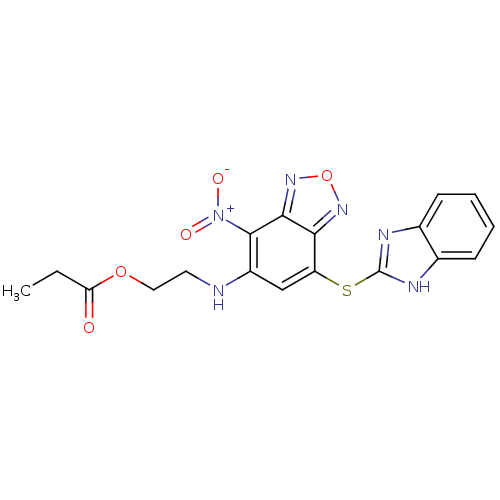
BDBM33121 2-[[7-(1H-benzimidazol-2-ylsulfanyl)-4-nitro-2,1,3-benzoxadiazol-5-yl]amino]ethyl propanoate::2-{[7-(1H-benzimidazol-2-ylthio)-4-nitro-2,1,3-benzoxadiazol-5-yl]amino}ethyl propionate::MLS000584446::SMR000207032::cid_12005314::propanoic acid 2-[[7-(1H-benzimidazol-2-ylthio)-4-nitro-2,1,3-benzoxadiazol-5-yl]amino]ethyl ester::propionic acid 2-[[7-(1H-benzimidazol-2-ylthio)-4-nitro-benzofurazan-5-yl]amino]ethyl ester
SMILES CCC(=O)OCCNc1cc(Sc2nc3ccccc3[nH]2)c2nonc2c1[N+]([O-])=O
InChI Key InChIKey=QOIZVPXCIKQNNN-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 3 hits for monomerid = 33121
Found 3 hits for monomerid = 33121
Affinity DataEC50: 1.63E+3nMpH: 7.4 T: 2°CAssay Description:The multiplex is constructed by using beads for each protein target that have been labeled with varying intensities of red color, so that each assay ...More data for this Ligand-Target Pair
TargetKappa-type opioid receptor(Homo sapiens (Human))
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Affinity DataIC50: 2.56E+3nMAssay Description:Data Source: Sanford-Burnham Center for Chemical Genomics (SBCCG) Source Affiliation: Sanford-Burnham Medical Research Institute (SBMRI, San Diego, C...More data for this Ligand-Target Pair
TargetKappa-type opioid receptor(Homo sapiens (Human))
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Affinity DataIC50: 1.91E+4nMAssay Description:Data Source: Sanford-Burnham Center for Chemical Genomics (SBCCG) Source Affiliation: Sanford-Burnham Medical Research Institute (SBMRI, San Diego, C...More data for this Ligand-Target Pair