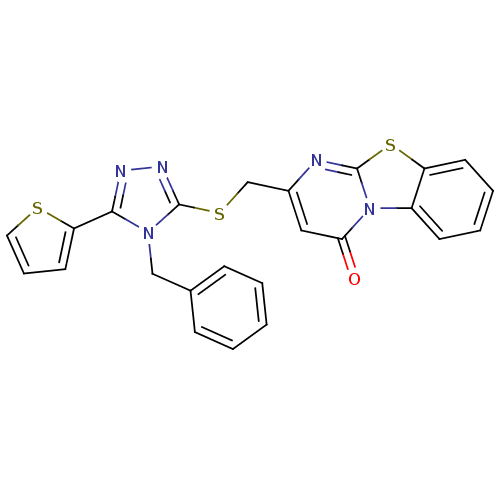
BDBM48349 2-[(4-benzyl-5-thiophen-2-yl-1,2,4-triazol-3-yl)sulfanylmethyl]pyrimido[2,1-b][1,3]benzothiazol-4-one::2-[[4-(phenylmethyl)-5-thiophen-2-yl-1,2,4-triazol-3-yl]sulfanylmethyl]pyrimido[2,1-b][1,3]benzothiazol-4-one::2-[[[4-(phenylmethyl)-5-thiophen-2-yl-1,2,4-triazol-3-yl]thio]methyl]-4-pyrimido[2,1-b][1,3]benzothiazolone::2-[[[4-benzyl-5-(2-thienyl)-1,2,4-triazol-3-yl]thio]methyl]pyrimido[2,1-b][1,3]benzothiazol-4-one::MLS000056286::SMR000068396::cid_2552706
SMILES O=c1cc(CSc2nnc(-c3cccs3)n2Cc2ccccc2)nc2sc3ccccc3n12
InChI Key InChIKey=JOXOOTKMEMJWMA-UHFFFAOYSA-N
Data 4 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 4 hits for monomerid = 48349
Found 4 hits for monomerid = 48349
Affinity DataIC50: 1.93E+4nMAssay Description:NIH Molecular Libraries Screening Centers Network [MLSCN] Emory Chemical Biology Discovery Center in MLSCN Assay provider: John A. Katzenellenbogen, ...More data for this Ligand-Target Pair
TargetGalanin receptor type 3(Homo sapiens (Human))
The Scripps Research Institute Molecular Screening Center
Curated by PubChem BioAssay
The Scripps Research Institute Molecular Screening Center
Curated by PubChem BioAssay
TargetCarboxy-terminal domain RNA polymerase II polypeptide A small phosphatase 1(Homo sapiens (Human))
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Affinity DataIC50: 5.42E+3nMAssay Description:Data Source: Sanford-Burnham Center for Chemical Genomics (SBCCG) Source Affiliation: Sanford-Burnham Medical Research Institute (SBMRI, San Diego, C...More data for this Ligand-Target Pair
Affinity DataIC50: 1.56E+3nMAssay Description:NIH Molecular Libraries Screening Centers Network [MLSCN] Emory Chemical Biology Discovery Center in MLSCN Assay provider: John A. Katzenellenbogen, ...More data for this Ligand-Target Pair