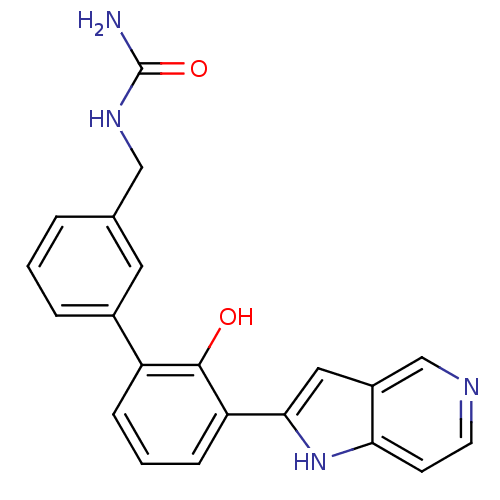
BDBM14864 5-azaindole analog 2::{[3-(2-hydroxy-3-{1H-pyrrolo[3,2-c]pyridin-2-yl}phenyl)phenyl]methyl}urea
SMILES NC(=O)NCc1cccc(c1)-c1cccc(-c2cc3cnccc3[nH]2)c1O
InChI Key InChIKey=SRPOHNDQBDHONJ-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 4 hits for monomerid = 14864
Found 4 hits for monomerid = 14864
Affinity DataKi: 800nM ΔG°: -8.23kcal/molepH: 7.4 T: 2°CAssay Description:The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su...More data for this Ligand-Target Pair
Affinity DataKi: 4.65E+4nM ΔG°: -5.85kcal/molepH: 7.4 T: 2°CAssay Description:The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su...More data for this Ligand-Target Pair
Affinity DataKi: 7.15E+4nM ΔG°: -5.59kcal/molepH: 7.4 T: 2°CAssay Description:The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su...More data for this Ligand-Target Pair
Affinity DataKi: 1.25E+5nM ΔG°: -5.27kcal/molepH: 7.4 T: 2°CAssay Description:The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su...More data for this Ligand-Target Pair
 3D Structure (crystal)
3D Structure (crystal)