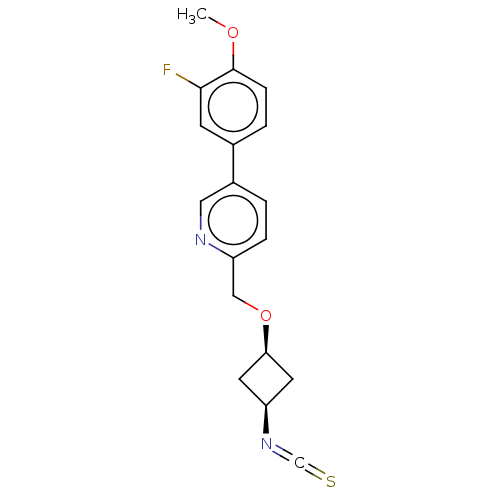
BDBM447507 US10689357, Example 43
SMILES COc1ccc(cc1F)-c1ccc(CO[C@H]2C[C@H](C2)N=C=S)nc1
InChI Key InChIKey=GAVRQMDMBSIEDN-IYBDPMFKSA-N
Data 7 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 7 hits for monomerid = 447507
Found 7 hits for monomerid = 447507
TargetN-acylethanolamine-hydrolyzing acid amidase(Homo sapiens (Human))
Northeastern University
US Patent
Northeastern University
US Patent
Affinity DataIC50: 55nMAssay Description:In order to have an assay method more conducive to high-throughput screening than those published for measuring the NAE hydrolyzing activity of NAAA,...More data for this Ligand-Target Pair
TargetN-acylethanolamine-hydrolyzing acid amidase(Homo sapiens (Human))
Northeastern University
US Patent
Northeastern University
US Patent
Affinity DataIC50: 20nMAssay Description:Inhibition of human NAAA using N-(4-methyl coumarin)-palmitamide as fluorogenic substrate preincubated for 90 mins followed by substrate addition by ...More data for this Ligand-Target Pair
Affinity DataIC50: 121nMAssay Description:Inhibition of recombinant human Cathepsin K using Z-Phe-Arg-AMC as fluorogenic substrate preincubated for 120 mins followed by substrate addition by ...More data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
Northeastern University
Curated by ChEMBL
Northeastern University
Curated by ChEMBL
Affinity DataIC50: 1.90E+4nMAssay Description:Inhibition of human ERGMore data for this Ligand-Target Pair
Affinity DataIC50: 3.30E+3nMAssay Description:Inhibition of CYP2C19 (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 2.60E+3nMAssay Description:Inhibition of CYP3A4 (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 1.80E+3nMAssay Description:Inhibition of CYP1A2 (unknown origin)More data for this Ligand-Target Pair