Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Ectonucleoside triphosphate diphosphohydrolase 8
Ligand
BDBM50179360
Substrate
n/a
Meas. Tech.
ChEBML_1716387
IC50
>100000±n/a nM
Citation
 Kuhrt, D; Ejaz, SA; Afzal, S; Khan, SU; Lecka, J; S�vigny, J; Ehlers, P; Spannenberg, A; Iqbal, J; Langer, P Chemoselective synthesis and biological evaluation of arylated 2-(Trifluoromethyl) quinolines as nucleotide pyrophosphatase (NPPs) inhibitors. Eur J Med Chem 138:816-829 (2017) [PubMed] Article
Kuhrt, D; Ejaz, SA; Afzal, S; Khan, SU; Lecka, J; S�vigny, J; Ehlers, P; Spannenberg, A; Iqbal, J; Langer, P Chemoselective synthesis and biological evaluation of arylated 2-(Trifluoromethyl) quinolines as nucleotide pyrophosphatase (NPPs) inhibitors. Eur J Med Chem 138:816-829 (2017) [PubMed] Article More Info.:
Target
Name:
Ectonucleoside triphosphate diphosphohydrolase 8
Synonyms:
ENTP8_HUMAN | ENTPD8
Type:
PROTEIN
Mol. Mass.:
53895.40
Organism:
Homo sapiens (Human)
Description:
ChEMBL_514295
Residue:
495
Sequence:
MGLSRKEQVFLALLGASGVSGLTALILLLVEATSVLLPTDIKFGIVFDAGSSHTSLFLYQWLANKENGTGVVSQALACQVEGPGISSYTSNAAQAGESLQGCLEEALVLIPEAQHRKTPTFLGATAGMRLLSRKNSSQARDIFAAVTQVLGRSPVDFWGAELLAGQAEGAFGWITVNYGLGTLVKYSFTGEWIQPPEEMLVGALDMGGASTQITFVPGGPILDKSTQADFRLYGSDYSVYTHSYLCFGRDQMLSRLLVGLVQSRPAALLRHPCYLSGYQTTLALGPLYESPCVHATPPLSLPQNLTVEGTGNPGACVSAIRELFNFSSCQGQEDCAFDGVYQPPLRGQFYAFSNFYYTFHFLNLTSRQPLSTVNATIWEFCQRPWKLVEASYPGQDRWLRDYCASGLYILTLLHEGYGFSEETWPSLEFRKQAGGVDIGWTLGYMLNLTGMIPADAPAQWRAESYGVWVAKVVFMVLALVAVVGAALVQLFWLQD
Inhibitor
Name:
BDBM50179360
Synonyms:
CHEMBL3040216
Type:
Small organic molecule
Emp. Form.:
C51H40N6O23S6
Mol. Mass.:
1297.28
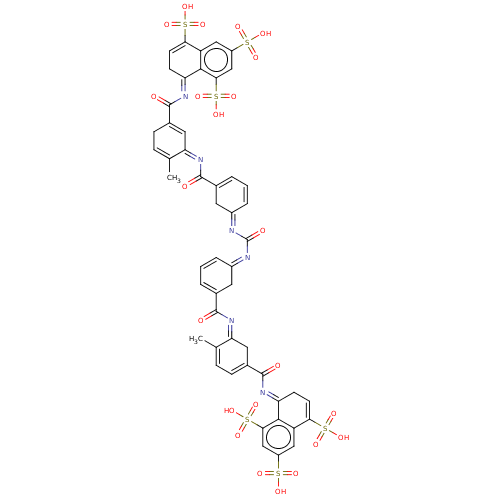
SMILES:
CC1=CCC(=C\C1=N\C(=O)C1=CC=C\C(C1)=N/C(=O)/N=C1/CC(=CC=C1)C(=O)\N=C1\CC(=CC=C1C)C(=O)\N=C1/CC=C(c2cc(cc(c12)S(O)(=O)=O)S(O)(=O)=O)S(O)(=O)=O)C(=O)\N=C1/CC=C(c2cc(cc(c12)S(O)(=O)=O)S(O)(=O)=O)S(O)(=O)=O |c:4,13,24,26,34,36,45,72,t:1,11|