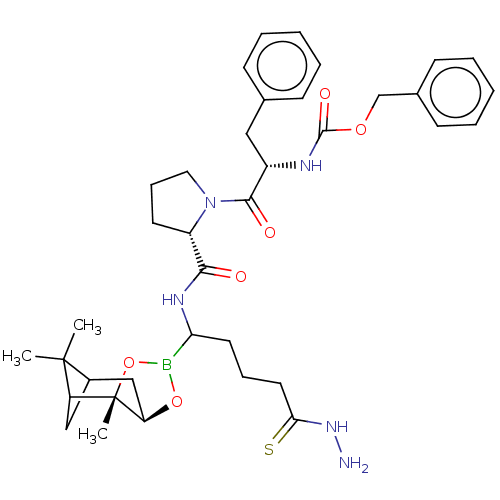
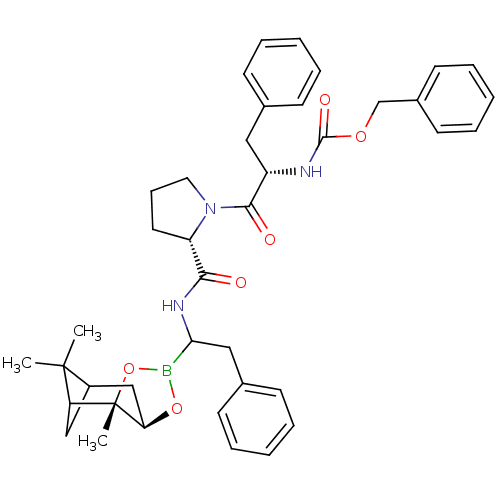
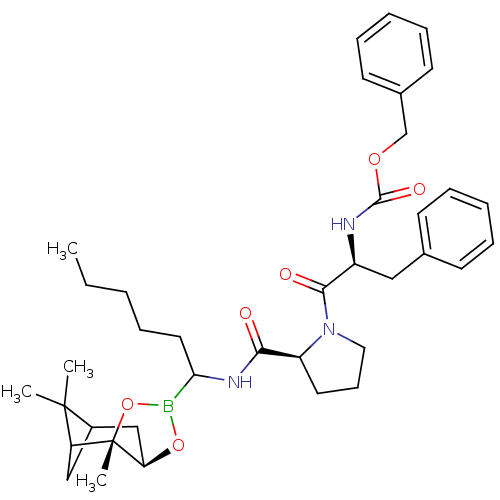
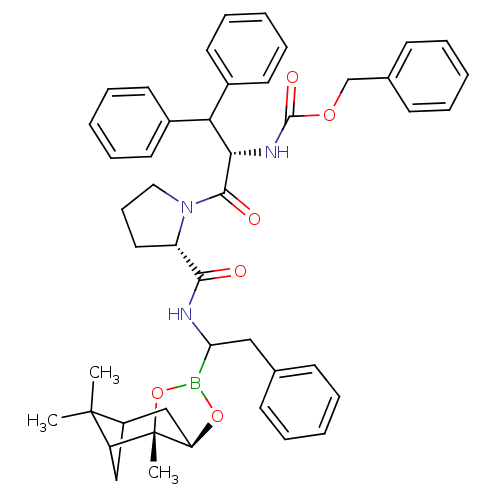
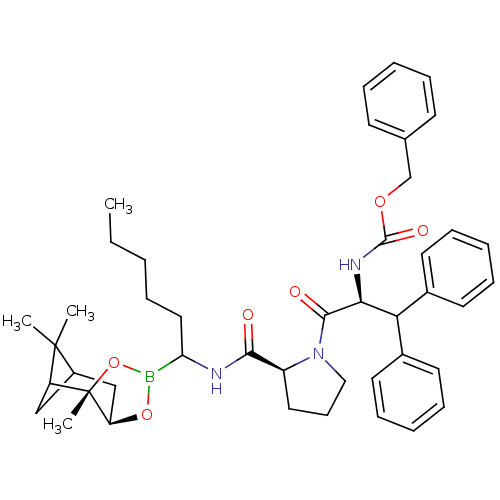
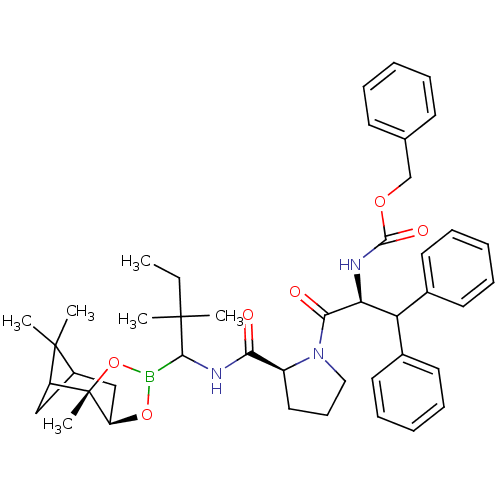
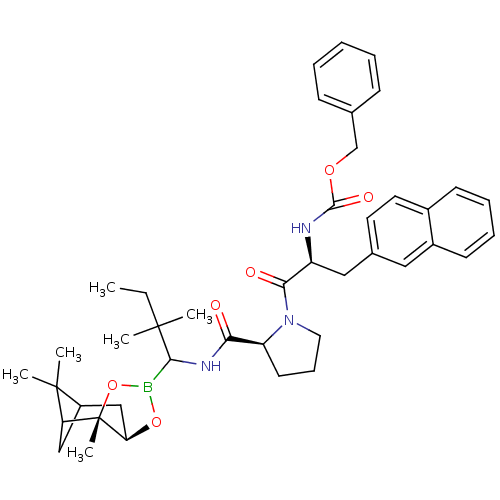
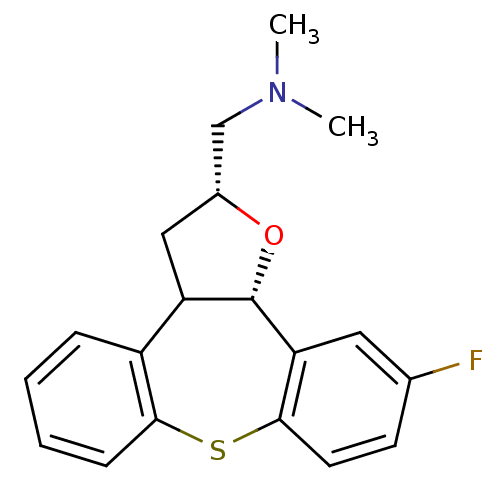
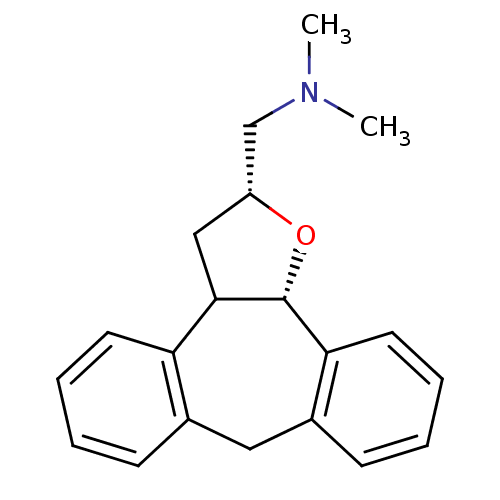
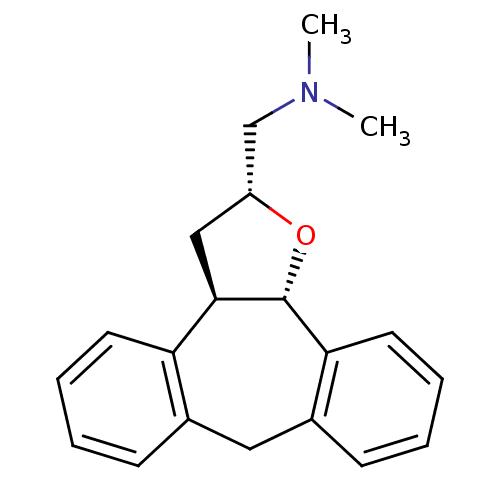
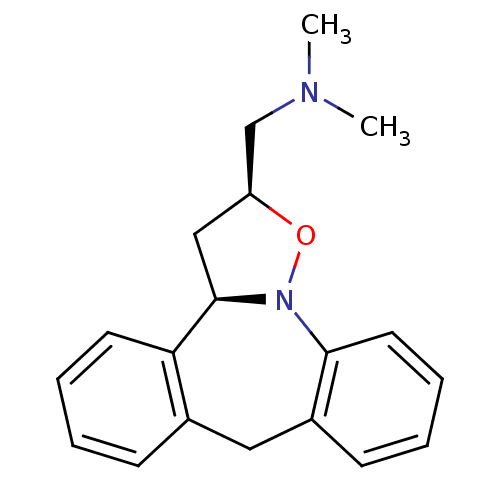
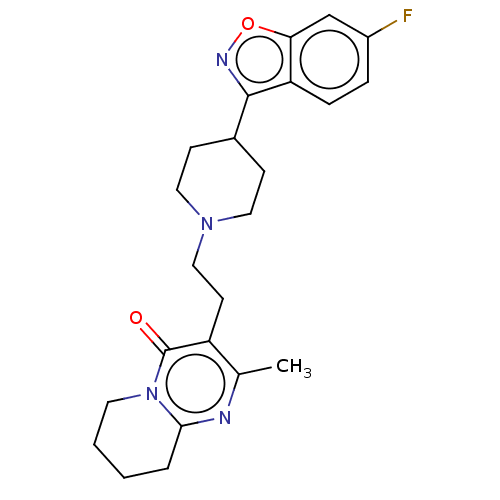
Affinity DataKi: 0.00150nMAssay Description:Binding affinity against alpha thrombinMore data for this Ligand-Target Pair
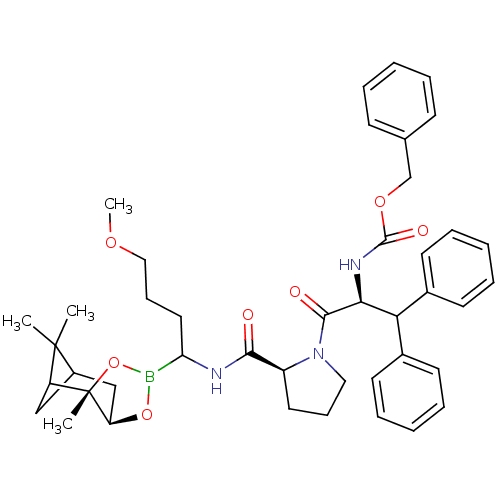
Affinity DataKi: 0.00200nMAssay Description:Binding affinity against alpha thrombinMore data for this Ligand-Target Pair
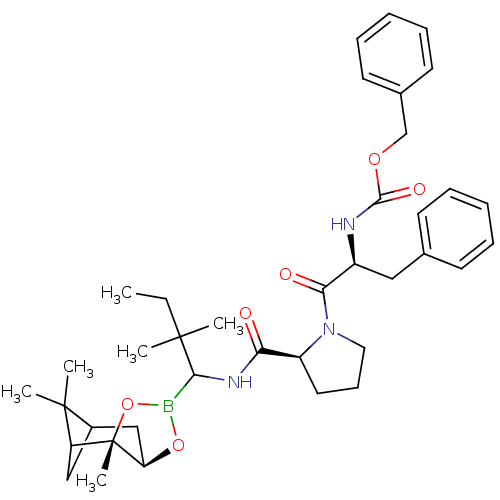
Affinity DataKi: 0.00400nMAssay Description:Binding affinity against gamma thrombinMore data for this Ligand-Target Pair
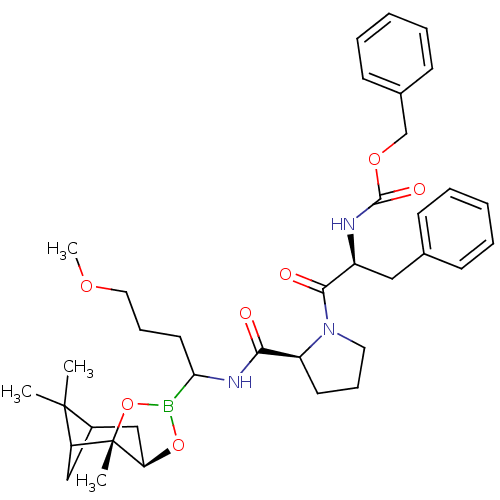
Affinity DataKi: 0.00490nMAssay Description:Binding affinity against kallikreinMore data for this Ligand-Target Pair
Affinity DataKi: 0.00700nMAssay Description:Binding affinity against alpha thrombinMore data for this Ligand-Target Pair
Affinity DataKi: 0.00700nMAssay Description:Binding affinity against alpha thrombinMore data for this Ligand-Target Pair
Affinity DataKi: 0.0110nMAssay Description:Binding affinity against gamma thrombinMore data for this Ligand-Target Pair
Affinity DataKi: 0.0110nMAssay Description:Binding affinity against ChymotrypsinMore data for this Ligand-Target Pair
Affinity DataKi: 0.0180nMAssay Description:Binding affinity against alpha thrombinMore data for this Ligand-Target Pair
Affinity DataKi: 0.0190nMAssay Description:Binding affinity against alpha thrombinMore data for this Ligand-Target Pair
Affinity DataKi: 0.0190nMAssay Description:Binding affinity against gamma thrombinMore data for this Ligand-Target Pair
Affinity DataKi: 0.0210nMAssay Description:Binding affinity against ChymotrypsinMore data for this Ligand-Target Pair
Affinity DataKi: 0.0240nMAssay Description:Binding affinity against gamma thrombinMore data for this Ligand-Target Pair
Affinity DataKi: 0.0270nMAssay Description:Binding affinity against gamma thrombinMore data for this Ligand-Target Pair
Affinity DataKi: 0.0280nMAssay Description:Binding affinity against alpha thrombinMore data for this Ligand-Target Pair
Affinity DataKi: 0.0280nMAssay Description:Binding affinity against gamma thrombinMore data for this Ligand-Target Pair
Affinity DataKi: 0.0340nMAssay Description:Binding affinity against gamma thrombinMore data for this Ligand-Target Pair
Affinity DataKi: 0.0420nMAssay Description:Binding affinity against alpha thrombinMore data for this Ligand-Target Pair
Affinity DataKi: 0.0540nMAssay Description:Binding affinity against TrypsinMore data for this Ligand-Target Pair
Affinity DataKi: 0.0590nMAssay Description:Binding affinity against gamma thrombinMore data for this Ligand-Target Pair
Affinity DataKi: 0.0820nMAssay Description:Binding affinity against alpha thrombinMore data for this Ligand-Target Pair
Affinity DataKi: 0.115nMAssay Description:Binding affinity against ChymotrypsinMore data for this Ligand-Target Pair
Affinity DataKi: 0.130nMAssay Description:Binding affinity against alpha thrombinMore data for this Ligand-Target Pair
Affinity DataKi: 0.140nMAssay Description:Binding affinity against gamma thrombinMore data for this Ligand-Target Pair
Affinity DataKi: 0.150nMAssay Description:Binding affinity against TrypsinMore data for this Ligand-Target Pair
Affinity DataKi: 0.152nMAssay Description:Binding affinity against ChymotrypsinMore data for this Ligand-Target Pair
Affinity DataKi: 0.174nMAssay Description:Binding affinity against cathepsin GMore data for this Ligand-Target Pair
Affinity DataKi: 0.187nMAssay Description:Binding affinity against cathepsin GMore data for this Ligand-Target Pair
Target5-hydroxytryptamine receptor 2C(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.210nMAssay Description:Inhibition of [3H]-mesulergine binding to human 5-hydroxytryptamine 2C receptorMore data for this Ligand-Target Pair
Affinity DataKi: 0.270nMAssay Description:Binding affinity against TrypsinMore data for this Ligand-Target Pair
Affinity DataKi: 0.320nMAssay Description:Binding affinity against TrypsinMore data for this Ligand-Target Pair
TargetChymotrypsin-like elastase family member 2A(Sus scrofa)
Thrombosis Research Institute
Curated by ChEMBL
Thrombosis Research Institute
Curated by ChEMBL
Affinity DataKi: 0.365nMAssay Description:Binding affinity of the compound towards porcine pancreatic elastaseMore data for this Ligand-Target Pair
TargetHistamine H1 receptor(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.430nMAssay Description:Inhibition of [3H]-pyrilamine binding to human Histamine H1 receptor More data for this Ligand-Target Pair
Affinity DataKi: 0.440nMAssay Description:Binding affinity against TrypsinMore data for this Ligand-Target Pair
Affinity DataKi: 0.450nMAssay Description:Binding affinity against TrypsinMore data for this Ligand-Target Pair
TargetHistamine H1 receptor(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.520nMAssay Description:Inhibition of [3H]-pyrilamine binding to human Histamine H1 receptor More data for this Ligand-Target Pair
TargetHistamine H1 receptor(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.520nMAssay Description:Inhibition of [3H]-pyrilamine binding to human Histamine H1 receptor More data for this Ligand-Target Pair
TargetCarbonic anhydrase 12(Homo sapiens (Human))
Temple University School Of Pharmacy
Curated by ChEMBL
Temple University School Of Pharmacy
Curated by ChEMBL
Affinity DataKi: 0.520nMAssay Description:Inhibition of human recombinant carbonic anhydrase 12 preincubated for 15 mins prior to testing by stopped-flow CO2 hydration assayMore data for this Ligand-Target Pair
TargetHistamine H1 receptor(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.540nMAssay Description:Ability to displace [3H]-pyrilamine from human cloned histamine H1 receptor expressed in CHO cellsMore data for this Ligand-Target Pair
Affinity DataKi: 0.560nMAssay Description:Binding affinity against TrypsinMore data for this Ligand-Target Pair
Target5-hydroxytryptamine receptor 2A(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.560nMAssay Description:Inhibition of [125I]-R91150 binding to human 5-hydroxytryptamine 2A receptorMore data for this Ligand-Target Pair
Target5-hydroxytryptamine receptor 2A(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.560nMAssay Description:Inhibition of [125I]-R91150 binding to human 5-hydroxytryptamine 2A receptorMore data for this Ligand-Target Pair
Affinity DataKi: 0.580nMAssay Description:Binding affinity against Coagulation factor XMore data for this Ligand-Target Pair
TargetHistamine H1 receptor(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.630nMAssay Description:Ability to displace [3H]-pyrilamine from human cloned histamine H1 receptor expressed in CHO cellsMore data for this Ligand-Target Pair
TargetHistamine H1 receptor(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.640nMAssay Description:Ability to displace [3H]-pyrilamine from human cloned histamine H1 receptor expressed in CHO cellsMore data for this Ligand-Target Pair
Target5-hydroxytryptamine receptor 2C(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.650nMAssay Description:Inhibition of [3H]-mesulergine binding to human 5-hydroxytryptamine 2C receptorMore data for this Ligand-Target Pair
Affinity DataKi: 0.670nMAssay Description:Binding affinity against TrypsinMore data for this Ligand-Target Pair
TargetHistamine H1 receptor(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.700nMAssay Description:Ability to displace [3H]-pyrilamine from human cloned histamine H1 receptor expressed in CHO cellsMore data for this Ligand-Target Pair
TargetDelta-type opioid receptor(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.740nMAssay Description:Displacement of [3H]DPDPE from cloned human delta opioid receptorMore data for this Ligand-Target Pair
Target5-hydroxytryptamine receptor 2A(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.810nMAssay Description:Inhibition of [125I]-R91150 binding to human 5-hydroxytryptamine 2A receptorMore data for this Ligand-Target Pair