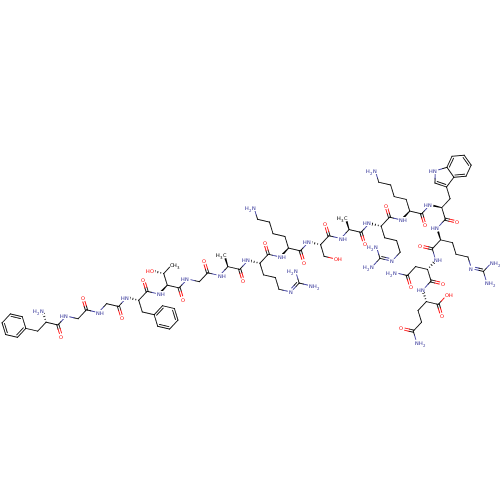
Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Nociceptin receptor
Ligand
BDBM50296841
Substrate
n/a
Meas. Tech.
ChEMBL_582618 (CHEMBL1054181)
IC50
0.23±n/a nM
Citation
 Nishimura, H; Li, J; Isozaki, K; Okada, K; Matsushima, A; Nose, T; Costa, T; Shimohigashi, Y Discriminatory synergistic effect of Trp-substitutions in superagonist [(Arg/Lys)(14), (Arg/Lys)(15)]nociceptin on ORL1 receptor binding and activation. Bioorg Med Chem 17:5683-7 (2009) [PubMed] Article
Nishimura, H; Li, J; Isozaki, K; Okada, K; Matsushima, A; Nose, T; Costa, T; Shimohigashi, Y Discriminatory synergistic effect of Trp-substitutions in superagonist [(Arg/Lys)(14), (Arg/Lys)(15)]nociceptin on ORL1 receptor binding and activation. Bioorg Med Chem 17:5683-7 (2009) [PubMed] Article More Info.:
Target
Name:
Nociceptin receptor
Synonyms:
Nociceptin/Orphanin FQ, NOP receptor | Nociceptin/mu opioid receptor | OPRX_RAT | Oor | Oprl | Oprl1
Type:
Enzyme Catalytic Domain
Mol. Mass.:
40531.08
Organism:
RAT
Description:
Nociceptin/Orphanin FQ, NOP receptor 0 RAT::P35370
Residue:
367
Sequence:
MESLFPAPYWEVLYGSHFQGNLSLLNETVPHHLLLNASHSAFLPLGLKVTIVGLYLAVCIGGLLGNCLVMYVILRHTKMKTATNIYIFNLALADTLVLLTLPFQGTDILLGFWPFGNALCKTVIAIDYYNMFTSTFTLTAMSVDRYVAICHPIRALDVRTSSKAQAVNVAIWALASVVGVPVAIMGSAQVEDEEIECLVEIPAPQDYWGPVFAICIFLFSFIIPVLIISVCYSLMIRRLRGVRLLSGSREKDRNLRRITRLVLVVVAVFVGCWTPVQVFVLVQGLGVQPGSETAVAILRFCTALGYVNSCLNPILYAFLDENFKACFRKFCCASSLHREMQVSDRVRSIAKDVGLGCKTSETVPRPA
Inhibitor
Name:
BDBM50296841
Synonyms:
CHEMBL556388 | FGGFTGARKSARKWRNQ
Type:
Small organic molecule
Emp. Form.:
C87H135N31O22
Mol. Mass.:
1967.1973
SMILES:
C[C@@H](O)[C@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)CNC(=O)CNC(=O)[C@@H](N)Cc1ccccc1)C(=O)NCC(=O)N[C@@H](C)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CO)C(=O)N[C@@H](C)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCC(N)=O)C(O)=O |r,wU:1.1,73.75,89.90,42.44,112.115,26.27,131.134,58.59,3.3,wD:78.79,7.15,47.48,98.99,123.126,67.68,(7.64,-2.95,;8.97,-3.72,;10.31,-2.96,;8.96,-5.26,;7.62,-6.03,;6.29,-5.25,;6.3,-3.71,;4.95,-6.01,;4.94,-7.55,;6.27,-8.33,;6.25,-9.86,;7.58,-10.64,;8.92,-9.88,;8.93,-8.33,;7.61,-7.57,;3.62,-5.23,;2.29,-6,;2.28,-7.54,;.96,-5.22,;-.38,-5.98,;-1.71,-5.2,;-1.7,-3.66,;-3.05,-5.97,;-4.38,-5.19,;-5.72,-5.95,;-5.73,-7.49,;-7.04,-5.17,;-8.38,-5.94,;-7.03,-3.63,;-8.36,-2.86,;-8.36,-1.31,;-9.69,-.53,;-11.03,-1.3,;-11.03,-2.85,;-9.7,-3.61,;10.29,-6.04,;10.28,-7.58,;11.63,-5.28,;12.96,-6.06,;14.29,-5.29,;14.3,-3.75,;15.62,-6.07,;16.96,-5.31,;16.97,-3.77,;18.29,-6.09,;18.28,-7.63,;19.63,-5.32,;20.96,-6.1,;20.95,-7.64,;19.61,-8.4,;19.6,-9.94,;18.26,-10.71,;18.26,-12.25,;16.92,-13.01,;19.59,-13.02,;22.3,-5.34,;22.31,-3.8,;23.63,-6.12,;24.96,-5.35,;24.97,-3.81,;26.31,-3.05,;26.32,-1.51,;27.66,-.75,;27.67,.79,;26.29,-6.13,;26.28,-7.67,;27.63,-5.37,;28.96,-6.15,;28.95,-7.69,;27.61,-8.45,;30.3,-5.38,;30.31,-3.84,;31.63,-6.16,;32.97,-5.4,;32.98,-3.86,;34.29,-6.18,;34.28,-7.72,;35.63,-5.41,;36.96,-6.19,;36.95,-7.73,;35.61,-8.49,;35.61,-10.03,;34.27,-10.8,;34.26,-12.34,;32.93,-13.1,;35.59,-13.11,;38.3,-5.43,;38.31,-3.89,;39.63,-6.21,;40.97,-5.44,;40.98,-3.9,;42.32,-3.14,;42.33,-1.6,;43.67,-.84,;43.68,.7,;42.3,-6.22,;42.29,-7.76,;43.64,-5.46,;44.96,-6.24,;44.95,-7.78,;46.28,-8.55,;47.68,-7.93,;48.7,-9.08,;47.93,-10.4,;48.39,-11.85,;47.36,-12.97,;45.87,-12.64,;45.41,-11.19,;46.44,-10.07,;46.3,-5.47,;46.31,-3.93,;47.63,-6.25,;48.97,-5.49,;48.98,-3.95,;50.31,-3.19,;50.32,-1.66,;51.65,-.9,;51.66,.63,;52.99,1.39,;50.34,1.41,;50.3,-6.27,;50.29,-7.81,;51.64,-5.51,;52.97,-6.28,;52.96,-7.82,;54.29,-8.61,;55.62,-7.84,;54.28,-10.15,;54.31,-5.53,;54.32,-3.99,;55.63,-6.3,;56.97,-5.54,;56.98,-4,;58.32,-3.23,;58.33,-1.69,;57,-.92,;59.67,-.94,;58.3,-6.31,;59.64,-5.56,;58.29,-7.85,)|