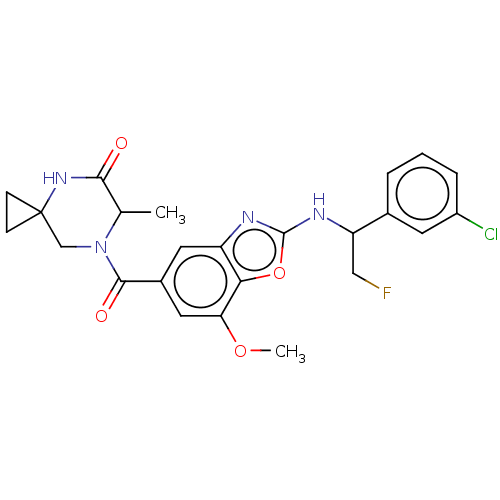
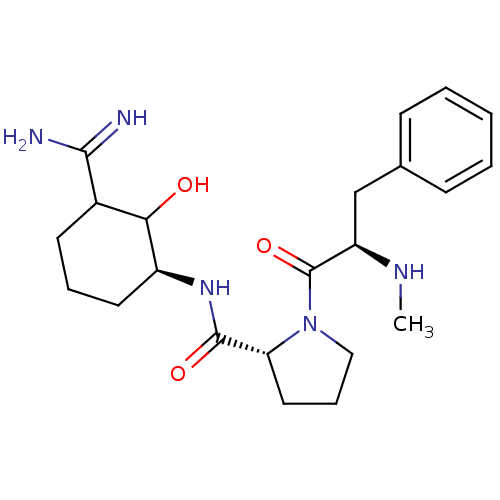
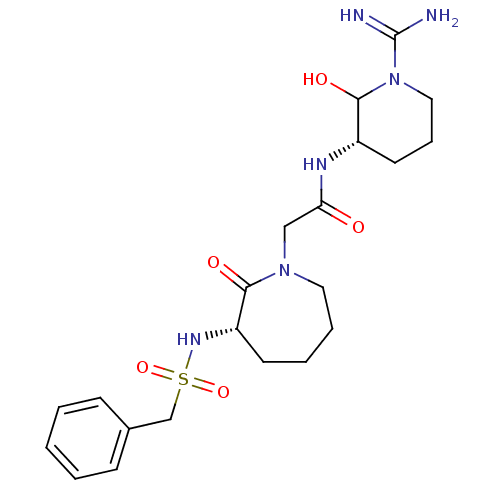
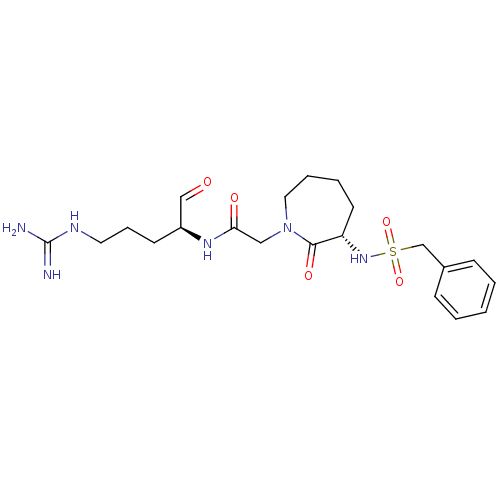
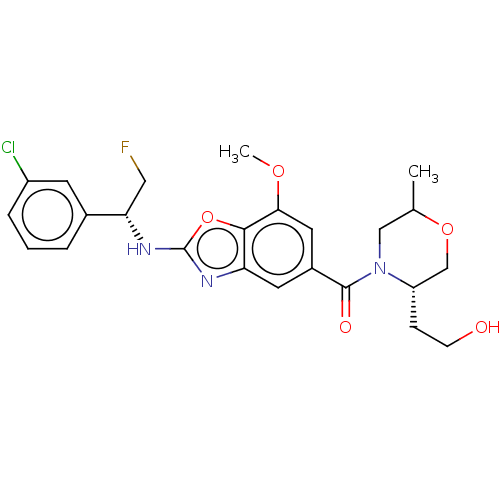
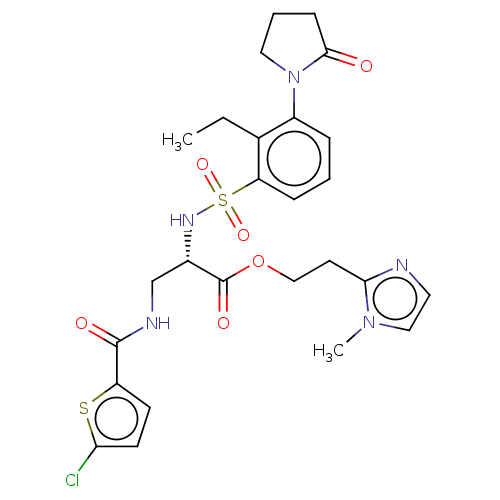
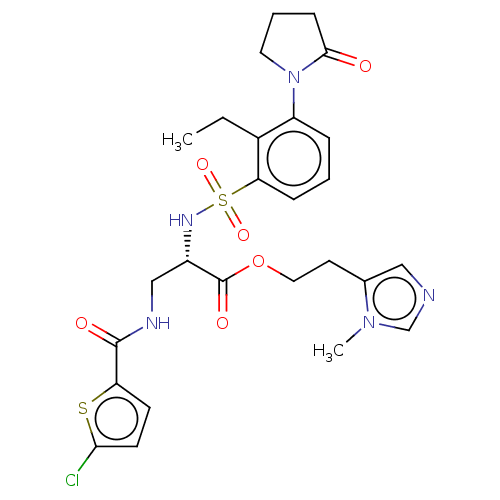
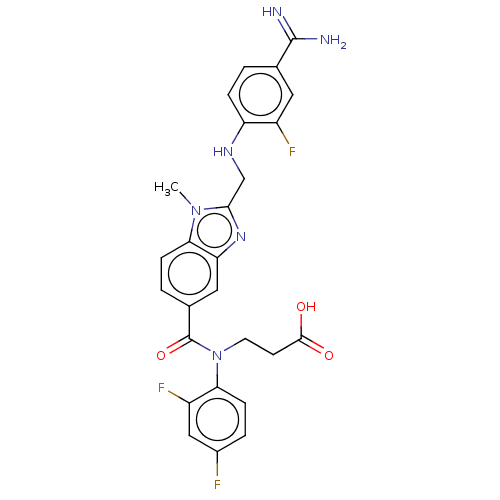
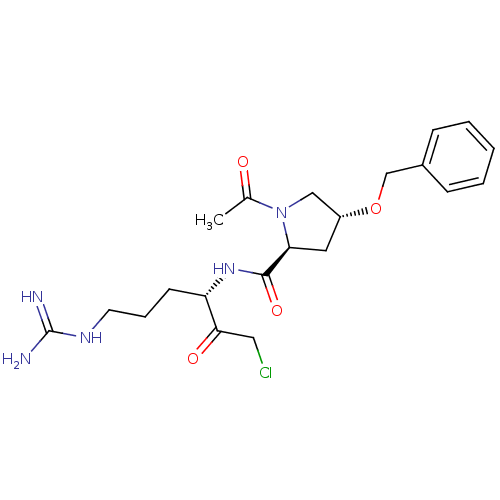
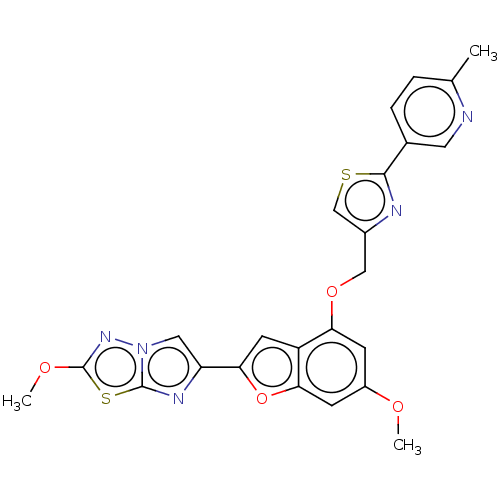
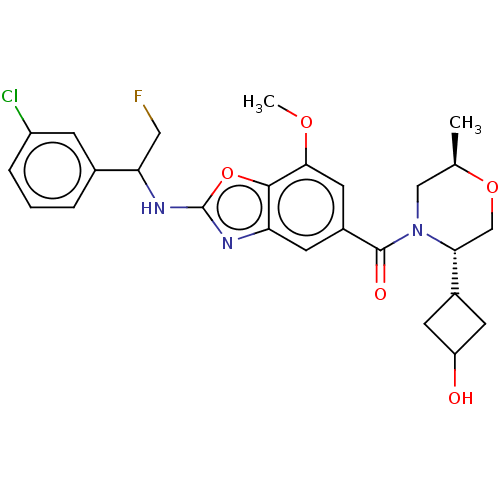
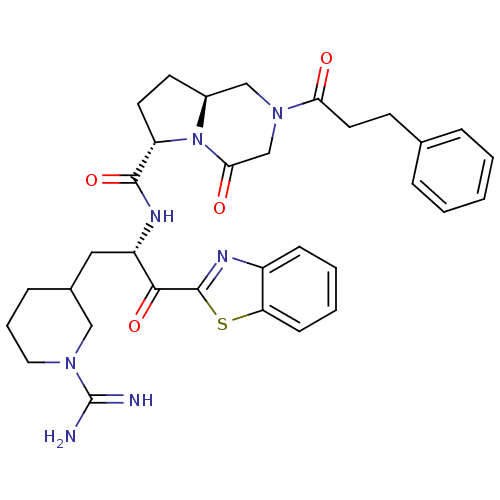
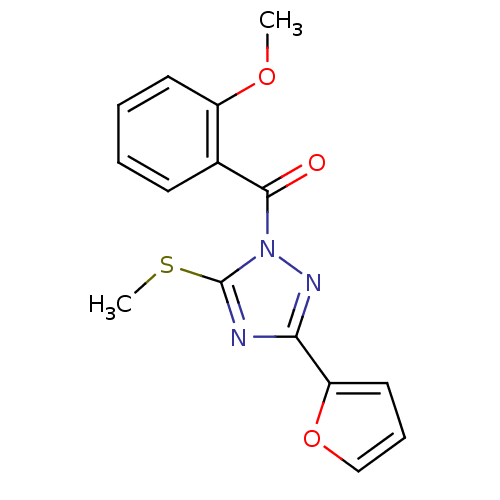
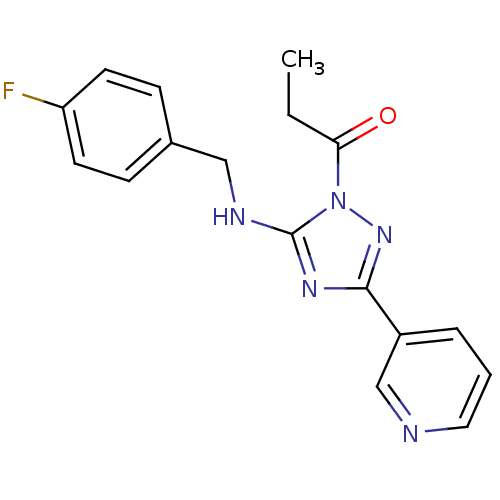
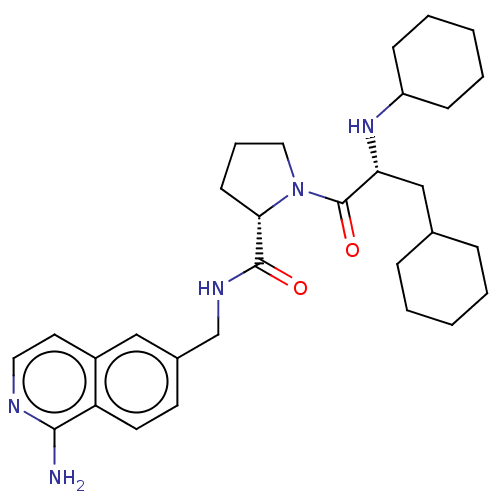
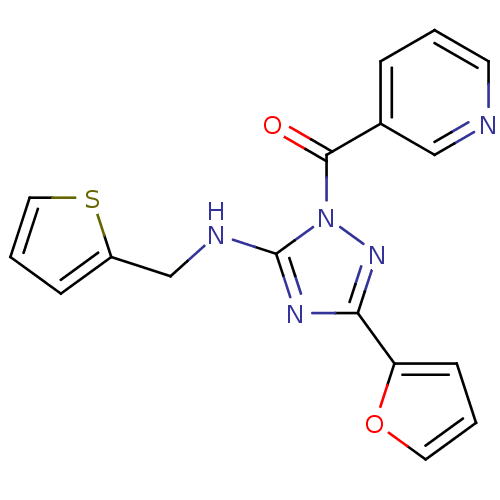
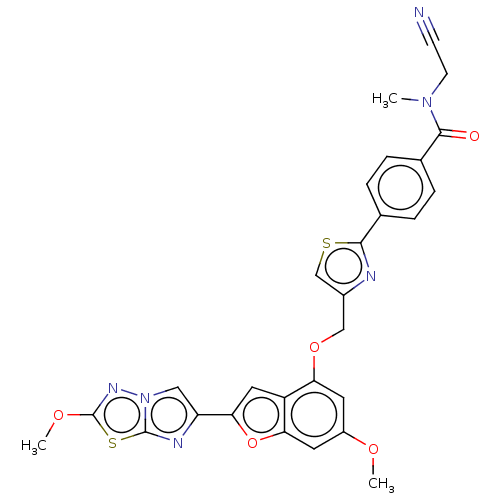
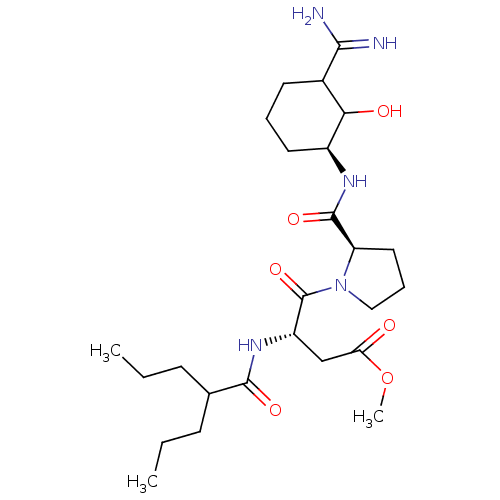
Affinity DataIC50: 0.00170nMAssay Description:In vitro inhibitory concentration of compound against human thrombinMore data for this Ligand-Target Pair
Affinity DataIC50: 0.00800nMAssay Description:Concentration of the compound required to inhibit thrombin was determinedMore data for this Ligand-Target Pair
Affinity DataIC50: 0.0100nMpH: 7.4 T: 2°CAssay Description:To determine the thrombin inhibition of the substances listed above, a biochemical test system is constructed in which the conversion of a thrombin s...More data for this Ligand-Target Pair
Affinity DataIC50: 0.0300nMAssay Description:Concentration of the compound required to inhibit thrombin was determinedMore data for this Ligand-Target Pair
Affinity DataIC50: 0.100nMAssay Description:Inhibitory activity against thrombinMore data for this Ligand-Target Pair
Affinity DataIC50: 0.100nMpH: 7.4 T: 2°CAssay Description:To determine the thrombin inhibition of the substances listed above, a biochemical test system is constructed in which the conversion of a thrombin s...More data for this Ligand-Target Pair
Affinity DataIC50: 0.100nMAssay Description:In vitro activity of the compound against human alpha thrombin was determinedMore data for this Ligand-Target Pair
Affinity DataIC50: 0.103nMAssay Description:Inhibition of human thrombin using Z-GPR-AMC as substrate after 30 mins by fluorimetric analysisMore data for this Ligand-Target Pair
Affinity DataIC50: 0.160nMpH: 7.4 T: 2°CAssay Description:To determine the thrombin inhibition of the substances listed above, a biochemical test system is constructed in which the conversion of a thrombin s...More data for this Ligand-Target Pair
Affinity DataIC50: 0.190nMAssay Description:Determination of the Selectivity To demonstrate the selectivity of the substances with respect to thrombin and factor Xa inhibition, the test substan...More data for this Ligand-Target Pair
Affinity DataIC50: 0.190nMpH: 7.4 T: 2°CAssay Description:To determine the thrombin inhibition of the substances listed above, a biochemical test system is constructed in which the conversion of a thrombin s...More data for this Ligand-Target Pair
Affinity DataIC50: 0.200nMpH: 7.4 T: 2°CAssay Description:To determine the thrombin inhibition of the substances listed above, a biochemical test system is constructed in which the conversion of a thrombin s...More data for this Ligand-Target Pair
Affinity DataIC50: 0.300nMpH: 7.4 T: 2°CAssay Description:To determine the thrombin inhibition of the substances listed above, a biochemical test system is constructed in which the conversion of a thrombin s...More data for this Ligand-Target Pair
Affinity DataIC50: 0.370nMpH: 7.4 T: 2°CAssay Description:To determine the thrombin inhibition of the substances listed above, a biochemical test system is constructed in which the conversion of a thrombin s...More data for this Ligand-Target Pair
Affinity DataIC50: 0.380nMpH: 7.4 T: 2°CAssay Description:To determine the thrombin inhibition of the substances listed above, a biochemical test system is constructed in which the conversion of a thrombin s...More data for this Ligand-Target Pair
Affinity DataIC50: 0.467nMAssay Description:Inhibitory concentration of the compound required to inhibit human thrombin enzyme was determinedMore data for this Ligand-Target Pair
Affinity DataIC50: 0.470nMpH: 7.4 T: 2°CAssay Description:To determine the thrombin inhibition of the substances listed above, a biochemical test system is constructed in which the conversion of a thrombin s...More data for this Ligand-Target Pair
Affinity DataIC50: 0.505nMAssay Description:Concentration required for the in vitro inhibitory activity against human enzyme, thrombin cleavage of the chromogenic substrateMore data for this Ligand-Target Pair
Affinity DataIC50: 0.505nMAssay Description:Inhibitory concentration of the compound required to inhibit human thrombin enzyme was determinedMore data for this Ligand-Target Pair
Affinity DataIC50: 0.540nMpH: 7.4 T: 2°CAssay Description:To determine the thrombin inhibition of the substances listed above, a biochemical test system is constructed in which the conversion of a thrombin s...More data for this Ligand-Target Pair
Affinity DataIC50: 0.670nMAssay Description:Compound was evaluated for inhibition of amidolytic activity for chromogenic substrate thrombin F11a.More data for this Ligand-Target Pair
Affinity DataIC50: 0.700nMpH: 7.4 T: 2°CAssay Description:To determine the thrombin inhibition of the substances listed above, a biochemical test system is constructed in which the conversion of a thrombin s...More data for this Ligand-Target Pair
Affinity DataIC50: 0.700nMAssay Description:In vitro for inhibition of serine protease thrombin(FIIa).More data for this Ligand-Target Pair
Affinity DataIC50: 0.710nMAssay Description:The compound was tested in vitro for inhibitory activity against Coagulation factor IIMore data for this Ligand-Target Pair
Affinity DataIC50: 0.710nMAssay Description:In vitro inhibitory activity of the compound against human enzyme thrombinMore data for this Ligand-Target Pair
Affinity DataIC50: 0.710nMAssay Description:In vitro for inhibition of serine protease thrombin(FIIa).More data for this Ligand-Target Pair
Affinity DataIC50: 0.710nMAssay Description:Compound was evaluated for inhibition of amidolytic activity for chromogenic substrate thrombin F11a.More data for this Ligand-Target Pair
Affinity DataIC50: 0.720nMpH: 7.4 T: 2°CAssay Description:To determine the thrombin inhibition of the substances listed above, a biochemical test system is constructed in which the conversion of a thrombin s...More data for this Ligand-Target Pair
Affinity DataIC50: 0.740nMAssay Description:Determination of the Selectivity To demonstrate the selectivity of the substances with respect to thrombin and factor Xa inhibition, the test substan...More data for this Ligand-Target Pair
Affinity DataIC50: 0.830nMAssay Description:Determination of the Selectivity To demonstrate the selectivity of the substances with respect to thrombin and factor Xa inhibition, the test substan...More data for this Ligand-Target Pair
Affinity DataIC50: 0.840nMAssay Description:Inhibition of human thrombin preincubated for 10 mins followed by Ac-FVR-AMC substrate addition measured every 20s for 10 mins by fluorescence assayMore data for this Ligand-Target Pair
Affinity DataIC50: 0.900nMAssay Description:Inhibition of thrombinMore data for this Ligand-Target Pair
Affinity DataIC50: 0.920nMAssay Description:Inhibition of human thrombin pre-incubated for 10 mins before Ac-FVR-AMC substrate addition and measured after 10 mins by fluorescence based assayMore data for this Ligand-Target Pair
Affinity DataIC50: 0.940nMT: 2°CAssay Description:The ability of the compounds of the current invention to inhibit platelet aggregation induced by gamma-thrombin was tested in a 96-well microplate ag...More data for this Ligand-Target Pair
Affinity DataIC50: 0.960nMT: 2°CAssay Description:The ability of the compounds of the current invention to inhibit platelet aggregation induced by gamma-thrombin was tested in a 96-well microplate ag...More data for this Ligand-Target Pair
Affinity DataIC50: 0.980nMAssay Description:Inhibition of thrombinMore data for this Ligand-Target Pair
Affinity DataIC50: 0.980nMpH: 7.4 T: 2°CAssay Description:To determine the thrombin inhibition of the substances listed above, a biochemical test system is constructed in which the conversion of a thrombin s...More data for this Ligand-Target Pair
Affinity DataIC50: <1nMAssay Description:Inhibition of human ThrombinMore data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:Inhibition of human alpha-thrombinMore data for this Ligand-Target Pair
Affinity DataIC50: <1nMAssay Description:Inhibition of human ThrombinMore data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:Inhibition of thrombin by chromogenic assayMore data for this Ligand-Target Pair
Affinity DataIC50: 1nMpH: 7.5 T: 2°CAssay Description:HTS was performed using 217,350 compounds of the MLSCN library individually plated into 10ul 1536 compound plates at a concentration of 2.5 mM each, ...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMpH: 7.5 T: 2°CAssay Description:HTS was performed using 217,350 compounds of the MLSCN library individually plated into 10ul 1536 compound plates at a concentration of 2.5 mM each, ...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:Inhibition of thrombin (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 1nMpH: 7.5 T: 2°CAssay Description:HTS was performed using 217,350 compounds of the MLSCN library individually plated into 10ul 1536 compound plates at a concentration of 2.5 mM each, ...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:In vitro inhibitory activity against thrombin(FIIa)More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:Inhibition of thrombin by chromogenic assayMore data for this Ligand-Target Pair
Affinity DataIC50: 1nMT: 2°CAssay Description:The ability of the compounds of the current invention to inhibit platelet aggregation induced by gamma-thrombin was tested in a 96-well microplate ag...More data for this Ligand-Target Pair
Affinity DataIC50: 1.10nMAssay Description:In vitro for inhibition of serine protease thrombin(FIIa).More data for this Ligand-Target Pair

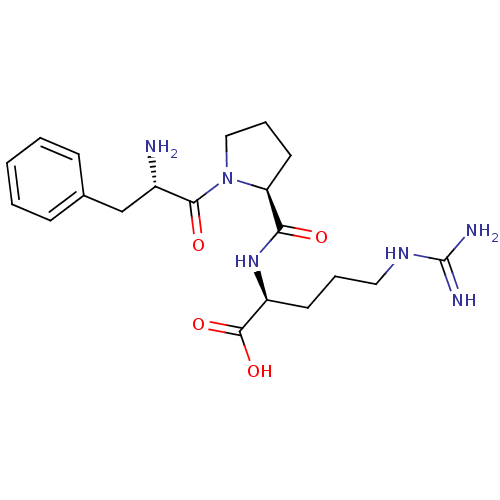
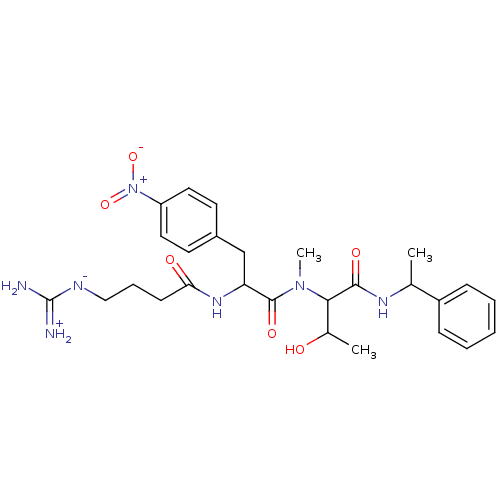
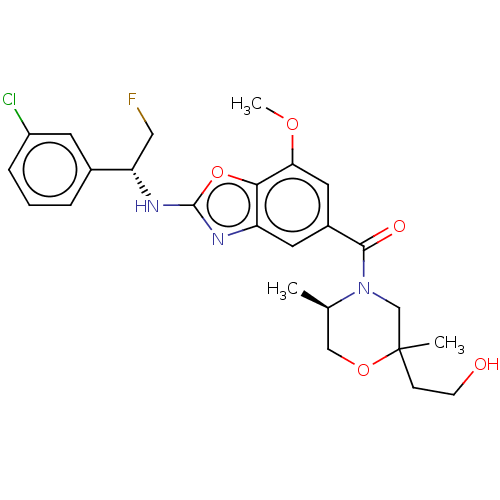
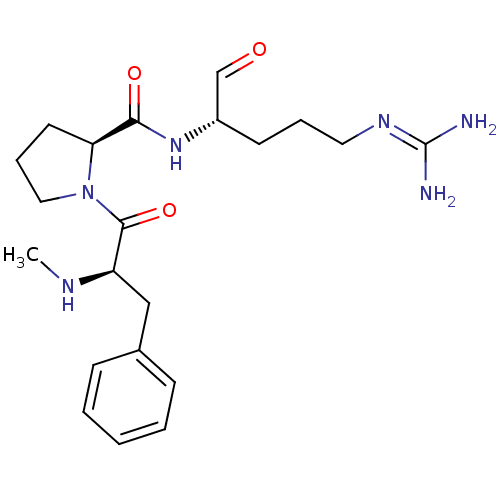
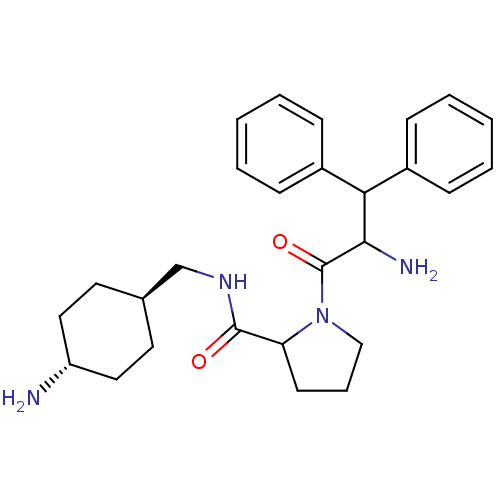
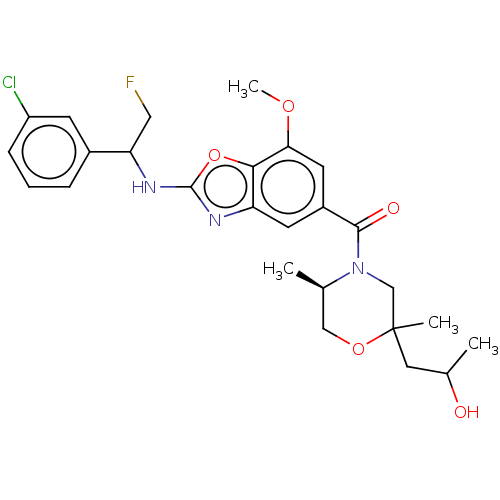
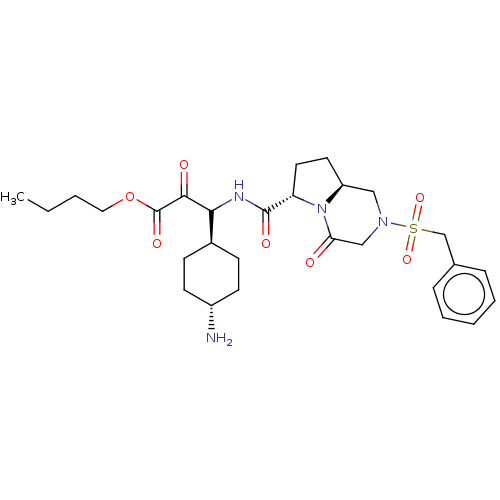
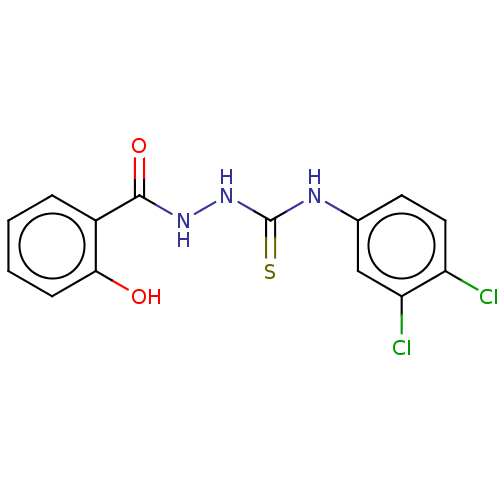

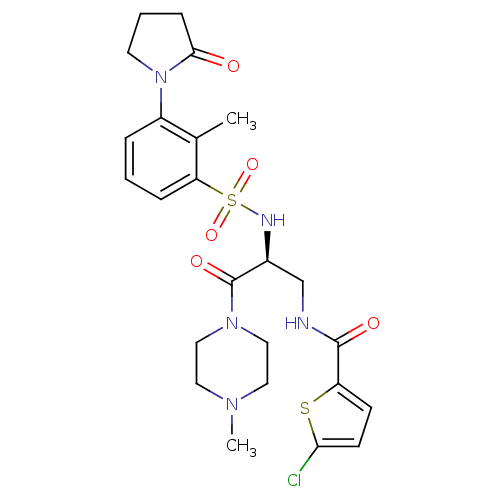
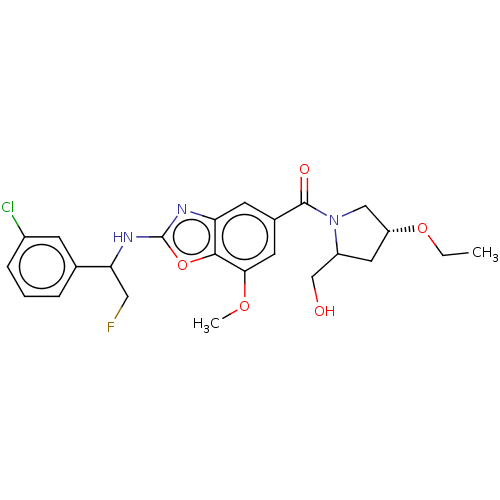
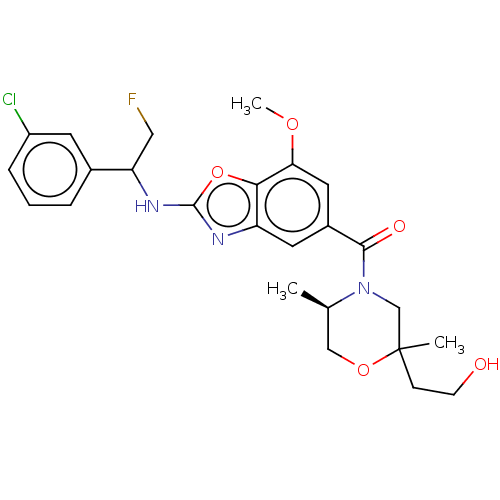
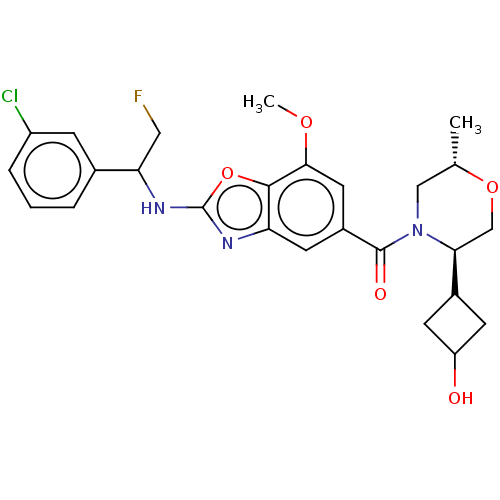
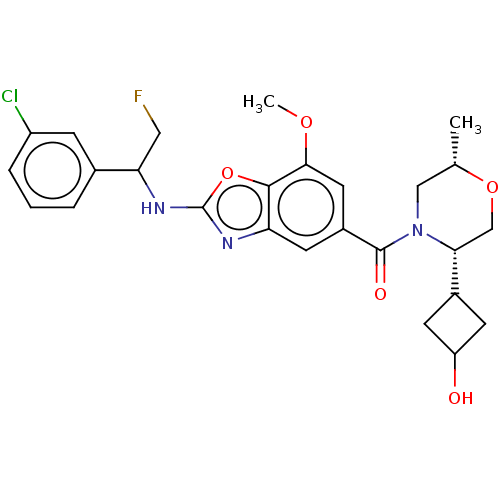
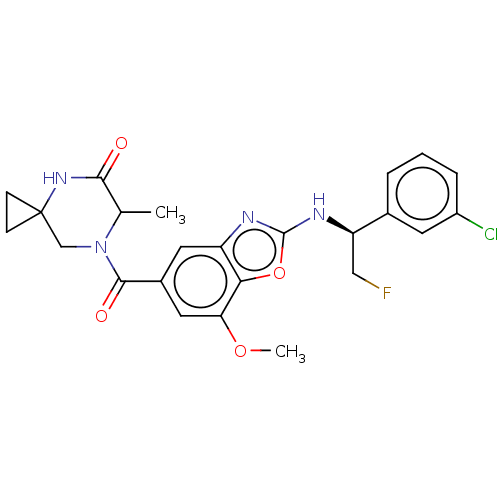
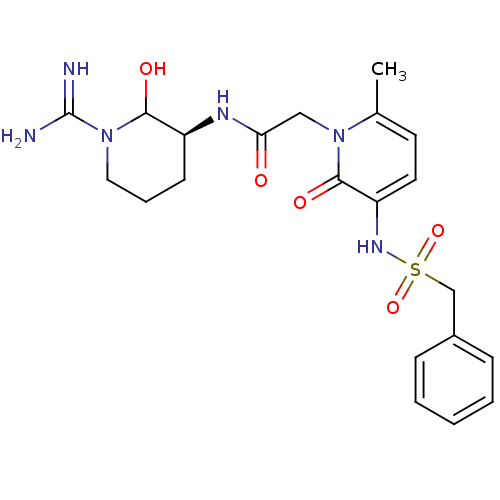
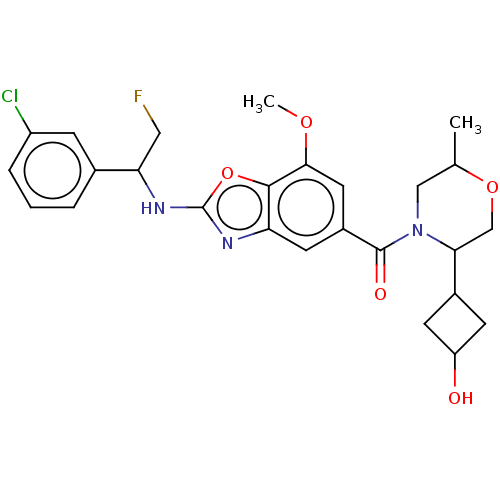
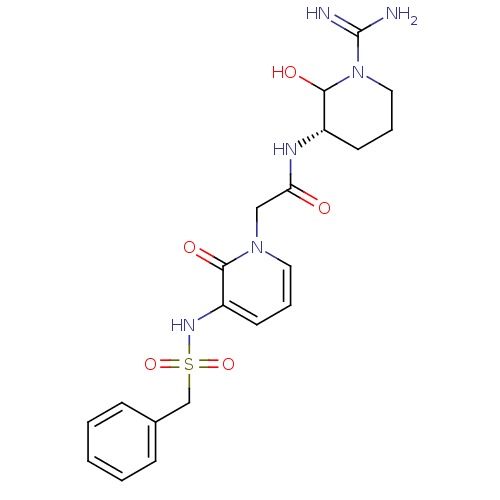
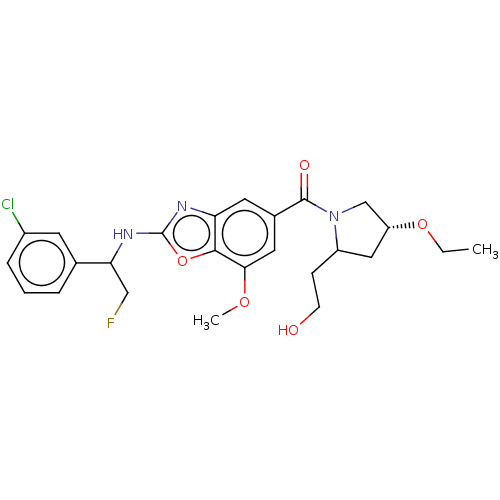
 3D Structure (crystal)
3D Structure (crystal)