Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Endothelin receptor type B
Ligand
BDBM50143784
Substrate
n/a
Ki
0.1±n/a nM
Comments
PDSP_999
Citation
 Nishikibe, M; Ohta, H; Okada, M; Ishikawa, K; Hayama, T; Fukuroda, T; Noguchi, K; Saito, M; Kanoh, T; Ozaki, S; Kamei, T; Hara, K; William, D; Kivlighn, S; Krause, S; Gabel, R; Zingaro, G; Nolan, N; O'Brien, J; Clayton, F; Lynch, J; Pettibone, D; Siegl, P Pharmacological properties of J-104132 (L-753,037), a potent, orally active, mixed ETA/ETB endothelin receptor antagonist. J Pharmacol Exp Ther 289:1262-70 (1999) [PubMed]
Nishikibe, M; Ohta, H; Okada, M; Ishikawa, K; Hayama, T; Fukuroda, T; Noguchi, K; Saito, M; Kanoh, T; Ozaki, S; Kamei, T; Hara, K; William, D; Kivlighn, S; Krause, S; Gabel, R; Zingaro, G; Nolan, N; O'Brien, J; Clayton, F; Lynch, J; Pettibone, D; Siegl, P Pharmacological properties of J-104132 (L-753,037), a potent, orally active, mixed ETA/ETB endothelin receptor antagonist. J Pharmacol Exp Ther 289:1262-70 (1999) [PubMed] More Info.:
Target
Name:
Endothelin receptor type B
Synonyms:
EDNRB | EDNRB_HUMAN | ENDOTHELIN B | ET-B | ETRB | Endothelin receptor ET-B | Endothelin receptor non-selective type | Endothelin receptor, ET-A/ET-B
Type:
Enzyme Catalytic Domain
Mol. Mass.:
49664.00
Organism:
Homo sapiens (Human)
Description:
ENDOTHELIN B EDNRB HUMAN::P24530
Residue:
442
Sequence:
MQPPPSLCGRALVALVLACGLSRIWGEERGFPPDRATPLLQTAEIMTPPTKTLWPKGSNASLARSLAPAEVPKGDRTAGSPPRTISPPPCQGPIEIKETFKYINTVVSCLVFVLGIIGNSTLLRIIYKNKCMRNGPNILIASLALGDLLHIVIDIPINVYKLLAEDWPFGAEMCKLVPFIQKASVGITVLSLCALSIDRYRAVASWSRIKGIGVPKWTAVEIVLIWVVSVVLAVPEAIGFDIITMDYKGSYLRICLLHPVQKTAFMQFYKTAKDWWLFSFYFCLPLAITAFFYTLMTCEMLRKKSGMQIALNDHLKQRREVAKTVFCLVLVFALCWLPLHLSRILKLTLYNQNDPNRCELLSFLLVLDYIGINMASLNSCINPIALYLVSKRFKNCFKSCLCCWCQSFEEKQSLEEKQSCLKFKANDHGYDNFRSSNKYSSS
Inhibitor
Name:
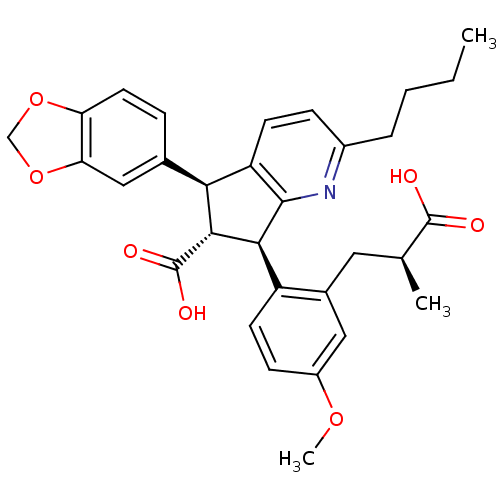
BDBM50143784
Synonyms:
(5S,6R,7R)-5-Benzo[1,3]dioxol-5-yl-2-butyl-7-[2-((S)-2-carboxy-propyl)-4-methoxy-phenyl]-6,7-dihydro-5H-[1]pyrindine-6-carboxylic acid | CHEMBL305576 | J-104132
Type:
Small organic molecule
Emp. Form.:
C31H33NO7
Mol. Mass.:
531.5962
SMILES:
CCCCc1ccc2[C@@H]([C@H]([C@@H](c2n1)c1ccc(OC)cc1C[C@H](C)C(O)=O)C(O)=O)c1ccc2OCOc2c1