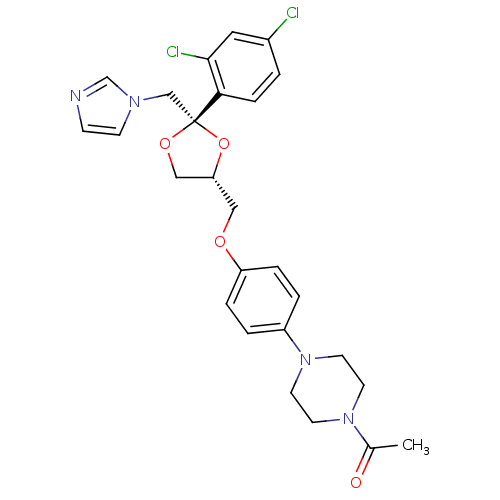
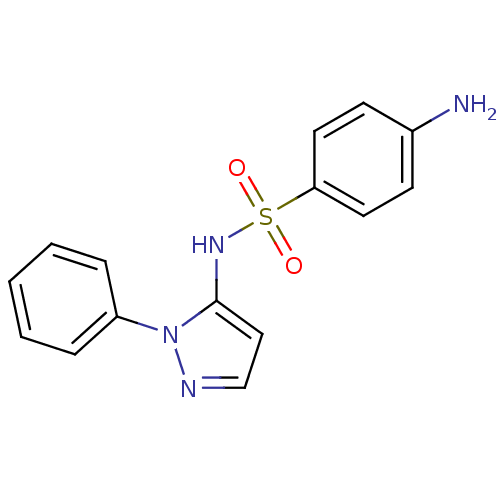
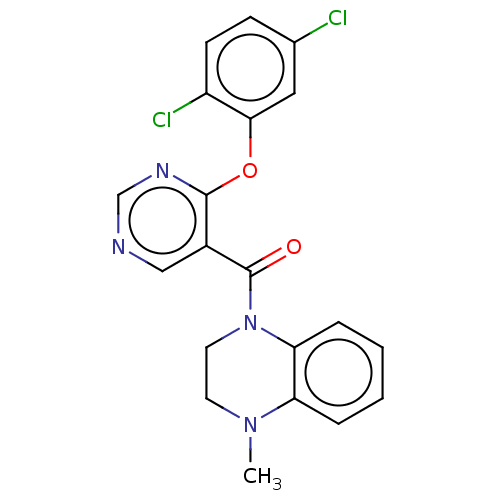
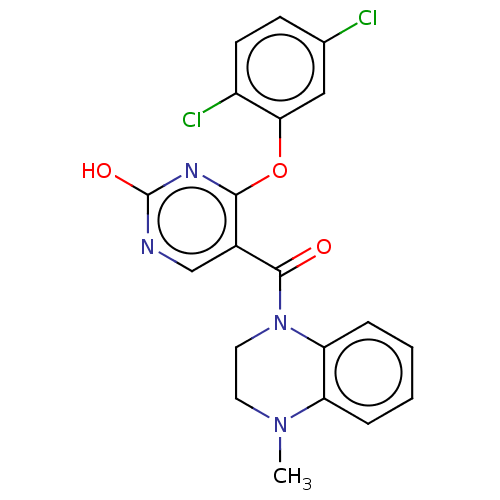
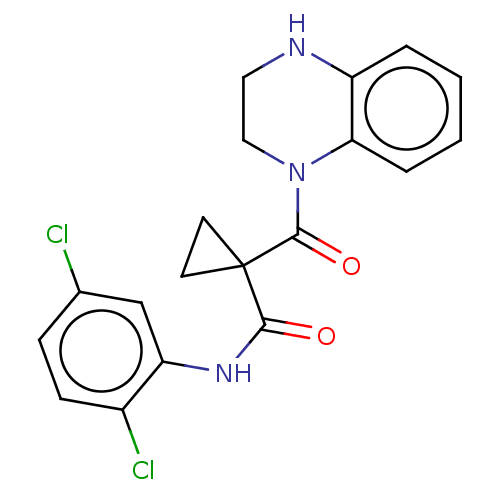
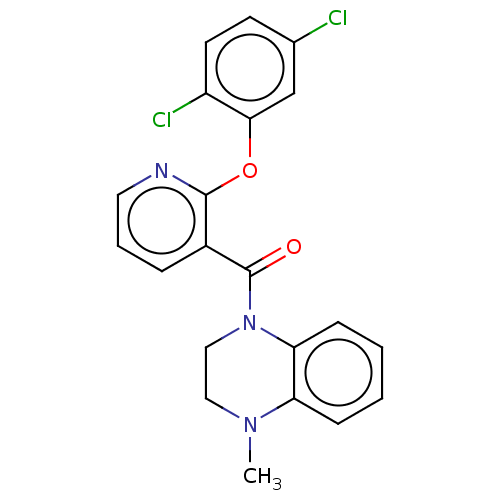
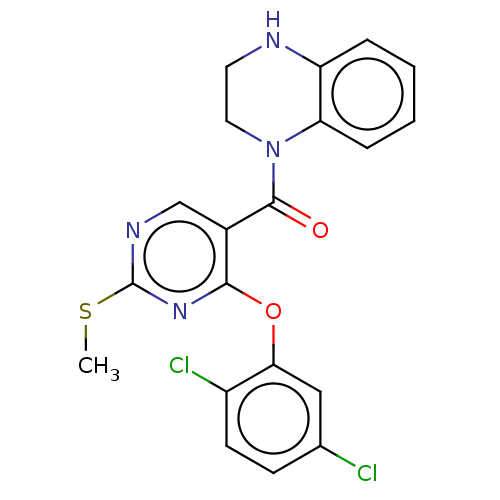
Affinity DataIC50: <6nMAssay Description:Inhibition of CYP2D6 in human liver microsomeMore data for this Ligand-Target Pair
Affinity DataIC50: <62nMAssay Description:Inhibition of CYP3A4 in human liver microsomeMore data for this Ligand-Target Pair
Affinity DataIC50: 180nMAssay Description:Inhibition of CYP2C9 in human liver microsomeMore data for this Ligand-Target Pair
Affinity DataIC50: 250nMAssay Description:Inhibition of CYP3A4 in human liver microsomeMore data for this Ligand-Target Pair
Affinity DataIC50: 260nMAssay Description:Inhibition of CYP3A4 in human liver microsomeMore data for this Ligand-Target Pair
Affinity DataIC50: 270nMAssay Description:Inhibition of CYP3A4 in human liver microsomeMore data for this Ligand-Target Pair
Affinity DataIC50: 780nMAssay Description:Inhibition of CYP2C19 in human liver microsomeMore data for this Ligand-Target Pair
Affinity DataIC50: 1.29E+3nMAssay Description:Inhibition of CYP2C9 in human liver microsomeMore data for this Ligand-Target Pair
Affinity DataIC50: 1.30E+3nMAssay Description:Inhibition of CYP2C9 in human liver microsomeMore data for this Ligand-Target Pair
Affinity DataIC50: 1.54E+3nMAssay Description:Inhibition of CYP2C9 in human liver microsomeMore data for this Ligand-Target Pair
Affinity DataIC50: 1.61E+3nMAssay Description:Inhibition of CYP2C19 in human liver microsomeMore data for this Ligand-Target Pair
Affinity DataIC50: 1.90E+3nMAssay Description:Inhibition of CYP2C19 in human liver microsomeMore data for this Ligand-Target Pair
Affinity DataIC50: 3.62E+3nMAssay Description:Inhibition of CYP1A2 in human liver microsomeMore data for this Ligand-Target Pair
Affinity DataIC50: 9.62E+3nMAssay Description:Inhibition of CYP2D6 in human liver microsomeMore data for this Ligand-Target Pair
Affinity DataIC50: 1.67E+4nMAssay Description:Inhibition of CYP2C19 in human liver microsomeMore data for this Ligand-Target Pair
Affinity DataIC50: 2.00E+4nMAssay Description:Inhibition of CYP2D6 in human liver microsomeMore data for this Ligand-Target Pair
Affinity DataIC50: 2.00E+4nMAssay Description:Inhibition of CYP2D6 in human liver microsomeMore data for this Ligand-Target Pair
Affinity DataIC50: 2.00E+4nMAssay Description:Inhibition of CYP1A2 in human liver microsomeMore data for this Ligand-Target Pair
Affinity DataIC50: 2.00E+4nMAssay Description:Inhibition of CYP1A2 in human liver microsomeMore data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+4nMAssay Description:Inhibition of CYP1A2 in human liver microsomeMore data for this Ligand-Target Pair
TargetG-protein coupled bile acid receptor 1(Homo sapiens (Human))
Chung-Ang University
Curated by ChEMBL
Chung-Ang University
Curated by ChEMBL
Affinity DataEC50: 178nMAssay Description:Agonist activity at human TGR5 expressed in HEK293 cells assessed as cAMP level after 48 hrs by fluorescent assayMore data for this Ligand-Target Pair
TargetG-protein coupled bile acid receptor 1(Homo sapiens (Human))
Chung-Ang University
Curated by ChEMBL
Chung-Ang University
Curated by ChEMBL
Affinity DataEC50: 293nMAssay Description:Agonist activity at human TGR5 expressed in HEK293 cells assessed as cAMP level after 48 hrs by fluorescent assayMore data for this Ligand-Target Pair
TargetG-protein coupled bile acid receptor 1(Homo sapiens (Human))
Chung-Ang University
Curated by ChEMBL
Chung-Ang University
Curated by ChEMBL
Affinity DataEC50: 6.10nMAssay Description:Agonist activity at human TGR5 expressed in HEK293 cells assessed as cAMP level after 48 hrs by fluorescent assayMore data for this Ligand-Target Pair
TargetG-protein coupled bile acid receptor 1(Homo sapiens (Human))
Chung-Ang University
Curated by ChEMBL
Chung-Ang University
Curated by ChEMBL
Affinity DataEC50: 3.5nMAssay Description:Agonist activity at human TGR5 expressed in HEK293 cells assessed as cAMP level after 48 hrs by fluorescent assayMore data for this Ligand-Target Pair
TargetG-protein coupled bile acid receptor 1(Homo sapiens (Human))
Chung-Ang University
Curated by ChEMBL
Chung-Ang University
Curated by ChEMBL
Affinity DataEC50: 3.90nMAssay Description:Agonist activity at human TGR5 expressed in HEK293 cells assessed as cAMP level after 48 hrs by fluorescent assayMore data for this Ligand-Target Pair
TargetG-protein coupled bile acid receptor 1(Homo sapiens (Human))
Chung-Ang University
Curated by ChEMBL
Chung-Ang University
Curated by ChEMBL
Affinity DataEC50: >1.00E+3nMAssay Description:Agonist activity at human TGR5 expressed in HEK293 cells assessed as cAMP level after 48 hrs by fluorescent assayMore data for this Ligand-Target Pair
TargetG-protein coupled bile acid receptor 1(Homo sapiens (Human))
Chung-Ang University
Curated by ChEMBL
Chung-Ang University
Curated by ChEMBL
Affinity DataEC50: 20nMAssay Description:Agonist activity at human TGR5 expressed in HEK293 cells assessed as cAMP level after 48 hrs by fluorescent assayMore data for this Ligand-Target Pair
TargetG-protein coupled bile acid receptor 1(Homo sapiens (Human))
Chung-Ang University
Curated by ChEMBL
Chung-Ang University
Curated by ChEMBL
Affinity DataEC50: 12nMAssay Description:Agonist activity at human TGR5 expressed in HEK293 cells assessed as cAMP level after 48 hrs by fluorescent assayMore data for this Ligand-Target Pair
TargetG-protein coupled bile acid receptor 1(Homo sapiens (Human))
Chung-Ang University
Curated by ChEMBL
Chung-Ang University
Curated by ChEMBL
Affinity DataEC50: 41nMAssay Description:Agonist activity at human TGR5 expressed in HEK293 cells assessed as cAMP level after 48 hrs by fluorescent assayMore data for this Ligand-Target Pair
TargetG-protein coupled bile acid receptor 1(Homo sapiens (Human))
Chung-Ang University
Curated by ChEMBL
Chung-Ang University
Curated by ChEMBL
Affinity DataEC50: 48nMAssay Description:Agonist activity at human TGR5 expressed in HEK293 cells assessed as cAMP level after 48 hrs by fluorescent assayMore data for this Ligand-Target Pair
TargetG-protein coupled bile acid receptor 1(Homo sapiens (Human))
Chung-Ang University
Curated by ChEMBL
Chung-Ang University
Curated by ChEMBL
Affinity DataEC50: 122nMAssay Description:Agonist activity at human TGR5 expressed in HEK293 cells assessed as cAMP level after 48 hrs by fluorescent assayMore data for this Ligand-Target Pair
TargetG-protein coupled bile acid receptor 1(Homo sapiens (Human))
Chung-Ang University
Curated by ChEMBL
Chung-Ang University
Curated by ChEMBL
Affinity DataEC50: 5.10nMAssay Description:Agonist activity at human TGR5 expressed in HEK293 cells assessed as cAMP level after 48 hrs by fluorescent assayMore data for this Ligand-Target Pair
TargetG-protein coupled bile acid receptor 1(Homo sapiens (Human))
Chung-Ang University
Curated by ChEMBL
Chung-Ang University
Curated by ChEMBL
Affinity DataEC50: >1.00E+3nMAssay Description:Agonist activity at human TGR5 expressed in HEK293 cells assessed as cAMP level after 48 hrs by fluorescent assayMore data for this Ligand-Target Pair
TargetG-protein coupled bile acid receptor 1(Homo sapiens (Human))
Chung-Ang University
Curated by ChEMBL
Chung-Ang University
Curated by ChEMBL
Affinity DataEC50: >1.00E+3nMAssay Description:Agonist activity at human TGR5 expressed in HEK293 cells assessed as cAMP level after 48 hrs by fluorescent assayMore data for this Ligand-Target Pair


 3D Structure (crystal)
3D Structure (crystal)