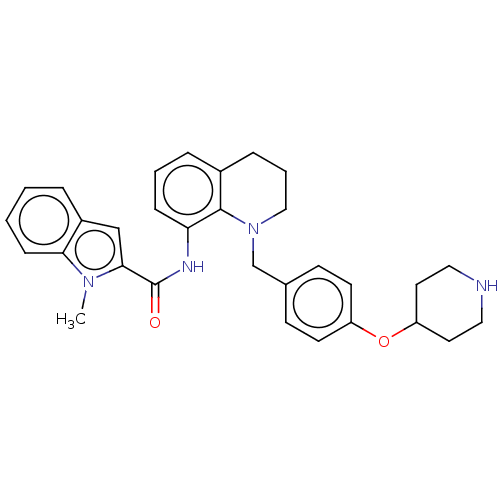
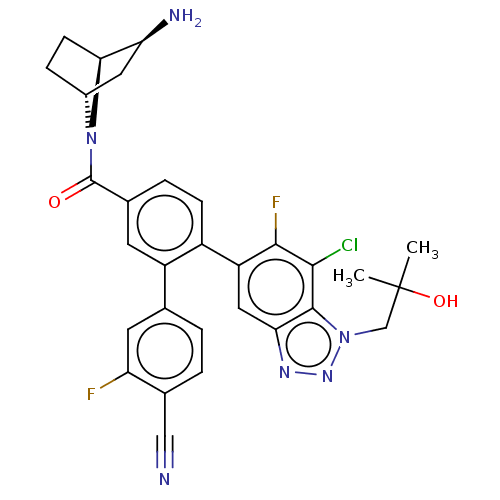
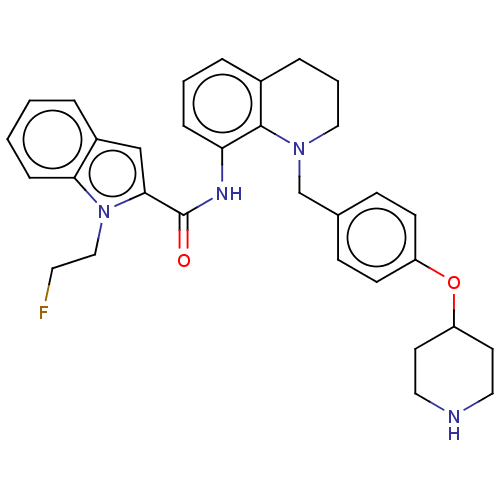
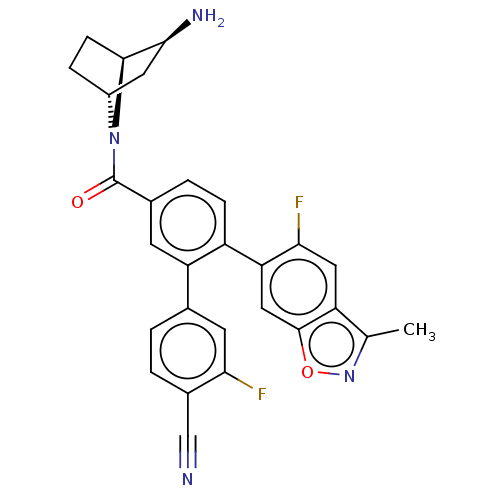
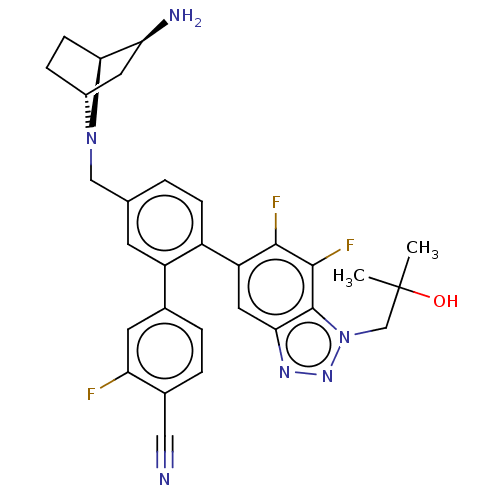
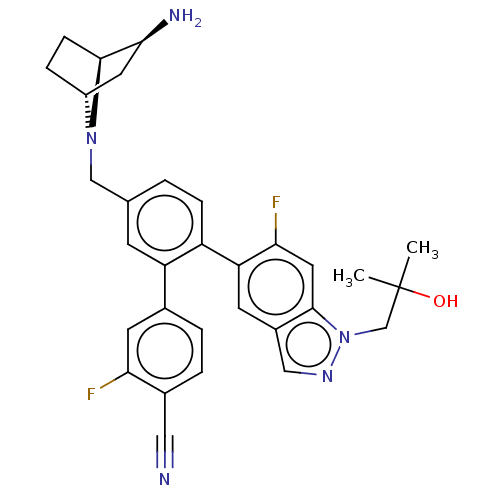
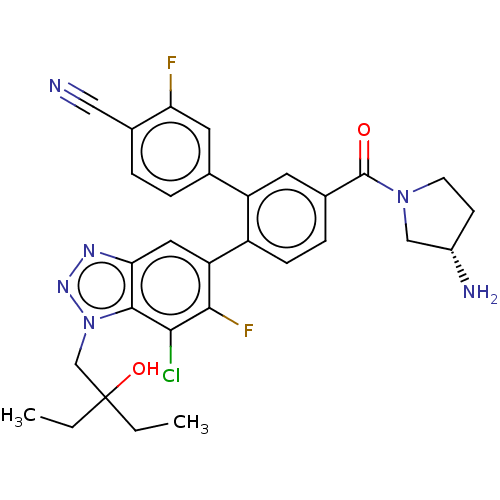
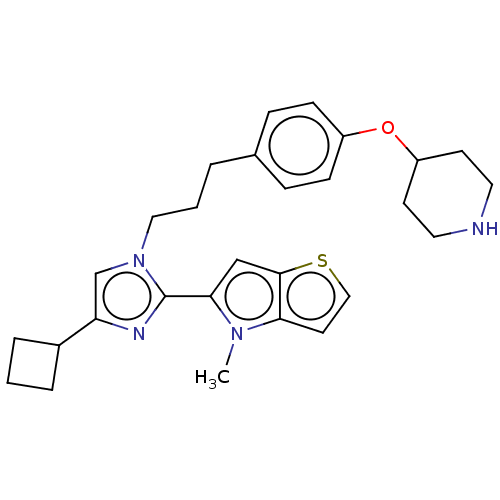
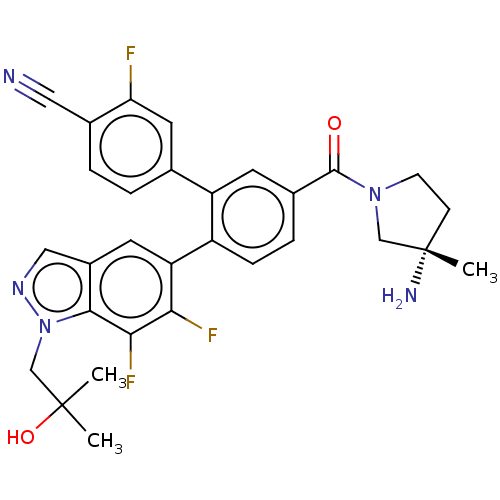
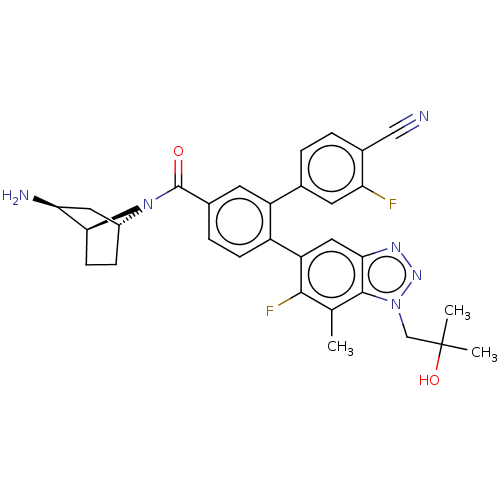
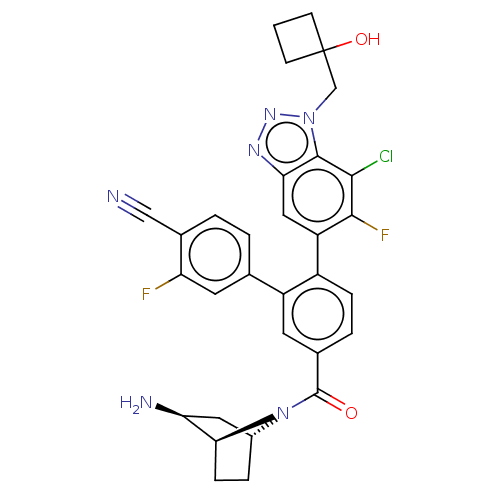
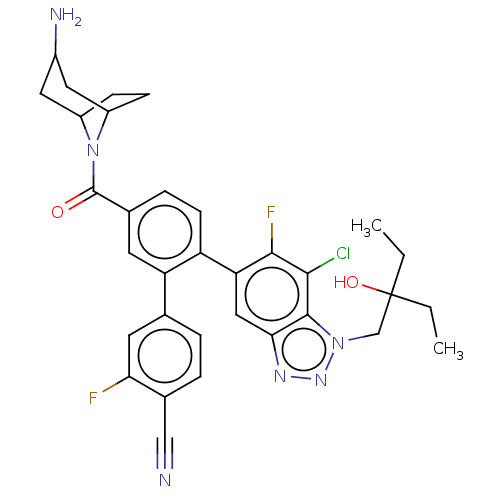
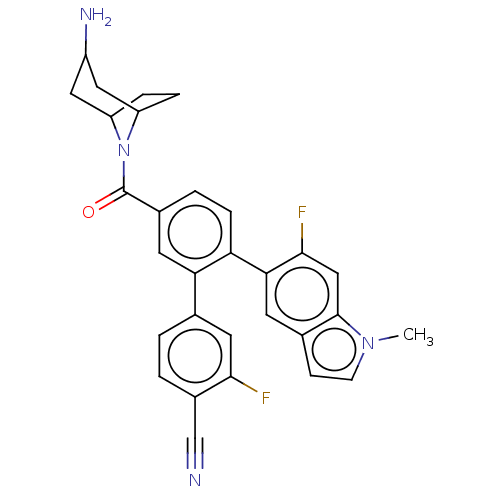
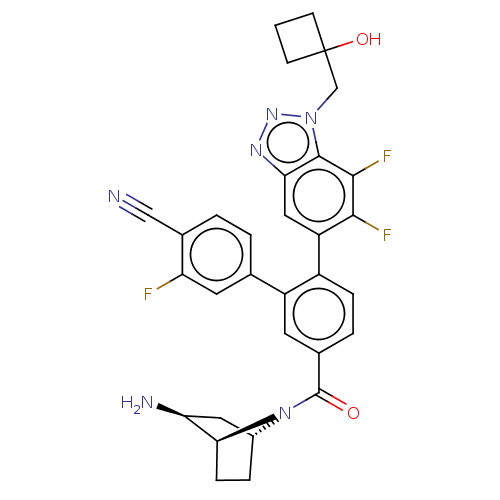
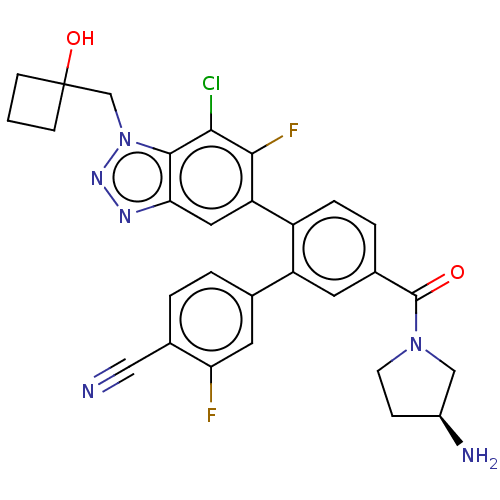
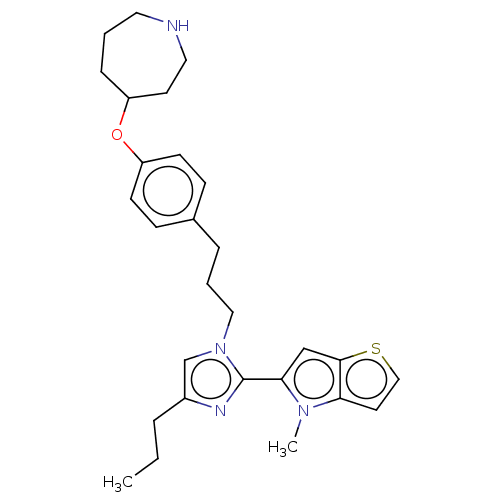
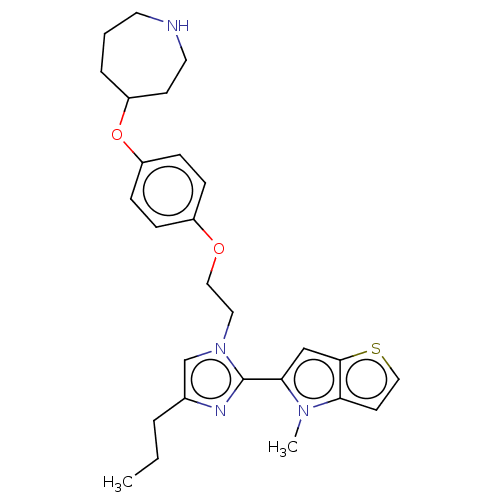
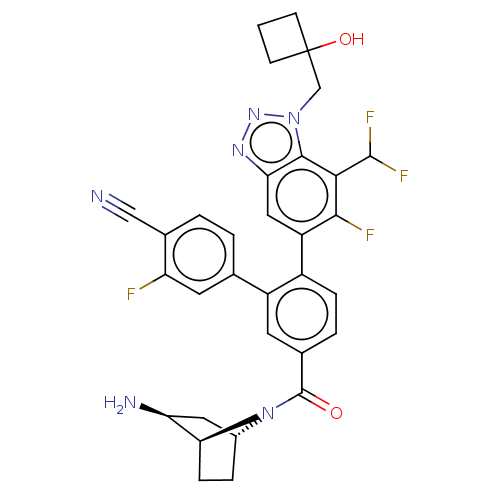
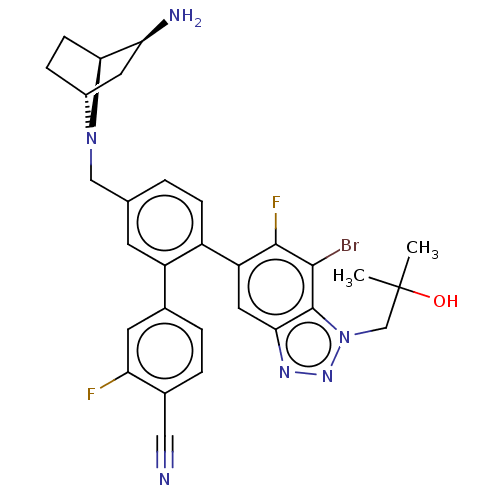
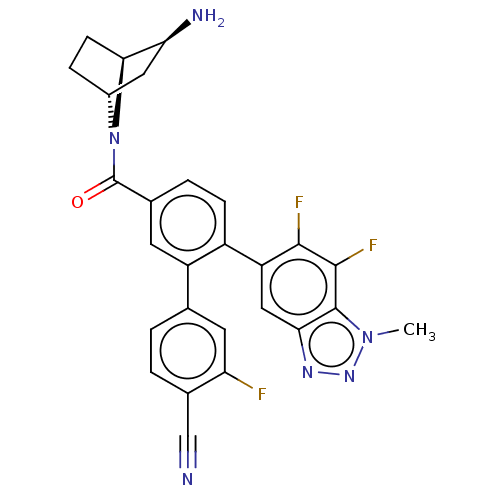
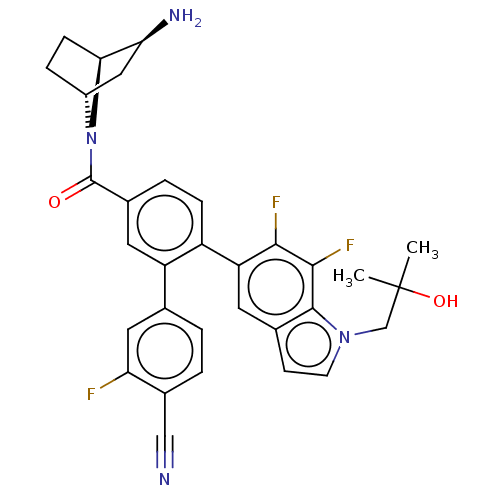
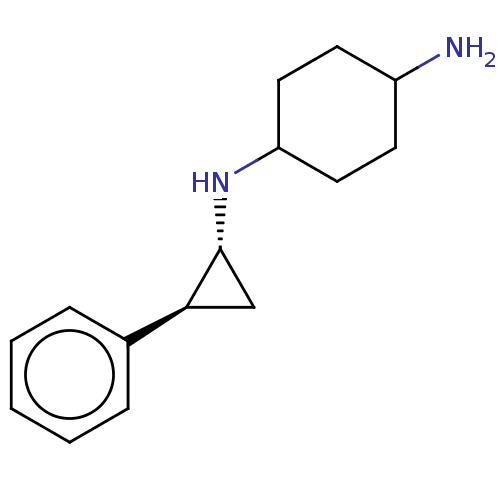
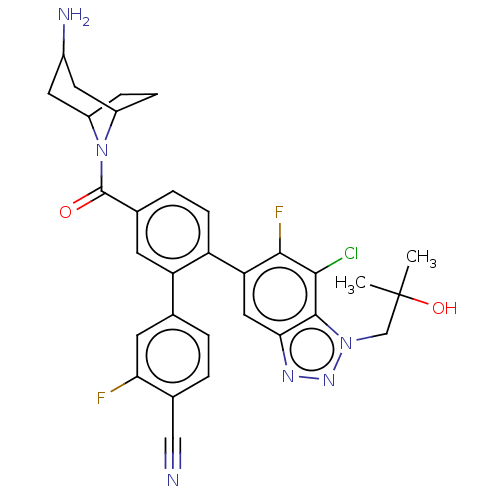
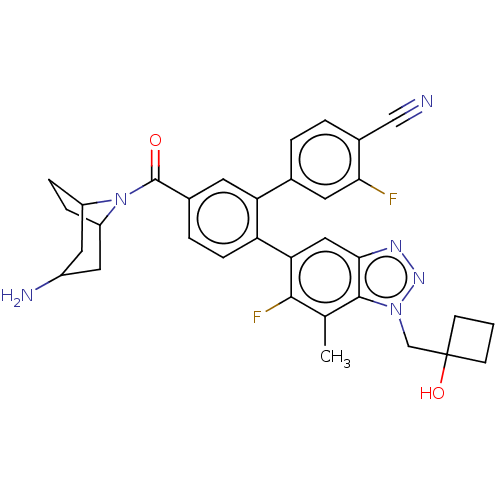
TargetLysine-specific histone demethylase 1A(Homo sapiens (Human))
Shenyang Pharmaceutical University
Curated by ChEMBL
Shenyang Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 0.0500nMAssay Description:Inhibition of human recombinant LSD1 using fluorogenic ADHP based substrate preincubated for 30 mins followed by substrate addition measured after 10...More data for this Ligand-Target Pair
TargetLysine-specific histone demethylase 1A(Homo sapiens (Human))
Shenyang Pharmaceutical University
Curated by ChEMBL
Shenyang Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 0.0600nMAssay Description:The conditions for measuring inhibitory activity of compounds against LSD1 activity were determined with reference to a document available from the w...More data for this Ligand-Target Pair
TargetLysine-specific histone demethylase 1A(Homo sapiens (Human))
Shenyang Pharmaceutical University
Curated by ChEMBL
Shenyang Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 0.0600nMAssay Description:The conditions for measuring inhibitory activity of compounds against LSD1 activity were determined with reference to a document available from the w...More data for this Ligand-Target Pair
TargetLysine-specific histone demethylase 1A(Homo sapiens (Human))
Shenyang Pharmaceutical University
Curated by ChEMBL
Shenyang Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 0.0600nMAssay Description:Inhibition of human recombinant LSD1 using fluorogenic ADHP based substrate preincubated for 30 mins followed by substrate addition measured after 10...More data for this Ligand-Target Pair
TargetLysine-specific histone demethylase 1A(Homo sapiens (Human))
Shenyang Pharmaceutical University
Curated by ChEMBL
Shenyang Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 0.0620nMAssay Description:The conditions for measuring inhibitory activity of compounds against LSD1 activity were determined with reference to a document available from the w...More data for this Ligand-Target Pair
TargetLysine-specific histone demethylase 1A(Homo sapiens (Human))
Shenyang Pharmaceutical University
Curated by ChEMBL
Shenyang Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 0.0700nMAssay Description:The conditions for measuring inhibitory activity of compounds against LSD1 activity were determined with reference to a document available from the w...More data for this Ligand-Target Pair
TargetLysine-specific histone demethylase 1A(Homo sapiens (Human))
Shenyang Pharmaceutical University
Curated by ChEMBL
Shenyang Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 0.0700nMAssay Description:The conditions for measuring inhibitory activity of compounds against LSD1 activity were determined with reference to a document available from the w...More data for this Ligand-Target Pair
TargetLysine-specific histone demethylase 1A(Homo sapiens (Human))
Shenyang Pharmaceutical University
Curated by ChEMBL
Shenyang Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 0.0750nMAssay Description:The conditions for measuring inhibitory activity of compounds against LSD1 activity were determined with reference to a document available from the w...More data for this Ligand-Target Pair
TargetLysine-specific histone demethylase 1A(Homo sapiens (Human))
Shenyang Pharmaceutical University
Curated by ChEMBL
Shenyang Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 0.0760nMAssay Description:The conditions for measuring inhibitory activity of compounds against LSD1 activity were determined with reference to a document available from the w...More data for this Ligand-Target Pair
TargetLysine-specific histone demethylase 1A(Homo sapiens (Human))
Shenyang Pharmaceutical University
Curated by ChEMBL
Shenyang Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 0.0900nMAssay Description:The conditions for measuring inhibitory activity of compounds against LSD1 activity were determined with reference to a document available from the w...More data for this Ligand-Target Pair
TargetLysine-specific histone demethylase 1A(Homo sapiens (Human))
Shenyang Pharmaceutical University
Curated by ChEMBL
Shenyang Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 0.0900nMAssay Description:The conditions for measuring inhibitory activity of compounds against LSD1 activity were determined with reference to a document available from the w...More data for this Ligand-Target Pair
TargetLysine-specific histone demethylase 1A(Homo sapiens (Human))
Shenyang Pharmaceutical University
Curated by ChEMBL
Shenyang Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 0.0910nMAssay Description:The conditions for measuring inhibitory activity of compounds against LSD1 activity were determined with reference to a document available from the w...More data for this Ligand-Target Pair
TargetLysine-specific histone demethylase 1A(Homo sapiens (Human))
Shenyang Pharmaceutical University
Curated by ChEMBL
Shenyang Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 0.0930nMAssay Description:The conditions for measuring inhibitory activity of compounds against LSD1 activity were determined with reference to a document available from the w...More data for this Ligand-Target Pair
TargetLysine-specific histone demethylase 1A(Homo sapiens (Human))
Shenyang Pharmaceutical University
Curated by ChEMBL
Shenyang Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 0.100nMAssay Description:The conditions for measuring inhibitory activity of compounds against LSD1 activity were determined with reference to a document available from the w...More data for this Ligand-Target Pair
TargetLysine-specific histone demethylase 1A(Homo sapiens (Human))
Shenyang Pharmaceutical University
Curated by ChEMBL
Shenyang Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 0.100nMAssay Description:The conditions for measuring inhibitory activity of compounds against LSD1 activity were determined with reference to a document available from the w...More data for this Ligand-Target Pair
TargetLysine-specific histone demethylase 1A(Homo sapiens (Human))
Shenyang Pharmaceutical University
Curated by ChEMBL
Shenyang Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 0.100nMAssay Description:The conditions for measuring inhibitory activity of compounds against LSD1 activity were determined with reference to a document available from the w...More data for this Ligand-Target Pair
TargetLysine-specific histone demethylase 1A(Homo sapiens (Human))
Shenyang Pharmaceutical University
Curated by ChEMBL
Shenyang Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 0.100nMAssay Description:The conditions for measuring inhibitory activity of compounds against LSD1 activity were determined with reference to a document available from the w...More data for this Ligand-Target Pair
TargetLysine-specific histone demethylase 1A(Homo sapiens (Human))
Shenyang Pharmaceutical University
Curated by ChEMBL
Shenyang Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 0.100nMAssay Description:Inhibition of human recombinant LSD1 using fluorogenic ADHP based substrate preincubated for 30 mins followed by substrate addition measured after 10...More data for this Ligand-Target Pair
TargetLysine-specific histone demethylase 1A(Homo sapiens (Human))
Shenyang Pharmaceutical University
Curated by ChEMBL
Shenyang Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 0.100nMAssay Description:The conditions for measuring inhibitory activity of compounds against LSD1 activity were determined with reference to a document available from the w...More data for this Ligand-Target Pair
TargetLysine-specific histone demethylase 1A(Homo sapiens (Human))
Shenyang Pharmaceutical University
Curated by ChEMBL
Shenyang Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 0.100nMAssay Description:Inhibition of recombinant human LSD1 expressed in Escherichia coli using biotinylated H3K4me as substrate by TR-FRET assayMore data for this Ligand-Target Pair
TargetLysine-specific histone demethylase 1A(Homo sapiens (Human))
Shenyang Pharmaceutical University
Curated by ChEMBL
Shenyang Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 0.110nMAssay Description:The conditions for measuring inhibitory activity of compounds against LSD1 activity were determined with reference to a document available from the w...More data for this Ligand-Target Pair
TargetLysine-specific histone demethylase 1A(Homo sapiens (Human))
Shenyang Pharmaceutical University
Curated by ChEMBL
Shenyang Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 0.110nMAssay Description:The conditions for measuring inhibitory activity of compounds against LSD1 activity were determined with reference to a document available from the w...More data for this Ligand-Target Pair
TargetLysine-specific histone demethylase 1A(Homo sapiens (Human))
Shenyang Pharmaceutical University
Curated by ChEMBL
Shenyang Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 0.110nMAssay Description:The conditions for measuring inhibitory activity of compounds against LSD1 activity were determined with reference to a document available from the w...More data for this Ligand-Target Pair
TargetLysine-specific histone demethylase 1A(Homo sapiens (Human))
Shenyang Pharmaceutical University
Curated by ChEMBL
Shenyang Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 0.110nMAssay Description:The conditions for measuring inhibitory activity of compounds against LSD1 activity were determined with reference to a document available from the w...More data for this Ligand-Target Pair
TargetLysine-specific histone demethylase 1A(Homo sapiens (Human))
Shenyang Pharmaceutical University
Curated by ChEMBL
Shenyang Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 0.120nMAssay Description:The conditions for measuring inhibitory activity of compounds against LSD1 activity were determined with reference to a document available from the w...More data for this Ligand-Target Pair
TargetLysine-specific histone demethylase 1A(Homo sapiens (Human))
Shenyang Pharmaceutical University
Curated by ChEMBL
Shenyang Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 0.120nMAssay Description:The conditions for measuring inhibitory activity of compounds against LSD1 activity were determined with reference to a document available from the w...More data for this Ligand-Target Pair
TargetLysine-specific histone demethylase 1A(Homo sapiens (Human))
Shenyang Pharmaceutical University
Curated by ChEMBL
Shenyang Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 0.120nMAssay Description:The conditions for measuring inhibitory activity of compounds against LSD1 activity were determined with reference to a document available from the w...More data for this Ligand-Target Pair
TargetLysine-specific histone demethylase 1A(Homo sapiens (Human))
Shenyang Pharmaceutical University
Curated by ChEMBL
Shenyang Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 0.120nMAssay Description:The conditions for measuring inhibitory activity of compounds against LSD1 activity were determined with reference to a document available from the w...More data for this Ligand-Target Pair
TargetLysine-specific histone demethylase 1A(Homo sapiens (Human))
Shenyang Pharmaceutical University
Curated by ChEMBL
Shenyang Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 0.120nMAssay Description:The conditions for measuring inhibitory activity of compounds against LSD1 activity were determined with reference to a document available from the w...More data for this Ligand-Target Pair
TargetLysine-specific histone demethylase 1A(Homo sapiens (Human))
Shenyang Pharmaceutical University
Curated by ChEMBL
Shenyang Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 0.120nMAssay Description:The conditions for measuring inhibitory activity of compounds against LSD1 activity were determined with reference to a document available from the w...More data for this Ligand-Target Pair
TargetLysine-specific histone demethylase 1A(Homo sapiens (Human))
Shenyang Pharmaceutical University
Curated by ChEMBL
Shenyang Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 0.130nMAssay Description:The conditions for measuring inhibitory activity of compounds against LSD1 activity were determined with reference to a document available from the w...More data for this Ligand-Target Pair
TargetLysine-specific histone demethylase 1A(Homo sapiens (Human))
Shenyang Pharmaceutical University
Curated by ChEMBL
Shenyang Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 0.130nMAssay Description:The conditions for measuring inhibitory activity of compounds against LSD1 activity were determined with reference to a document available from the w...More data for this Ligand-Target Pair
TargetLysine-specific histone demethylase 1A(Homo sapiens (Human))
Shenyang Pharmaceutical University
Curated by ChEMBL
Shenyang Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 0.130nMAssay Description:The conditions for measuring inhibitory activity of compounds against LSD1 activity were determined with reference to a document available from the w...More data for this Ligand-Target Pair
TargetLysine-specific histone demethylase 1A(Homo sapiens (Human))
Shenyang Pharmaceutical University
Curated by ChEMBL
Shenyang Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 0.130nMAssay Description:Inhibition of recombinant human LSD1 expressed in Escherichia coli using biotinylated H3K4me as substrate by TR-FRET assayMore data for this Ligand-Target Pair
TargetLysine-specific histone demethylase 1A(Homo sapiens (Human))
Shenyang Pharmaceutical University
Curated by ChEMBL
Shenyang Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 0.130nMAssay Description:The conditions for measuring inhibitory activity of compounds against LSD1 activity were determined with reference to a document available from the w...More data for this Ligand-Target Pair
TargetLysine-specific histone demethylase 1A(Homo sapiens (Human))
Shenyang Pharmaceutical University
Curated by ChEMBL
Shenyang Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 0.130nMAssay Description:Inhibition of recombinant human LSD1 expressed in Escherichia coli using biotinylated H3K4me as substrate by TR-FRET assayMore data for this Ligand-Target Pair
TargetLysine-specific histone demethylase 1A(Homo sapiens (Human))
Shenyang Pharmaceutical University
Curated by ChEMBL
Shenyang Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 0.140nMAssay Description:The conditions for measuring inhibitory activity of compounds against LSD1 activity were determined with reference to a document available from the w...More data for this Ligand-Target Pair
TargetLysine-specific histone demethylase 1A(Homo sapiens (Human))
Shenyang Pharmaceutical University
Curated by ChEMBL
Shenyang Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 0.140nMAssay Description:The conditions for measuring inhibitory activity of compounds against LSD1 activity were determined with reference to a document available from the w...More data for this Ligand-Target Pair
TargetLysine-specific histone demethylase 1A(Homo sapiens (Human))
Shenyang Pharmaceutical University
Curated by ChEMBL
Shenyang Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 0.140nMAssay Description:The conditions for measuring inhibitory activity of compounds against LSD1 activity were determined with reference to a document available from the w...More data for this Ligand-Target Pair
TargetLysine-specific histone demethylase 1A(Homo sapiens (Human))
Shenyang Pharmaceutical University
Curated by ChEMBL
Shenyang Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 0.140nMAssay Description:Inhibition of KDM1A (unknown origin)More data for this Ligand-Target Pair
TargetLysine-specific histone demethylase 1A(Homo sapiens (Human))
Shenyang Pharmaceutical University
Curated by ChEMBL
Shenyang Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 0.140nMAssay Description:The conditions for measuring inhibitory activity of compounds against LSD1 activity were determined with reference to a document available from the w...More data for this Ligand-Target Pair
TargetLysine-specific histone demethylase 1A(Homo sapiens (Human))
Shenyang Pharmaceutical University
Curated by ChEMBL
Shenyang Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 0.140nMAssay Description:The conditions for measuring inhibitory activity of compounds against LSD1 activity were determined with reference to a document available from the w...More data for this Ligand-Target Pair
TargetLysine-specific histone demethylase 1A(Homo sapiens (Human))
Shenyang Pharmaceutical University
Curated by ChEMBL
Shenyang Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 0.140nMAssay Description:The conditions for measuring inhibitory activity of compounds against LSD1 activity were determined with reference to a document available from the w...More data for this Ligand-Target Pair
TargetLysine-specific histone demethylase 1A(Homo sapiens (Human))
Shenyang Pharmaceutical University
Curated by ChEMBL
Shenyang Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 0.140nMAssay Description:Inhibition of recombinant human LSD1 expressed in Escherichia coli using biotinylated H3K4me as substrate by TR-FRET assayMore data for this Ligand-Target Pair
TargetLysine-specific histone demethylase 1A(Homo sapiens (Human))
Shenyang Pharmaceutical University
Curated by ChEMBL
Shenyang Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 0.140nMAssay Description:Inhibition of LSD1 (unknown origin)More data for this Ligand-Target Pair
TargetLysine-specific histone demethylase 1A(Homo sapiens (Human))
Shenyang Pharmaceutical University
Curated by ChEMBL
Shenyang Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 0.140nMAssay Description:The conditions for measuring inhibitory activity of compounds against LSD1 activity were determined with reference to a document available from the w...More data for this Ligand-Target Pair
TargetLysine-specific histone demethylase 1A(Homo sapiens (Human))
Shenyang Pharmaceutical University
Curated by ChEMBL
Shenyang Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 0.140nMAssay Description:The conditions for measuring inhibitory activity of compounds against LSD1 activity were determined with reference to a document available from the w...More data for this Ligand-Target Pair
TargetLysine-specific histone demethylase 1A(Homo sapiens (Human))
Shenyang Pharmaceutical University
Curated by ChEMBL
Shenyang Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 0.140nMAssay Description:The conditions for measuring inhibitory activity of compounds against LSD1 activity were determined with reference to a document available from the w...More data for this Ligand-Target Pair
TargetLysine-specific histone demethylase 1A(Homo sapiens (Human))
Shenyang Pharmaceutical University
Curated by ChEMBL
Shenyang Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 0.140nMAssay Description:The conditions for measuring inhibitory activity of compounds against LSD1 activity were determined with reference to a document available from the w...More data for this Ligand-Target Pair
TargetLysine-specific histone demethylase 1A(Homo sapiens (Human))
Shenyang Pharmaceutical University
Curated by ChEMBL
Shenyang Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 0.140nMAssay Description:The conditions for measuring inhibitory activity of compounds against LSD1 activity were determined with reference to a document available from the w...More data for this Ligand-Target Pair