More Info.:
Target
Name:
Potassium voltage-gated channel subfamily H member 2
Synonyms:
1,3-beta-glucan synthase component GLS2 | Cytochrome P450 3A4 | ERG | ERG1 | Eag-related protein 1 | Ether a-go-go related gene potassium channel (hERG) | Ether-a-go-go-related gene (HERG) | Ether-a-go-go-related gene potassium channel (hERG) | Ether-a-go-go-related gene potassium channel 1 | Ether-a-go-go-related gene potassium channel 1 (HERG) | Ether-a-go-go-related gene potassium channel 1 (hERG1) | Ether-a-go-go-related protein (hERG) | Ether-a-go-go-related protein 1 | Ether-a-go-go-related protein 1 (HERG) | H-ERG | HERG | KCNH2 | KCNH2_HUMAN | Potassium voltage-gated channel subfamily H member 2 (hERG) | Transcriptional regulator ERG | Voltage-gated potassium channel subunit Kv11.1 | eag homolog | hERG Potassium Channel 1 | putative potassium channel subunit
Type:
Multi-pass membrane protein
Mol. Mass.:
126672.65
Organism:
Homo sapiens (Human)
Description:
Q12809
Residue:
1159
Sequence:
MPVRRGHVAPQNTFLDTIIRKFEGQSRKFIIANARVENCAVIYCNDGFCELCGYSRAEVMQRPCTCDFLHGPRTQRRAAAQIAQALLGAEERKVEIAFYRKDGSCFLCLVDVVPVKNEDGAVIMFILNFEVVMEKDMVGSPAHDTNHRGPPTSWLAPGRAKTFRLKLPALLALTARESSVRSGGAGGAGAPGAVVVDVDLTPAAPSSESLALDEVTAMDNHVAGLGPAEERRALVGPGSPPRSAPGQLPSPRAHSLNPDASGSSCSLARTRSRESCASVRRASSADDIEAMRAGVLPPPPRHASTGAMHPLRSGLLNSTSDSDLVRYRTISKIPQITLNFVDLKGDPFLASPTSDREIIAPKIKERTHNVTEKVTQVLSLGADVLPEYKLQAPRIHRWTILHYSPFKAVWDWLILLLVIYTAVFTPYSAAFLLKETEEGPPATECGYACQPLAVVDLIVDIMFIVDILINFRTTYVNANEEVVSHPGRIAVHYFKGWFLIDMVAAIPFDLLIFGSGSEELIGLLKTARLLRLVRVARKLDRYSEYGAAVLFLLMCTFALIAHWLACIWYAIGNMEQPHMDSRIGWLHNLGDQIGKPYNSSGLGGPSIKDKYVTALYFTFSSLTSVGFGNVSPNTNSEKIFSICVMLIGSLMYASIFGNVSAIIQRLYSGTARYHTQMLRVREFIRFHQIPNPLRQRLEEYFQHAWSYTNGIDMNAVLKGFPECLQADICLHLNRSLLQHCKPFRGATKGCLRALAMKFKTTHAPPGDTLVHAGDLLTALYFISRGSIEILRGDVVVAILGKNDIFGEPLNLYARPGKSNGDVRALTYCDLHKIHRDDLLEVLDMYPEFSDHFWSSLEITFNLRDTNMIPGSPGSTELEGGFSRQRKRKLSFRRRTDKDTEQPGEVSALGPGRAGAGPSSRGRPGGPWGESPSSGPSSPESSEDEGPGRSSSPLRLVPFSSPRPPGEPPGGEPLMEDCEKSSDTCNPLSGAFSGVSNIFSFWGDSRGRQYQELPRCPAPTPSLLNIPLSSPGRRPRGDVESRLDALQRQLNRLETRLSADMATVLQLLQRQMTLVPPAYSAVTTPGPGPTSTSPLLPVSPLPTLTLDSLSQVSQFMACEELPPGAPELPQEGPTRRLSLPGQLGALTSQPLHRHGSDPGS
Inhibitor
Name:
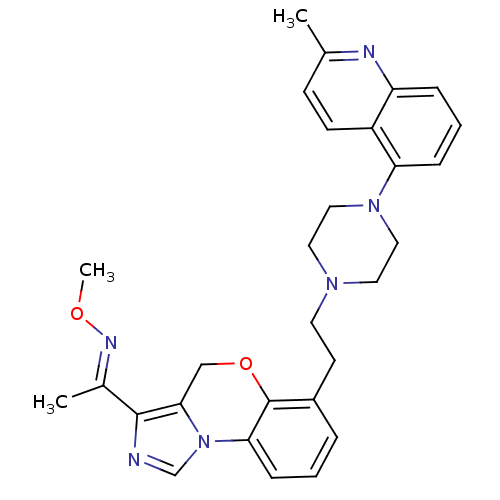
BDBM50326136
Synonyms:
1-(6-(2-(4-(2-methylquinolin-5-yl)piperazin-1-yl)ethyl)-4H-benzo[b]imidazo[1,5-d][1,4]oxazin-3-yl)ethanone O-methyl oxime | CHEMBL1241737
Type:
Small organic molecule
Emp. Form.:
C29H32N6O2
Mol. Mass.:
496.6034
SMILES:
CO\N=C(/C)c1ncn-2c1COc1c(CCN3CCN(CC3)c3cccc4nc(C)ccc34)cccc-21