Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Cytochrome P450 2C9
Ligand
BDBM21278
Substrate
n/a
Meas. Tech.
ChEMBL_651864 (CHEMBL1227229)
IC50
5.3±n/a nM
Citation
 Lee, J; Seo, HJ; Lee, SH; Kim, J; Jung, ME; Lee, SH; Song, KS; Lee, J; Kang, SY; Kim, MJ; Kim, MS; Son, EJ; Lee, M; Han, HK Discovery of 2-(4-((1H-1,2,4-triazol-1-yl)methyl)-5-(4-bromophenyl)-1-(2-chlorophenyl)-1H-pyrazol-3-yl)-5-tert-butyl-1,3,4-thiadiazole (GCC2680) as a potent, selective and orally efficacious cannabinoid-1 receptor antagonist. Bioorg Med Chem 18:6377-88 (2010) [PubMed] Article
Lee, J; Seo, HJ; Lee, SH; Kim, J; Jung, ME; Lee, SH; Song, KS; Lee, J; Kang, SY; Kim, MJ; Kim, MS; Son, EJ; Lee, M; Han, HK Discovery of 2-(4-((1H-1,2,4-triazol-1-yl)methyl)-5-(4-bromophenyl)-1-(2-chlorophenyl)-1H-pyrazol-3-yl)-5-tert-butyl-1,3,4-thiadiazole (GCC2680) as a potent, selective and orally efficacious cannabinoid-1 receptor antagonist. Bioorg Med Chem 18:6377-88 (2010) [PubMed] Article More Info.:
Target
Name:
Cytochrome P450 2C9
Synonyms:
(R)-limonene 6-monooxygenase | (S)-limonene 6-monooxygenase | CP2C9_HUMAN | CYP2C10 | CYP2C9 | CYPIIC9 | Cytochrome P450 2C9 (CYP2C9 ) | Cytochrome P450 2C9 (CYP2C9) | P-450MP | P450 MP-4/MP-8 | P450 PB-1 | S-mephenytoin 4-hydroxylase
Type:
Enzyme
Mol. Mass.:
55636.33
Organism:
Homo sapiens (Human)
Description:
P11712
Residue:
490
Sequence:
MDSLVVLVLCLSCLLLLSLWRQSSGRGKLPPGPTPLPVIGNILQIGIKDISKSLTNLSKVYGPVFTLYFGLKPIVVLHGYEAVKEALIDLGEEFSGRGIFPLAERANRGFGIVFSNGKKWKEIRRFSLMTLRNFGMGKRSIEDRVQEEARCLVEELRKTKASPCDPTFILGCAPCNVICSIIFHKRFDYKDQQFLNLMEKLNENIKILSSPWIQICNNFSPIIDYFPGTHNKLLKNVAFMKSYILEKVKEHQESMDMNNPQDFIDCFLMKMEKEKHNQPSEFTIESLENTAVDLFGAGTETTSTTLRYALLLLLKHPEVTAKVQEEIERVIGRNRSPCMQDRSHMPYTDAVVHEVQRYIDLLPTSLPHAVTCDIKFRNYLIPKGTTILISLTSVLHDNKEFPNPEMFDPHHFLDEGGNFKKSKYFMPFSAGKRICVGEALAGMELFLFLTSILQNFNLKSLVDPKNLDTTPVVNGFASVPPFYQLCFIPV
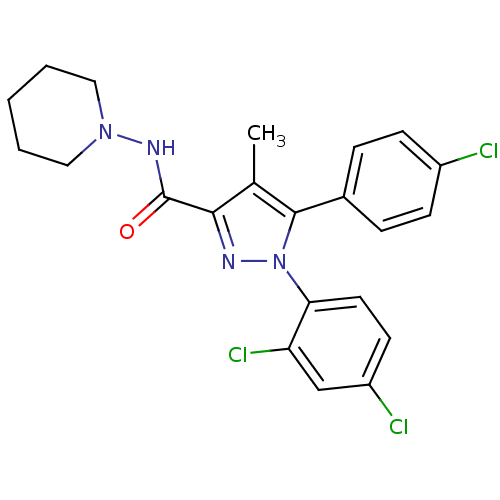
Inhibitor
Name:
BDBM21278
Synonyms:
5-(4-chlorophenyl)-1-(2,4-dichlorophenyl)-4-methyl-N-(piperidin-1-yl)-1H-pyrazole-3-carboxamide | Acomplia | CHEMBL111 | CHEMBL558598 | RIMONABANT HYDROCHLORIDE | Rimonabant | SR 141716A | SR141716 | SR141716A | [3H]Rimonabant | [3H]SR141716A
Type:
Small organic molecule
Emp. Form.:
C22H21Cl3N4O
Mol. Mass.:
463.787
SMILES:
Cc1c(nn(c1-c1ccc(Cl)cc1)-c1ccc(Cl)cc1Cl)C(=O)NN1CCCCC1