Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Renin
Ligand
BDBM50434420
Substrate
n/a
Meas. Tech.
ChEMBL_962065 (CHEMBL2388033)
IC50
7.8±n/a nM
Citation
 Nakamura, Y; Fujimoto, T; Ogawa, Y; Namiki, H; Suzuki, S; Asano, M; Sugita, C; Mochizuki, A; Miyazaki, S; Tamaki, K; Nagai, Y; Inoue, S; Nagayama, T; Kato, M; Chiba, K; Takasuna, K; Nishi, T Lead optimization of 5-amino-6-(2,2-dimethyl-5-oxo-4-phenylpiperazin-1-yl)-4-hydroxyhexanamides to reduce a cardiac safety issue: discovery of DS-8108b, an orally active renin inhibitor. Bioorg Med Chem 21:3175-96 (2013) [PubMed] Article
Nakamura, Y; Fujimoto, T; Ogawa, Y; Namiki, H; Suzuki, S; Asano, M; Sugita, C; Mochizuki, A; Miyazaki, S; Tamaki, K; Nagai, Y; Inoue, S; Nagayama, T; Kato, M; Chiba, K; Takasuna, K; Nishi, T Lead optimization of 5-amino-6-(2,2-dimethyl-5-oxo-4-phenylpiperazin-1-yl)-4-hydroxyhexanamides to reduce a cardiac safety issue: discovery of DS-8108b, an orally active renin inhibitor. Bioorg Med Chem 21:3175-96 (2013) [PubMed] Article More Info.:
Target
Name:
Renin
Synonyms:
Angiotensinogenase | REN | RENI_MACFA
Type:
PROTEIN
Mol. Mass.:
45012.96
Organism:
Macaca fascicularis
Description:
ChEMBL_979177
Residue:
406
Sequence:
MDGWRRMPRWGLLLLLWGSCTFGLPTDTTTFKRIFLKRMPSIRESLKERGVDMARLGPEWSQPMKRLALGNTTSSVILTNYMDTQYYGEIGIGTPPQTFKVVFDTGSSNVWVPSSKCSRLYTACVYHKLFDASDSSSYKHNGTELTLRYSTGTVSGFLSQDIITVGGITVTQMFGEVTEMPALPFMLAEFDGVVGMGFIEQAIGRVTPIFDNILSQGVLKEDVFSFYYNRDSENAQSLGGQIVLGGSDPQHYEGNFHYINLIKTGVWQIQMKGVSVGSSTLLCEDGCLALVDTGASYISGSTSSIEKLMEALGAKKRLFDYVVKCNEGPTLPDISFHLGGKEYTLTSADYVFQESYSSKKLCTLAIHAMDIPPPTGPTWALGATFIRKFYTEFDRRNNRIGFALAR
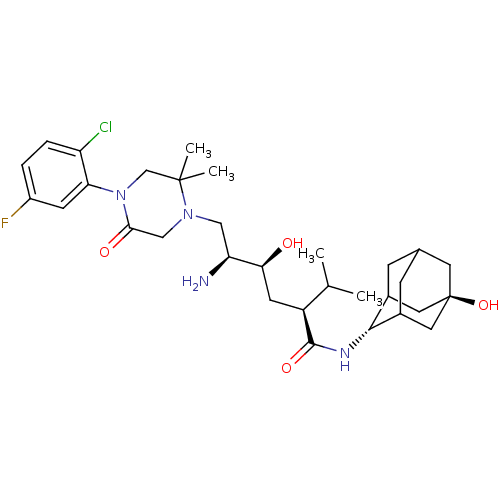
Inhibitor
Name:
BDBM50434420
Synonyms:
CHEMBL2387572
Type:
Small organic molecule
Emp. Form.:
C31H46ClFN4O4
Mol. Mass.:
593.173
SMILES:
CC(C)[C@H](C[C@H](O)[C@@H](N)CN1CC(=O)N(CC1(C)C)c1cc(F)ccc1Cl)C(=O)N[C@H]1C2CC3CC1C[C@](O)(C3)C2 |r,wU:5.5,3.3,30.31,wD:7.7,37.40,TLB:29:30:40.36.37:34.33.32,29:30:32:40.37.39,38:37:30:34.33.32,THB:36:35:32:40.37.39,36:37:30.35.34:32,38:37:30.35.34:32,39:37:30:34.33.32,39:33:30:40.36.37,(40.42,-16.49,;39.09,-17.26,;37.76,-16.49,;39.09,-18.8,;37.75,-19.57,;36.42,-18.8,;36.42,-17.26,;35.08,-19.56,;33.75,-18.79,;35.08,-21.1,;33.75,-21.87,;32.41,-21.1,;31.07,-21.87,;29.74,-21.11,;31.08,-23.41,;32.42,-24.18,;33.74,-23.41,;35.23,-23.81,;34.14,-24.9,;29.74,-24.19,;28.41,-23.43,;27.08,-24.2,;25.75,-23.43,;27.08,-25.74,;28.41,-26.51,;29.75,-25.74,;31.08,-26.51,;40.42,-19.57,;40.42,-21.11,;41.75,-18.8,;43.09,-19.58,;44.28,-18.3,;44.27,-16.81,;45.62,-16.34,;44.58,-17.56,;44.59,-19.16,;45.99,-19.72,;47.01,-18.44,;48.09,-19.53,;47.02,-16.91,;45.61,-18.79,)|