Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
GTPase KRas
Ligand
BDBM50527058
Substrate
n/a
Meas. Tech.
ChEMBL_1901970 (CHEMBL4404192)
IC50
52±n/a nM
Citation
 Kettle, JG; Bagal, SK; Bickerton, S; Bodnarchuk, MS; Breed, J; Carbajo, RJ; Cassar, DJ; Chakraborty, A; Cosulich, S; Cumming, I; Davies, M; Eatherton, A; Evans, L; Feron, L; Fillery, S; Gleave, ES; Goldberg, FW; Harlfinger, S; Hanson, L; Howard, M; Howells, R; Jackson, A; Kemmitt, P; Kingston, JK; Lamont, S; Lewis, HJ; Li, S; Liu, L; Ogg, D; Phillips, C; Polanski, R; Robb, G; Robinson, D; Ross, S; Smith, JM; Tonge, M; Whiteley, R; Yang, J; Zhang, L; Zhao, X Structure-Based Design and Pharmacokinetic Optimization of Covalent Allosteric Inhibitors of the Mutant GTPase KRAS J Med Chem 63:4468-4483 (2020) [PubMed] Article
Kettle, JG; Bagal, SK; Bickerton, S; Bodnarchuk, MS; Breed, J; Carbajo, RJ; Cassar, DJ; Chakraborty, A; Cosulich, S; Cumming, I; Davies, M; Eatherton, A; Evans, L; Feron, L; Fillery, S; Gleave, ES; Goldberg, FW; Harlfinger, S; Hanson, L; Howard, M; Howells, R; Jackson, A; Kemmitt, P; Kingston, JK; Lamont, S; Lewis, HJ; Li, S; Liu, L; Ogg, D; Phillips, C; Polanski, R; Robb, G; Robinson, D; Ross, S; Smith, JM; Tonge, M; Whiteley, R; Yang, J; Zhang, L; Zhao, X Structure-Based Design and Pharmacokinetic Optimization of Covalent Allosteric Inhibitors of the Mutant GTPase KRAS J Med Chem 63:4468-4483 (2020) [PubMed] Article More Info.:
Target
Name:
GTPase KRas
Synonyms:
GTPase KRas, N-terminally processed | K-Ras 2 | KRAS | KRAS2 | Ki-Ras | RASK2 | RASK_HUMAN | c-K-ras | c-Ki-ras
Type:
PROTEIN
Mol. Mass.:
21656.10
Organism:
Homo sapiens (Human)
Description:
ChEMBL_1476955
Residue:
189
Sequence:
MTEYKLVVVGAGGVGKSALTIQLIQNHFVDEYDPTIEDSYRKQVVIDGETCLLDILDTAGQEEYSAMRDQYMRTGEGFLCVFAINNTKSFEDIHHYREQIKRVKDSEDVPMVLVGNKCDLPSRTVDTKQAQDLARSYGIPFIETSAKTRQRVEDAFYTLVREIRQYRLKKISKEEKTPGCVKIKKCIIM
Inhibitor
Name:
BDBM50527058
Synonyms:
CHEMBL4455191
Type:
Small organic molecule
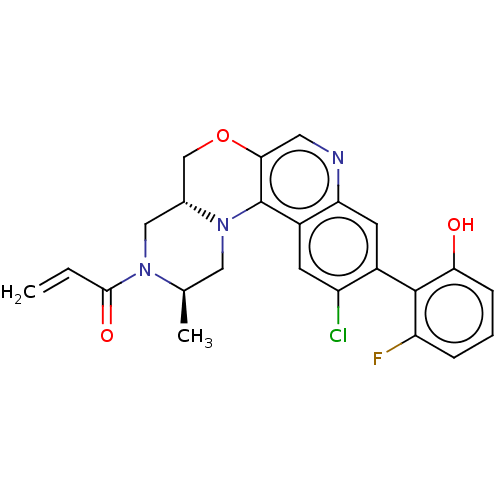
Emp. Form.:
C24H21ClFN3O3
Mol. Mass.:
453.893
SMILES:
[H][C@@]12COc3cnc4cc(c(Cl)cc4c3N1C[C@@H](C)N(C2)C(=O)C=C)-c1c(O)cccc1F |r,wU:1.0,17.20,(70.37,-6.5,;69.05,-7.29,;70.38,-8.05,;70.39,-9.59,;69.06,-10.36,;69.07,-11.91,;67.74,-12.69,;66.4,-11.92,;65.07,-12.7,;63.73,-11.93,;63.73,-10.38,;62.4,-9.61,;65.06,-9.61,;66.4,-10.38,;67.72,-9.6,;67.72,-8.06,;66.38,-7.3,;66.37,-5.76,;65.03,-5,;67.7,-4.98,;69.04,-5.75,;67.69,-3.44,;66.35,-2.68,;69.02,-2.67,;69.01,-1.13,;62.4,-12.7,;61.07,-11.92,;61.07,-10.38,;59.73,-12.69,;59.73,-14.23,;61.08,-15,;62.4,-14.23,;63.74,-15,)|