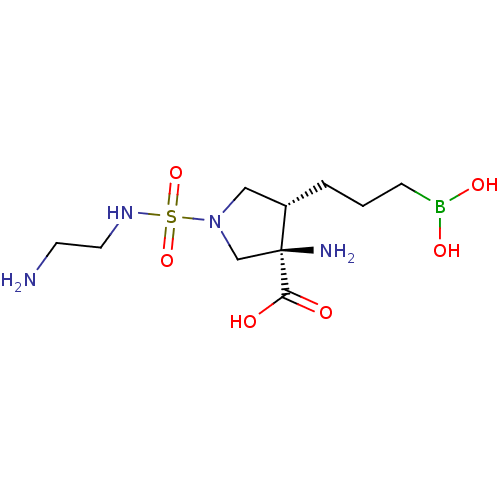
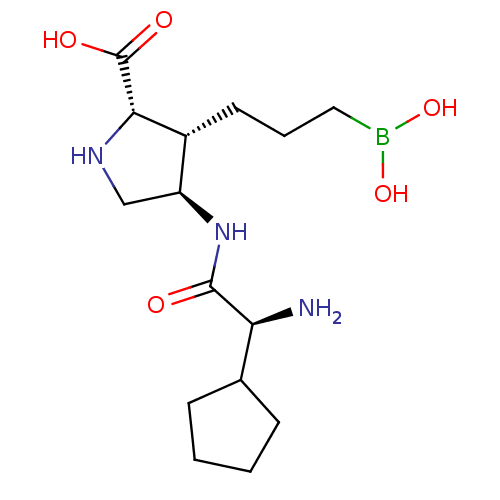
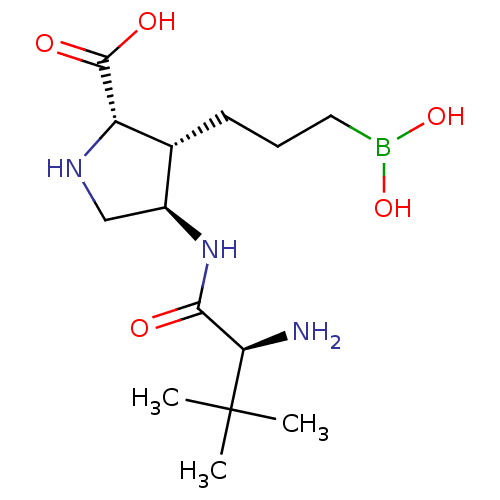
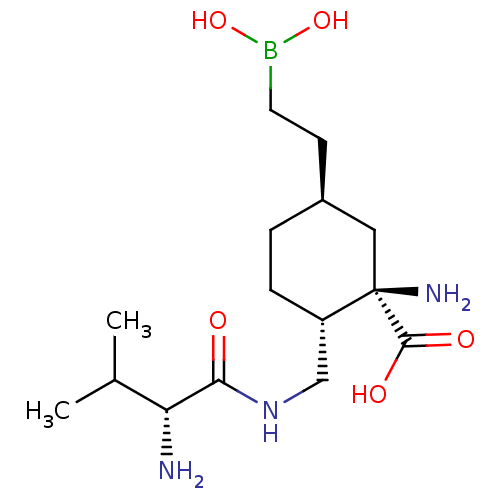
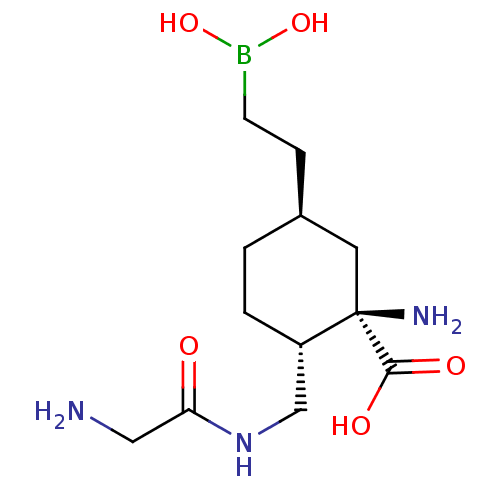
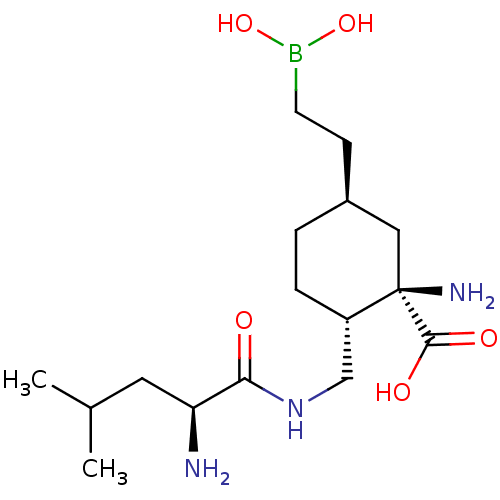
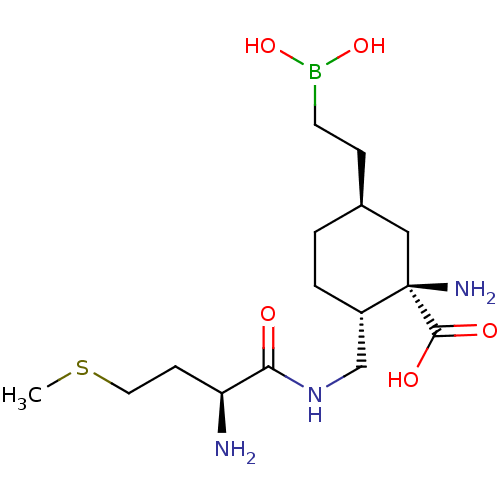
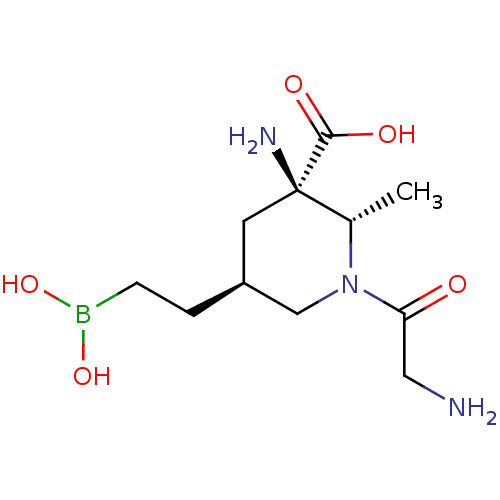
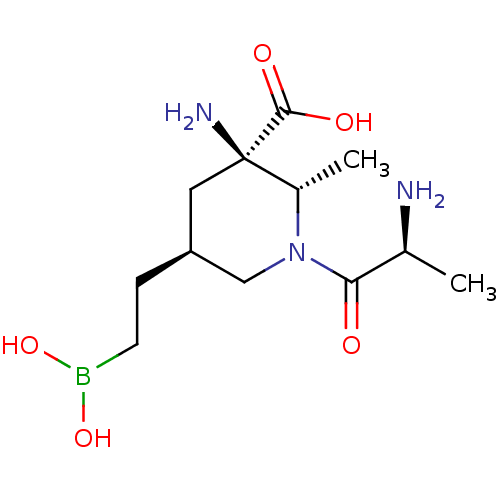
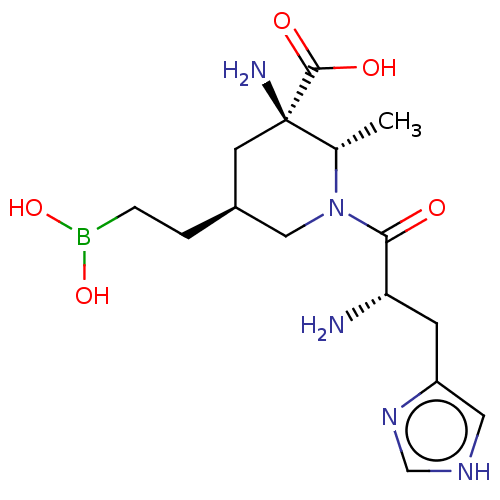
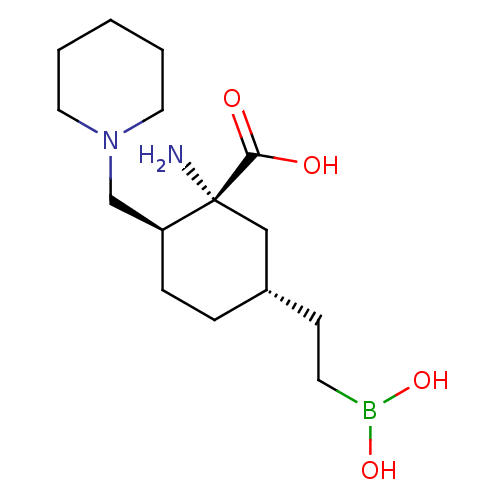
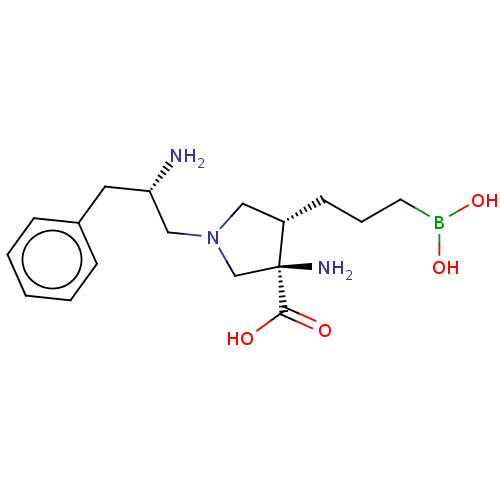
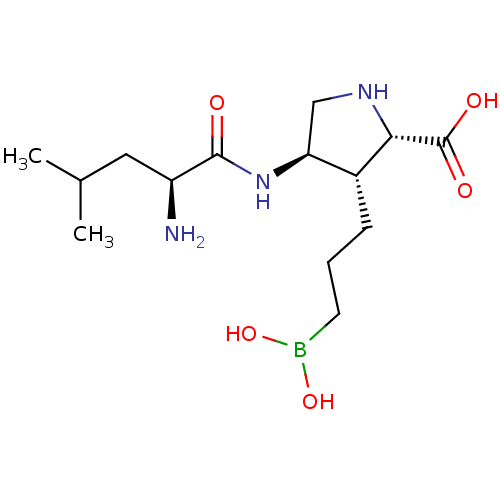
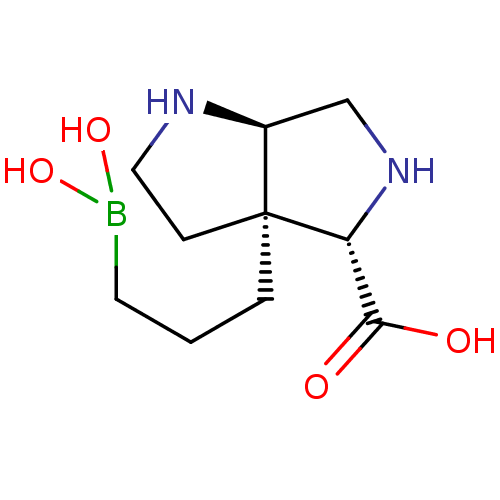
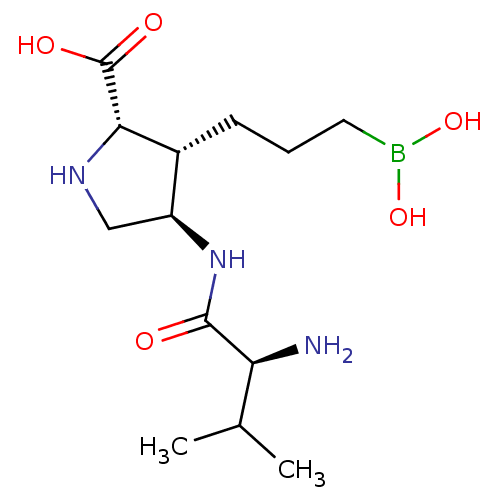
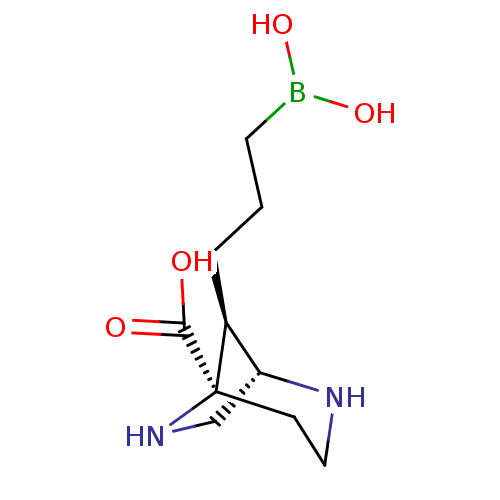
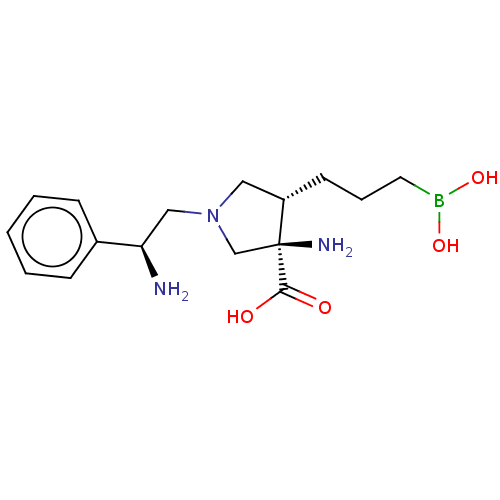
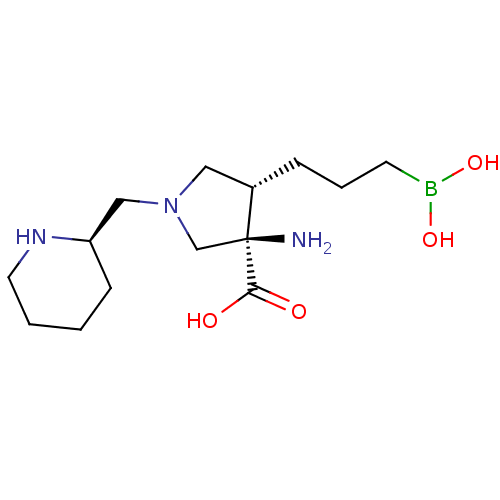
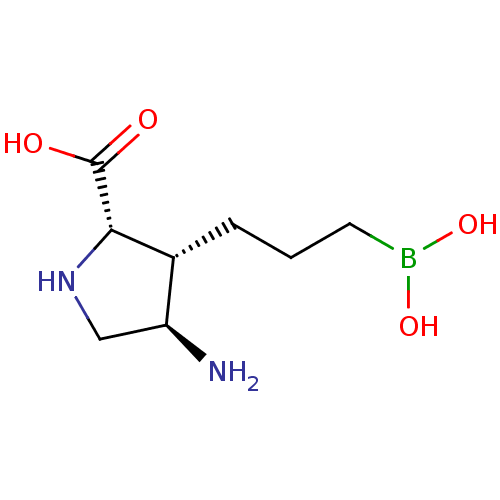
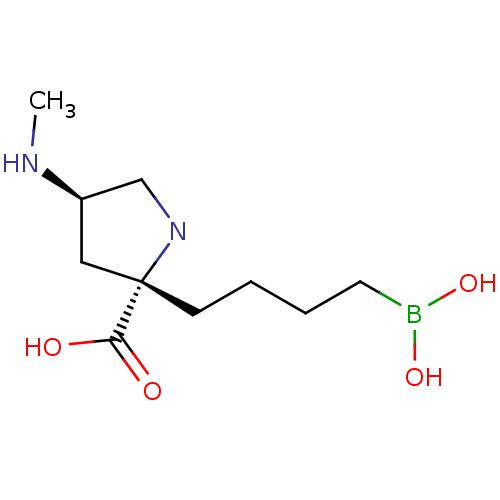
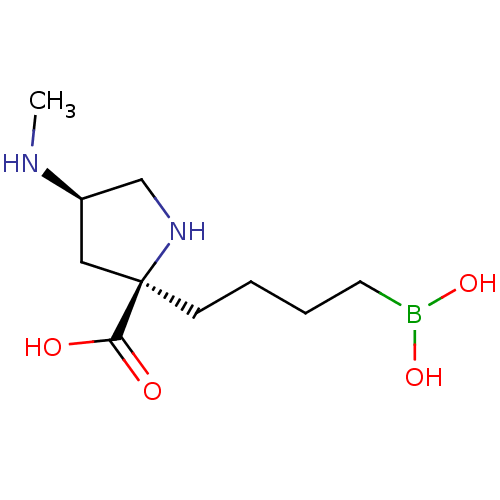
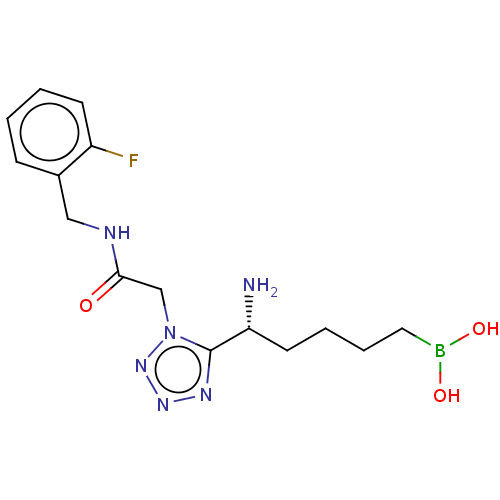
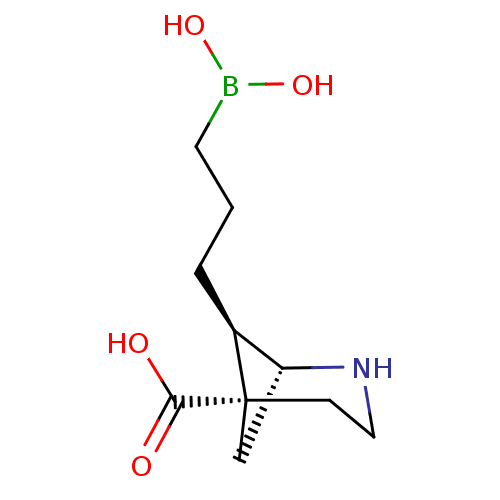
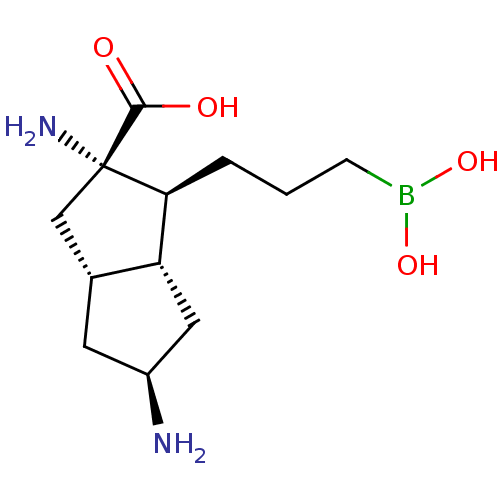
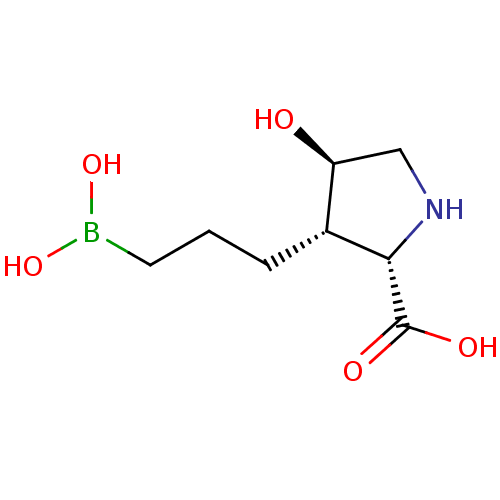
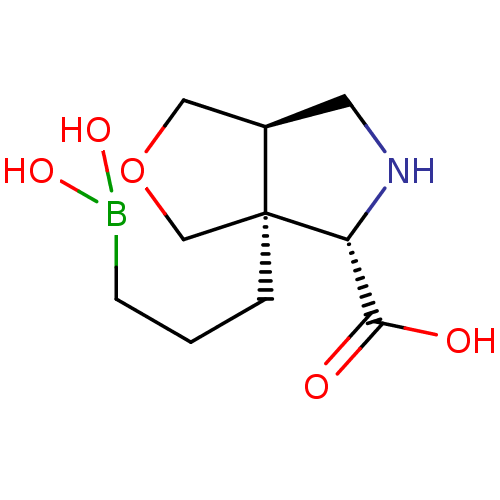
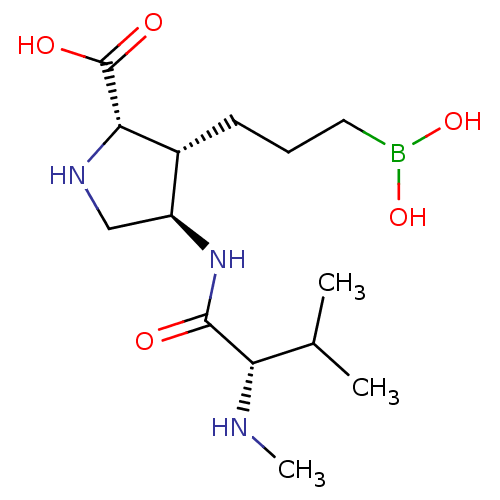
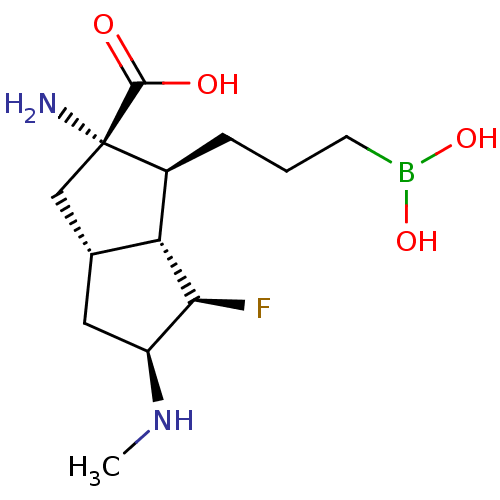
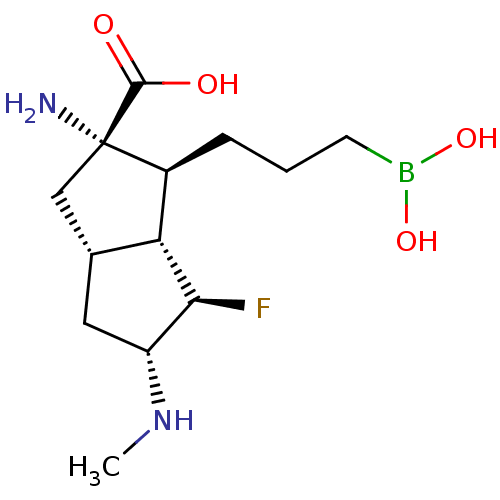
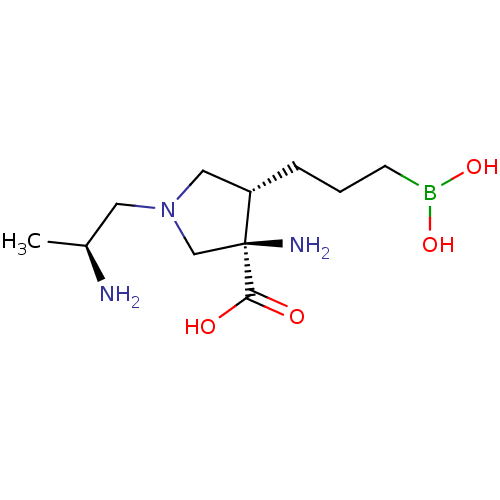
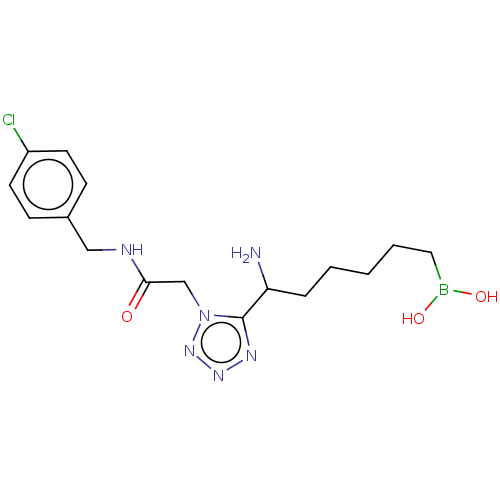
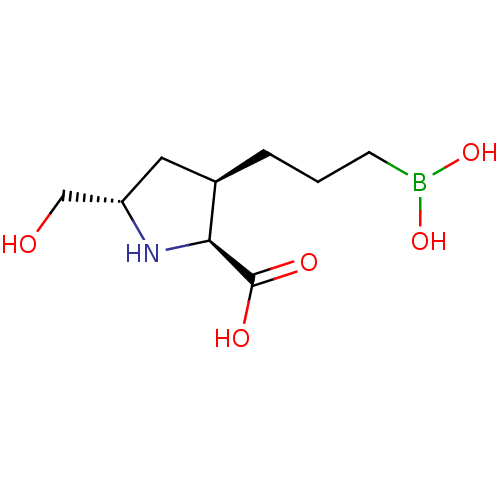
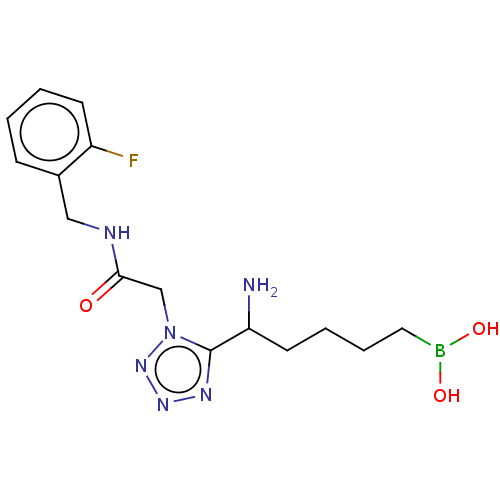
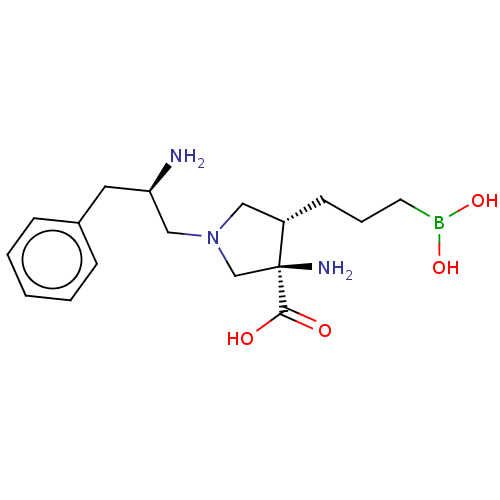
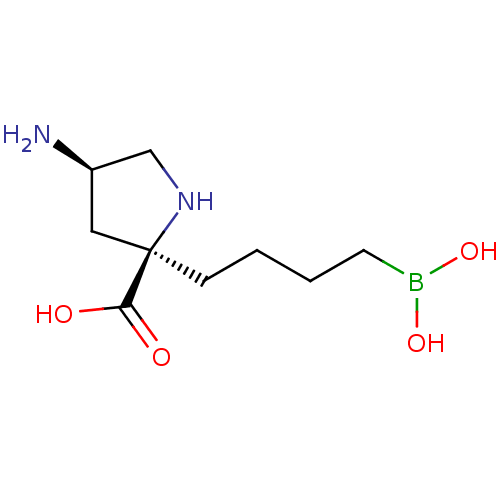
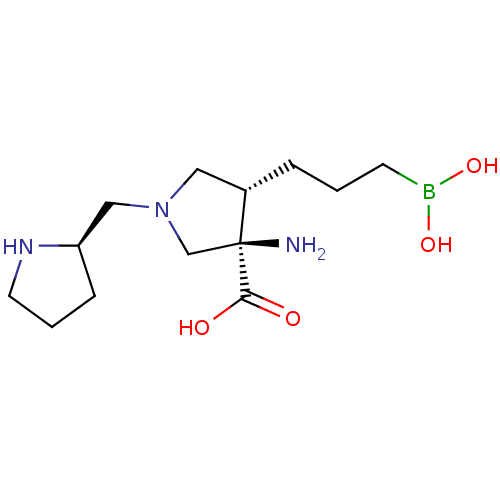
Affinity DataIC50: 0.100nMAssay Description:Inhibition of human Arg1More data for this Ligand-Target Pair
Affinity DataIC50: 0.800nMAssay Description:Inhibition of human Arg1 by TOGA assayMore data for this Ligand-Target Pair
Affinity DataIC50: 0.800nMAssay Description:Inhibition of human Arg1 by TOGA assayMore data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:Inhibition of recombinant human ARG1 expressed in Escherichia coli using L-arginine hydrochloride as substrate incubated for 1 hr by colorimetric ass...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:Inhibition of recombinant human ARG1 expressed in Escherichia coli using L-arginine hydrochloride as substrate incubated for 1 hr by colorimetric ass...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:Inhibition of recombinant human ARG1 expressed in Escherichia coli using L-arginine hydrochloride as substrate incubated for 1 hr by colorimetric ass...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:Inhibition of recombinant human ARG1 expressed in Escherichia coli using L-arginine hydrochloride as substrate incubated for 1 hr by colorimetric ass...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:Inhibition of recombinant human ARG1 expressed in Escherichia coli using L-arginine hydrochloride as substrate incubated for 1 hr by colorimetric ass...More data for this Ligand-Target Pair
Affinity DataIC50: <1nMAssay Description:Inhibition of human recombinant ARG1 expressed in Escherichia coli using thioarginine as a substrate preincubated for 30 mins followed by substrate a...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:Inhibition of human Arg1More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:Inhibition of human Arg1More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:Inhibition of human Arg1More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:Inhibition of human Arg1More data for this Ligand-Target Pair
Affinity DataIC50: 1.30nMAssay Description:Inhibition of human recombinant arginase 1 expressed in Escherichia coli BL21 (DE3) assessed as reduction in urea production using L-arginine as subs...More data for this Ligand-Target Pair
Affinity DataIC50: 1.60nMAssay Description:Inhibition of human Arg1 by TOGA assayMore data for this Ligand-Target Pair
Affinity DataIC50: 1.60nMAssay Description:Inhibition of ARG1 (unknown origin) assessed as reduction in thio-ornithine production using thioarginine as substrate by fluorimetric analysisMore data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:Inhibition of human recombinant ARG1 expressed in Escherichia coli using thioarginine as a substrate preincubated for 30 mins followed by substrate a...More data for this Ligand-Target Pair
Affinity DataIC50: 2.20nMAssay Description:Inhibition of ARG1 (unknown origin) assessed as reduction in thio-ornithine production using thioarginine as substrate by fluorimetric analysisMore data for this Ligand-Target Pair
Affinity DataIC50: 2.5nMAssay Description:Inhibition of human recombinant arginase 1 expressed in Escherichia coli BL21 (DE3) assessed as reduction in urea production using L-arginine as subs...More data for this Ligand-Target Pair
Affinity DataIC50: 2.60nMAssay Description:Inhibition of human recombinant arginase 1 expressed in Escherichia coli BL21 (DE3) assessed as reduction in urea production using L-arginine as subs...More data for this Ligand-Target Pair
Affinity DataIC50: 3nMAssay Description:Inhibition of human recombinant ARG1 expressed in Escherichia coli using thioarginine as a substrate preincubated for 30 mins followed by substrate a...More data for this Ligand-Target Pair
Affinity DataIC50: <3nMAssay Description:The inhibitory effects of Examples 1 to 30 on the activity of Human Arginase 1 and Arginase 2 activity were quantified by measuring the formation of ...More data for this Ligand-Target Pair
Affinity DataIC50: 3nMAssay Description:Inhibition of human Arg1 by TOGA assayMore data for this Ligand-Target Pair
Affinity DataIC50: 3.20nMAssay Description:Inhibition of human Arg1 by TOGA assayMore data for this Ligand-Target Pair
Affinity DataIC50: 3.20nMAssay Description:Inhibition of ARG1 (unknown origin) assessed as reduction in thio-ornithine production using thioarginine as substrate by fluorimetric analysisMore data for this Ligand-Target Pair
Affinity DataIC50: 3.40nMAssay Description:Inhibition of arginase I (ARG I) and arginase II (ARGG II) novel compounds is followed spectrophotometrically at 530 nm. The compound to be tested is...More data for this Ligand-Target Pair
Affinity DataIC50: 3.90nMAssay Description:Inhibition of ARG1 (unknown origin) assessed as reduction in thio-ornithine production using thioarginine as substrate by fluorimetric analysisMore data for this Ligand-Target Pair
Affinity DataIC50: 4nMAssay Description:Inhibition of human arginase1 in presence of DNTB by thio ornithine generation assayMore data for this Ligand-Target Pair
Affinity DataIC50: 4nMAssay Description:Inhibition of human recombinant ARG1 expressed in Escherichia coli using thioarginine as a substrate preincubated for 30 mins followed by substrate a...More data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:Inhibition of ARG1 (unknown origin) assessed as reduction in thio-ornithine production using thioarginine as substrate by fluorimetric analysisMore data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:Inhibition of human recombinant ARG1 expressed in Escherichia coli using thioarginine as a substrate preincubated for 30 mins followed by substrate a...More data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:Inhibition of human arginase1 in presence of DNTB by thio ornithine generation assayMore data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:Inhibition of human recombinant ARG1 expressed in Escherichia coli using thioarginine as a substrate preincubated for 30 mins followed by substrate a...More data for this Ligand-Target Pair
Affinity DataIC50: 5.5nMAssay Description:Inhibition of arginase I (ARG I) and arginase II (ARGG II) novel compounds is followed spectrophotometrically at 530 nm. The compound to be tested is...More data for this Ligand-Target Pair
Affinity DataIC50: 6nMAssay Description:Inhibition of human Arg1 by TOGA assayMore data for this Ligand-Target Pair
Affinity DataIC50: 7nMAssay Description:Inhibition of human recombinant ARG1 expressed in Escherichia coli using thioarginine as a substrate preincubated for 30 mins followed by substrate a...More data for this Ligand-Target Pair
Affinity DataIC50: 7nMAssay Description:Inhibition of human arginase1 in presence of DNTB by thio ornithine generation assayMore data for this Ligand-Target Pair
Affinity DataIC50: 7.70nMAssay Description:Inhibition of human recombinant arginase 1 expressed in Escherichia coli BL21 (DE3) assessed as reduction in urea production using L-arginine as subs...More data for this Ligand-Target Pair
Affinity DataIC50: 7.80nMAssay Description:Inhibition of arginase I (ARG I) and arginase II (ARGG II) novel compounds is followed spectrophotometrically at 530 nm. The compound to be tested is...More data for this Ligand-Target Pair
Affinity DataIC50: 8nMAssay Description:Inhibition of human Arg1 by TOGA assayMore data for this Ligand-Target Pair
Affinity DataIC50: 8nMAssay Description:Inhibition of human recombinant ARG1 expressed in Escherichia coli using thioarginine as a substrate preincubated for 30 mins followed by substrate a...More data for this Ligand-Target Pair
Affinity DataIC50: 9.10nMAssay Description:Inhibition of arginase I (ARG I) and arginase II (ARGG II) novel compounds is followed spectrophotometrically at 530 nm. The compound to be tested is...More data for this Ligand-Target Pair
Affinity DataIC50: 9.40nMAssay Description:Inhibition of arginase I (ARG I) and arginase II (ARGG II) novel compounds is followed spectrophotometrically at 530 nm. The compound to be tested is...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:Inhibition of human recombinant arginase 1 expressed in Escherichia coli BL21 (DE3) assessed as reduction in urea production using L-arginine as subs...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:Inhibition of human recombinant arginase 1 expressed in Escherichia coli BL21 (DE3) assessed as reduction in urea production using L-arginine as subs...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:The inhibitory effects of Examples 1 to 30 on the activity of Human Arginase 1 and Arginase 2 activity were quantified by measuring the formation of ...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:Inhibition of human Arg1 by TOGA assayMore data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:Inhibition of human recombinant arginase 1 expressed in Escherichia coli BL21 (DE3) assessed as reduction in urea production using L-arginine as subs...More data for this Ligand-Target Pair
Affinity DataIC50: 10.5nMAssay Description:Inhibition of arginase I (ARG I) and arginase II (ARGG II) novel compounds is followed spectrophotometrically at 530 nm. The compound to be tested is...More data for this Ligand-Target Pair
Affinity DataIC50: 10.6nMAssay Description:Inhibition of arginase I (ARG I) and arginase II (ARGG II) novel compounds is followed spectrophotometrically at 530 nm. The compound to be tested is...More data for this Ligand-Target Pair