Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Cannabinoid receptor 1
Ligand
BDBM21281
Substrate
n/a
Meas. Tech.
ChEMBL_444840 (CHEMBL895095)
EC50
12±n/a nM
Citation
 Dziadulewicz, EK; Bevan, SJ; Brain, CT; Coote, PR; Culshaw, AJ; Davis, AJ; Edwards, LJ; Fisher, AJ; Fox, AJ; Gentry, C; Groarke, A; Hart, TW; Huber, W; James, IF; Kesingland, A; La Vecchia, L; Loong, Y; Lyothier, I; McNair, K; O'Farrell, C; Peacock, M; Portmann, R; Schopfer, U; Yaqoob, M; Zadrobilek, J Naphthalen-1-yl-(4-pentyloxynaphthalen-1-yl)methanone: a potent, orally bioavailable human CB1/CB2 dual agonist with antihyperalgesic properties and restricted central nervous system penetration. J Med Chem 50:3851-6 (2007) [PubMed] Article
Dziadulewicz, EK; Bevan, SJ; Brain, CT; Coote, PR; Culshaw, AJ; Davis, AJ; Edwards, LJ; Fisher, AJ; Fox, AJ; Gentry, C; Groarke, A; Hart, TW; Huber, W; James, IF; Kesingland, A; La Vecchia, L; Loong, Y; Lyothier, I; McNair, K; O'Farrell, C; Peacock, M; Portmann, R; Schopfer, U; Yaqoob, M; Zadrobilek, J Naphthalen-1-yl-(4-pentyloxynaphthalen-1-yl)methanone: a potent, orally bioavailable human CB1/CB2 dual agonist with antihyperalgesic properties and restricted central nervous system penetration. J Med Chem 50:3851-6 (2007) [PubMed] Article More Info.:
Target
Name:
Cannabinoid receptor 1
Synonyms:
CANN6 | CANNABINOID CB1 | CB-R | CB1 | CNR | CNR1 | CNR1_HUMAN | Cannabinoid CB1 receptor | Cannabinoid receptor | Cannabinoid receptor 1 (CB1) | Cannabinoid receptor 1 (brain)
Type:
G Protein-Coupled Receptor (GPCR)
Mol. Mass.:
52868.96
Organism:
Homo sapiens (Human)
Description:
P21554
Residue:
472
Sequence:
MKSILDGLADTTFRTITTDLLYVGSNDIQYEDIKGDMASKLGYFPQKFPLTSFRGSPFQEKMTAGDNPQLVPADQVNITEFYNKSLSSFKENEENIQCGENFMDIECFMVLNPSQQLAIAVLSLTLGTFTVLENLLVLCVILHSRSLRCRPSYHFIGSLAVADLLGSVIFVYSFIDFHVFHRKDSRNVFLFKLGGVTASFTASVGSLFLTAIDRYISIHRPLAYKRIVTRPKAVVAFCLMWTIAIVIAVLPLLGWNCEKLQSVCSDIFPHIDETYLMFWIGVTSVLLLFIVYAYMYILWKAHSHAVRMIQRGTQKSIIIHTSEDGKVQVTRPDQARMDIRLAKTLVLILVVLIICWGPLLAIMVYDVFGKMNKLIKTVFAFCSMLCLLNSTVNPIIYALRSKDLRHAFRSMFPSCEGTAQPLDNSMGDSDCLHKHANNAASVHRAAESCIKSTVKIAKVTMSVSTDTSAEAL
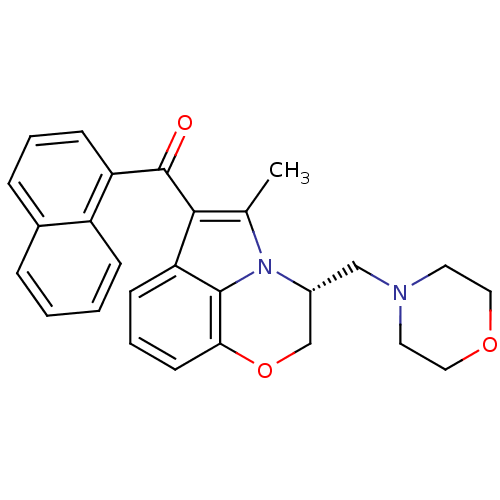
Inhibitor
Name:
BDBM21281
Synonyms:
(11R)-2-methyl-11-(morpholin-4-ylmethyl)-3-(naphthalen-1-ylcarbonyl)-9-oxa-1-azatricyclo[6.3.1.0^{4,12}]dodeca-2,4(12),5,7-tetraene | (2,3-dihydro-5-methyl-3-((4-morpholinyl)methyl)pyrrolo-(1,2,3-de)-1,4-benzoxazin-6-yl)(1-naphthalenyl)methanone monomethanesulfonat | CHEMBL188 | WIN 55,212-2 | WIN 55212-2 | WIN-55212 | WIN55212-2
Type:
Analgesic
Emp. Form.:
C27H26N2O3
Mol. Mass.:
426.5069
SMILES:
Cc1c(C(=O)c2cccc3ccccc23)c2cccc3OC[C@@H](CN4CCOCC4)n1c23 |r|