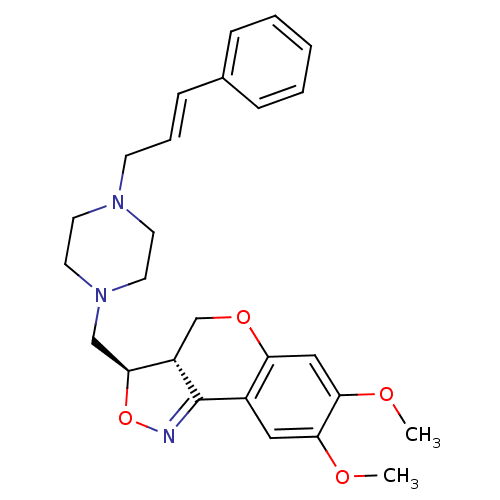
BDBM50131359 (3R,3aS)-7,8-Dimethoxy-3-[4-((E)-3-phenyl-allyl)-piperazin-1-ylmethyl]-3a,4-dihydro-3H-chromeno[4,3-c]isoxazole::(3R,3aS)-7,8-Dimethoxy-3-[4-(3-phenyl-allyl)-piperazin-1-ylmethyl]-3a,4-dihydro-3H-chromeno[4,3-c]isoxazole::CHEMBL328114
SMILES COc1cc2OC[C@H]3[C@H](CN4CCN(C\C=C\c5ccccc5)CC4)ON=C3c2cc1OC
InChI Key InChIKey=WZOMJNATDSYQHS-ZPWQMVEJSA-N
Data 29 KI
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 8 hits for monomerid = 50131359
Found 8 hits for monomerid = 50131359
TargetAlpha-2C adrenergic receptor(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.5nMAssay Description:In vitro binding affinity towards human adrenergic alpha-2C adrenergic receptor using [3H]-rauwolscineMore data for this Ligand-Target Pair
TargetAlpha-2C adrenergic receptor(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.5nMAssay Description:In vitro binding affinity towards human adrenergic alpha-2C adrenergic receptor using [3H]-rauwolscineMore data for this Ligand-Target Pair
TargetSodium-dependent serotonin transporter(Rattus norvegicus (rat))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 1.20nMAssay Description:In vitro binding affinity towards rat serotonin transporterMore data for this Ligand-Target Pair
TargetSodium-dependent serotonin transporter(Rattus norvegicus (rat))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 1.20nMAssay Description:In vitro binding affinity towards human alpha-2B adrenergic receptor using [3H]-rauwolscineMore data for this Ligand-Target Pair
TargetAlpha-2A adrenergic receptor(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 2.60nMAssay Description:In vitro binding affinity towards human alpha-2A adrenergic receptor using [3H]-rauwolscineMore data for this Ligand-Target Pair
TargetAlpha-2A adrenergic receptor(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 2.60nMAssay Description:In vitro binding affinity towards human alpha-2B adrenergic receptor using [3H]-rauwolscineMore data for this Ligand-Target Pair
TargetAlpha-2B adrenergic receptor(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 14nMAssay Description:In vitro binding affinity towards human alpha-2B adrenergic receptor using [3H]-rauwolscineMore data for this Ligand-Target Pair
TargetAlpha-2B adrenergic receptor(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 14nMAssay Description:In vitro binding affinity towards human alpha-2B adrenergic receptor using [3H]-rauwolscineMore data for this Ligand-Target Pair