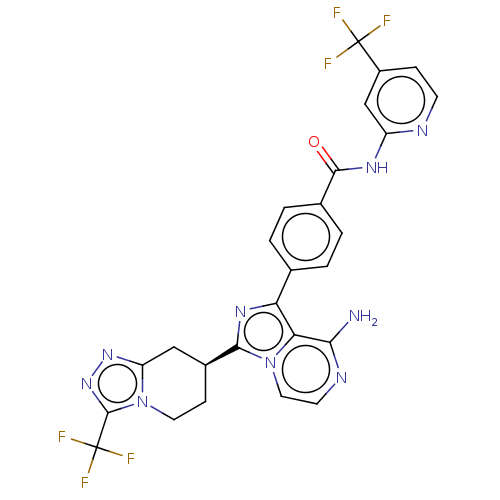
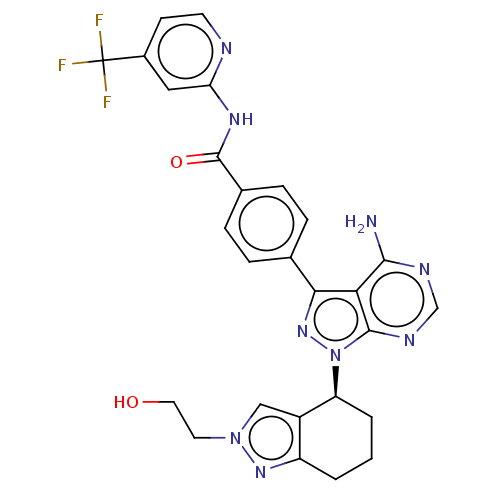
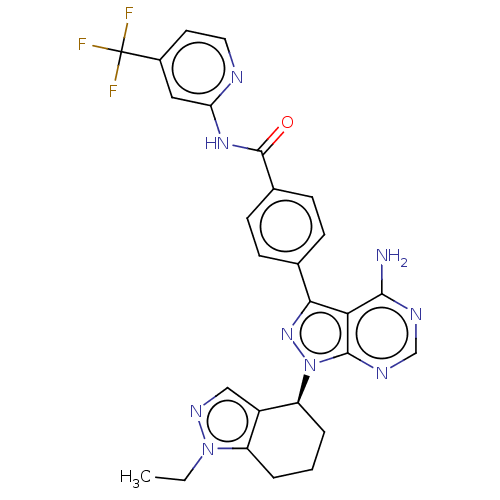
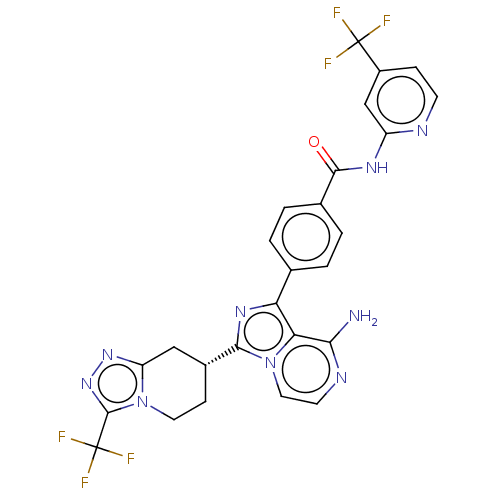
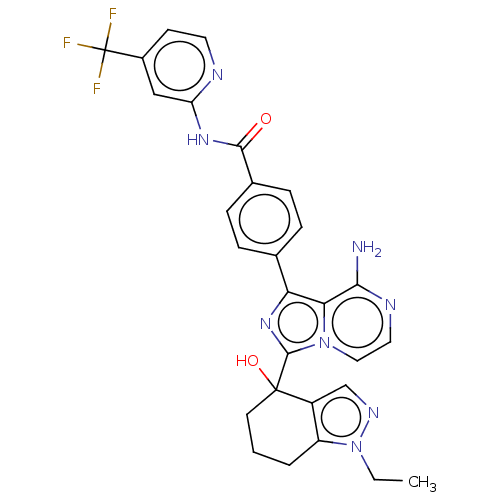
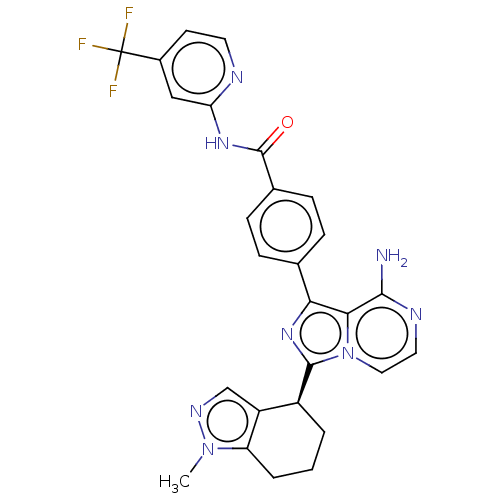
Affinity DataIC50: 0.330nMAssay Description:Assay Method B: BTK enzymatic activity was determined with the LANCE (Lanthanide Chelate Excite) TR-FRET (Time-resolved fluorescence resonance energy...More data for this Ligand-Target Pair
Affinity DataIC50: 0.450nMAssay Description:Assay Method B: BTK enzymatic activity was determined with the LANCE (Lanthanide Chelate Excite) TR-FRET (Time-resolved fluorescence resonance energy...More data for this Ligand-Target Pair
Affinity DataIC50: 0.520nMAssay Description:Assay Method B: BTK enzymatic activity was determined with the LANCE (Lanthanide Chelate Excite) TR-FRET (Time-resolved fluorescence resonance energy...More data for this Ligand-Target Pair
Affinity DataIC50: 0.810nMAssay Description:Assay Method B: BTK enzymatic activity was determined with the LANCE (Lanthanide Chelate Excite) TR-FRET (Time-resolved fluorescence resonance energy...More data for this Ligand-Target Pair
Affinity DataIC50: 1.10nMAssay Description:Assay Method B: BTK enzymatic activity was determined with the LANCE (Lanthanide Chelate Excite) TR-FRET (Time-resolved fluorescence resonance energy...More data for this Ligand-Target Pair
Affinity DataIC50: 1.30nMAssay Description:Assay Method B: BTK enzymatic activity was determined with the LANCE (Lanthanide Chelate Excite) TR-FRET (Time-resolved fluorescence resonance energy...More data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:Assay Method B: BTK enzymatic activity was determined with the LANCE (Lanthanide Chelate Excite) TR-FRET (Time-resolved fluorescence resonance energy...More data for this Ligand-Target Pair
Affinity DataIC50: 2.10nMAssay Description:Assay Method B: BTK enzymatic activity was determined with the LANCE (Lanthanide Chelate Excite) TR-FRET (Time-resolved fluorescence resonance energy...More data for this Ligand-Target Pair
Affinity DataIC50: 2.70nMAssay Description:Assay Method B: BTK enzymatic activity was determined with the LANCE (Lanthanide Chelate Excite) TR-FRET (Time-resolved fluorescence resonance energy...More data for this Ligand-Target Pair
Affinity DataIC50: 3.20nMAssay Description:Assay Method B: BTK enzymatic activity was determined with the LANCE (Lanthanide Chelate Excite) TR-FRET (Time-resolved fluorescence resonance energy...More data for this Ligand-Target Pair
Affinity DataIC50: 3.20nMAssay Description:Assay Method B: BTK enzymatic activity was determined with the LANCE (Lanthanide Chelate Excite) TR-FRET (Time-resolved fluorescence resonance energy...More data for this Ligand-Target Pair
Affinity DataIC50: 5.90nMAssay Description:Assay Method B: BTK enzymatic activity was determined with the LANCE (Lanthanide Chelate Excite) TR-FRET (Time-resolved fluorescence resonance energy...More data for this Ligand-Target Pair
Affinity DataIC50: 5.90nMAssay Description:Assay Method B: BTK enzymatic activity was determined with the LANCE (Lanthanide Chelate Excite) TR-FRET (Time-resolved fluorescence resonance energy...More data for this Ligand-Target Pair
Affinity DataIC50: 7.20nMpH: 7.2Assay Description:Assay Method A: BTK enzymatic activity was determined in an IMAP-FP assay (Immobilized Metal ion Affinity-based fluorescence Polarization; Molecular ...More data for this Ligand-Target Pair
Affinity DataIC50: 11nMAssay Description:Assay Method B: BTK enzymatic activity was determined with the LANCE (Lanthanide Chelate Excite) TR-FRET (Time-resolved fluorescence resonance energy...More data for this Ligand-Target Pair
Affinity DataIC50: 12.0nMpH: 7.2Assay Description:Assay Method A: BTK enzymatic activity was determined in an IMAP-FP assay (Immobilized Metal ion Affinity-based fluorescence Polarization; Molecular ...More data for this Ligand-Target Pair
Affinity DataIC50: 13nMAssay Description:Assay Method B: BTK enzymatic activity was determined with the LANCE (Lanthanide Chelate Excite) TR-FRET (Time-resolved fluorescence resonance energy...More data for this Ligand-Target Pair
Affinity DataIC50: 43nMAssay Description:Assay Method B: BTK enzymatic activity was determined with the LANCE (Lanthanide Chelate Excite) TR-FRET (Time-resolved fluorescence resonance energy...More data for this Ligand-Target Pair
Affinity DataIC50: 54nMAssay Description:Assay Method B: BTK enzymatic activity was determined with the LANCE (Lanthanide Chelate Excite) TR-FRET (Time-resolved fluorescence resonance energy...More data for this Ligand-Target Pair
Affinity DataIC50: 56nMAssay Description:Assay Method B: BTK enzymatic activity was determined with the LANCE (Lanthanide Chelate Excite) TR-FRET (Time-resolved fluorescence resonance energy...More data for this Ligand-Target Pair
Affinity DataIC50: 60nMAssay Description:Assay Method B: BTK enzymatic activity was determined with the LANCE (Lanthanide Chelate Excite) TR-FRET (Time-resolved fluorescence resonance energy...More data for this Ligand-Target Pair
Affinity DataIC50: 67nMAssay Description:Assay Method B: BTK enzymatic activity was determined with the LANCE (Lanthanide Chelate Excite) TR-FRET (Time-resolved fluorescence resonance energy...More data for this Ligand-Target Pair
Affinity DataIC50: 83nMAssay Description:Assay Method B: BTK enzymatic activity was determined with the LANCE (Lanthanide Chelate Excite) TR-FRET (Time-resolved fluorescence resonance energy...More data for this Ligand-Target Pair
Affinity DataIC50: 740nMAssay Description:Assay Method B: BTK enzymatic activity was determined with the LANCE (Lanthanide Chelate Excite) TR-FRET (Time-resolved fluorescence resonance energy...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+3nMAssay Description:Assay Method B: BTK enzymatic activity was determined with the LANCE (Lanthanide Chelate Excite) TR-FRET (Time-resolved fluorescence resonance energy...More data for this Ligand-Target Pair