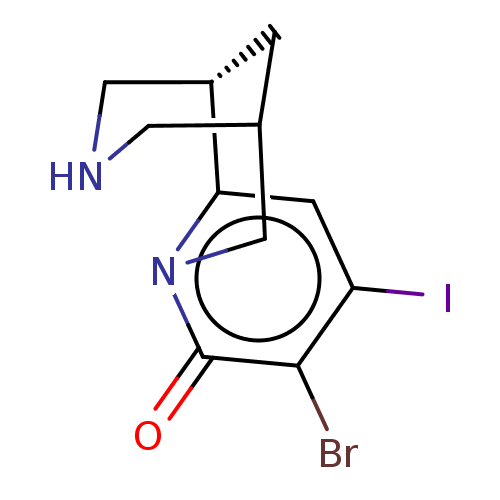
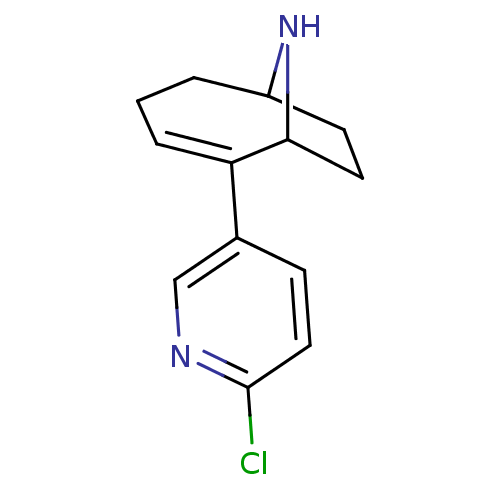
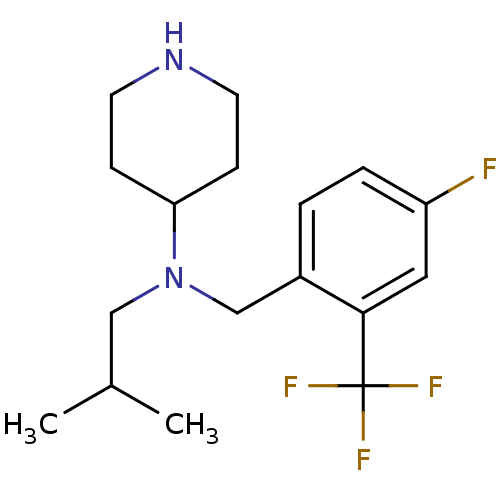
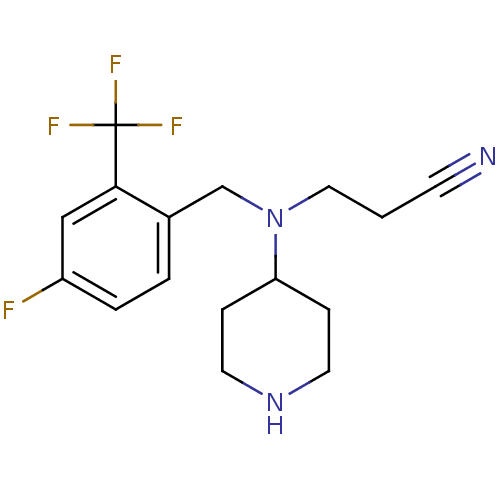
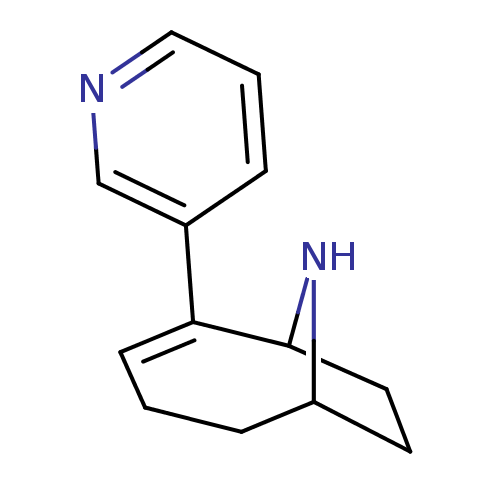
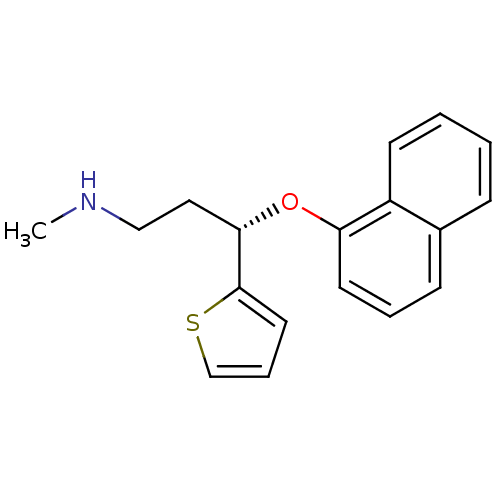
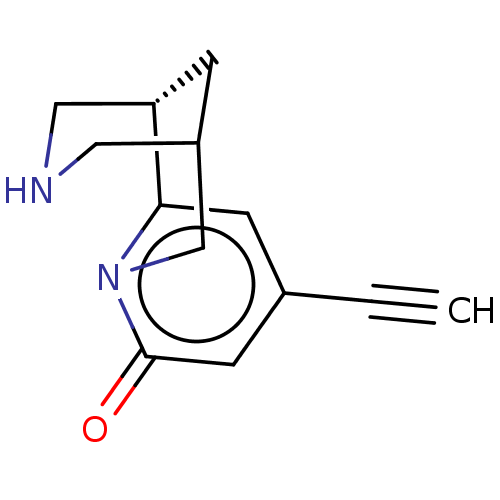
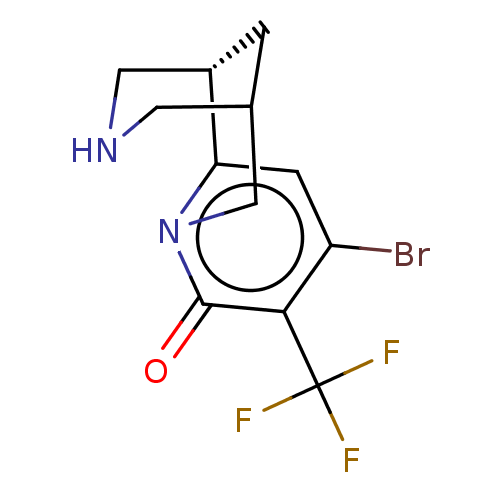
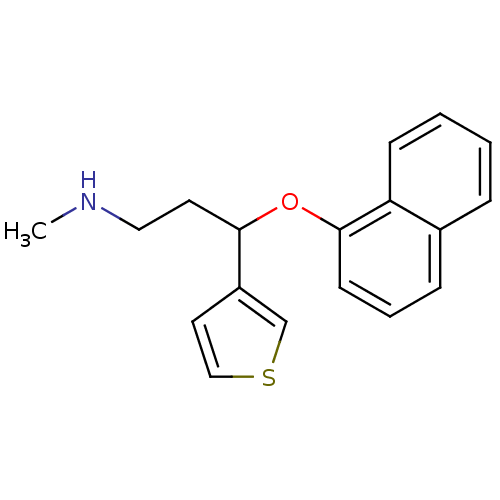
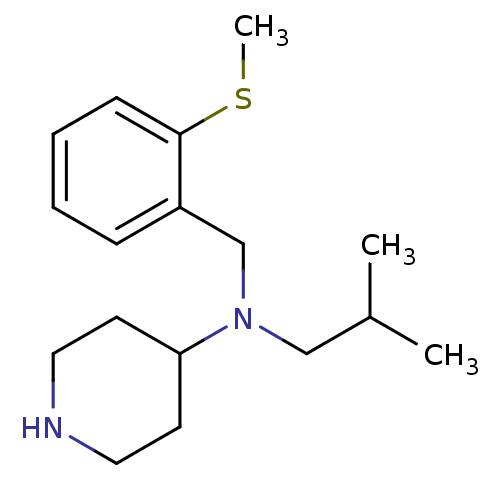
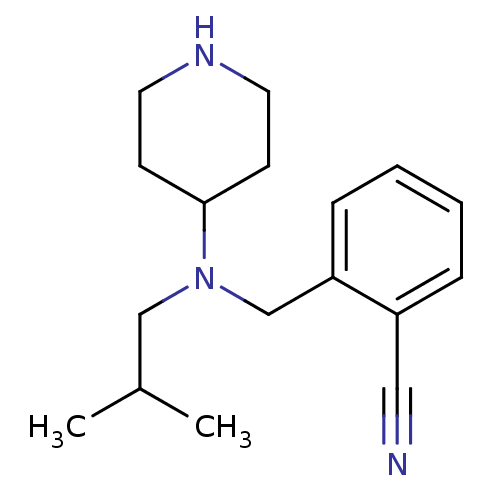
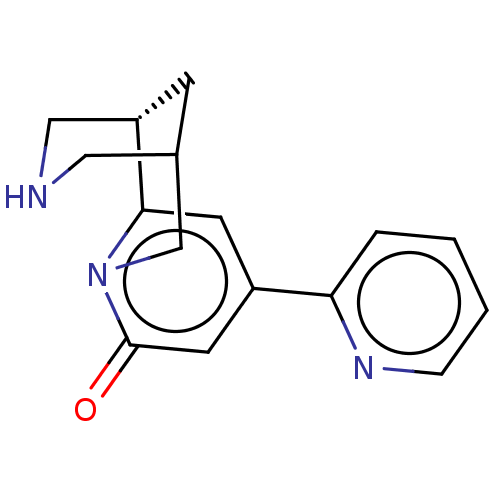
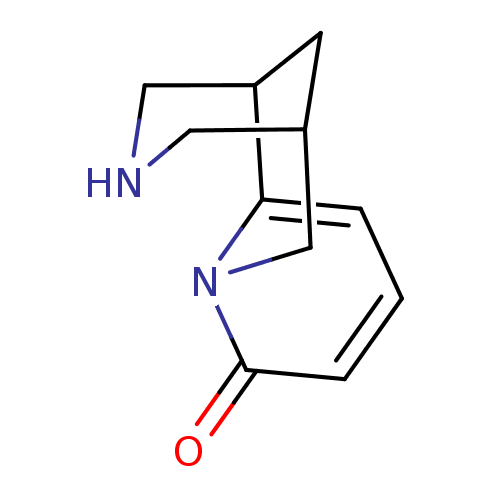
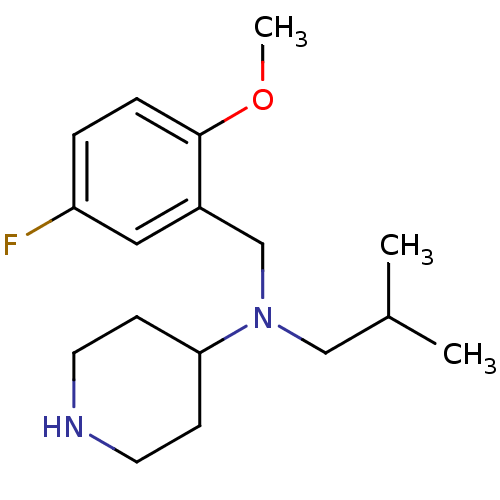
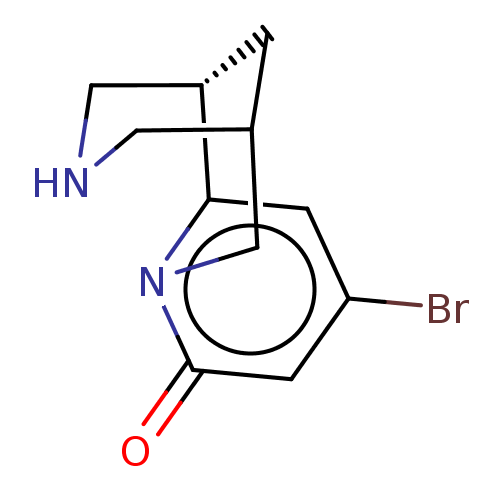
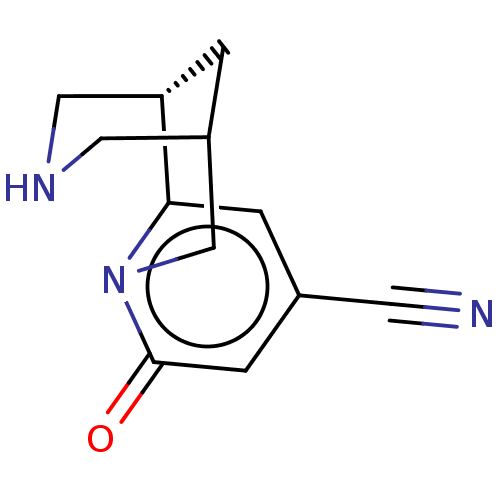
Affinity DataKi: 0.0700nMAssay Description:Displacement of [3H]citalopram from SERTMore data for this Ligand-Target Pair
TargetNeuronal acetylcholine receptor subunit alpha-4/beta-2(Homo sapiens (Human))
University of Bristol
US Patent
University of Bristol
US Patent
Affinity DataKi: 0.238nMAssay Description:The binding of a group of compounds mentioned above were tested for their affinity at different nAChR subtypes, specifically the α4β2, the ...More data for this Ligand-Target Pair
Affinity DataKi: 0.260nMAssay Description:Displacement of [3H]citalopram from SERTMore data for this Ligand-Target Pair
TargetNeuronal acetylcholine receptor subunit alpha-4/beta-2(Rattus norvegicus (Rat))
University Of Bristol
Curated by ChEMBL
University Of Bristol
Curated by ChEMBL
Affinity DataKi: 0.270nMAssay Description:Binding affinity towards alpha3-beta4 subtype of neuronal nicotinic acetylcholine receptor (nAChR) using [3H]-epibatidine as radioligandMore data for this Ligand-Target Pair
Affinity DataKi: 0.280nMAssay Description:Displacement of [3H]citalopram from SERTMore data for this Ligand-Target Pair
Affinity DataKi: 0.300nMAssay Description:Displacement of [3H]citalopram from SERTMore data for this Ligand-Target Pair
Affinity DataKi: 0.390nMAssay Description:Displacement of [3H]citalopram from SERTMore data for this Ligand-Target Pair
Affinity DataKi: 0.400nMAssay Description:Displacement of [3H]citalopram from SERTMore data for this Ligand-Target Pair
Affinity DataKi: 0.400nMAssay Description:Displacement of [3H]citalopram from SERTMore data for this Ligand-Target Pair
Affinity DataKi: 0.400nMAssay Description:Displacement of [3H]citalopram from SERTMore data for this Ligand-Target Pair
TargetNeuronal acetylcholine receptor subunit alpha-4/beta-2(Rattus norvegicus (Rat))
University Of Bristol
Curated by ChEMBL
University Of Bristol
Curated by ChEMBL
Affinity DataKi: 0.430nMAssay Description:Binding affinity towards alpha3-beta4 subtype of neuronal nicotinic acetylcholine receptor (nAChR) using [3H]-epibatidine as radioligandMore data for this Ligand-Target Pair
Affinity DataKi: 0.480nMAssay Description:Displacement of [3H]citalopram from SERTMore data for this Ligand-Target Pair
Affinity DataKi: 0.480nMAssay Description:Displacement of [3H]citalopram from SERTMore data for this Ligand-Target Pair
Affinity DataKi: 0.580nMAssay Description:Displacement of [3H]citalopram from SERTMore data for this Ligand-Target Pair
Affinity DataKi: 0.700nMAssay Description:Binding affinity to serotonin transporter, using [3H]-citalopram as radioligandMore data for this Ligand-Target Pair
Affinity DataKi: 0.700nMAssay Description:Displacement of [3H]citalopram from SERTMore data for this Ligand-Target Pair
Affinity DataKi: 0.800nMAssay Description:Displacement of [3H]citalopram from SERTMore data for this Ligand-Target Pair
Affinity DataKi: 0.800nMAssay Description:Displacement of [3H]citalopram from SERTMore data for this Ligand-Target Pair
Affinity DataKi: 0.800nMAssay Description:Ability to inhibit the reuptake of 5-HT at human serotonin transporterMore data for this Ligand-Target Pair
TargetNeuronal acetylcholine receptor subunit alpha-4/beta-2(Homo sapiens (Human))
University of Bristol
US Patent
University of Bristol
US Patent
Affinity DataKi: 0.860nMAssay Description:The binding of a group of compounds mentioned above were tested for their affinity at different nAChR subtypes, specifically the α4β2, the ...More data for this Ligand-Target Pair
TargetNeuronal acetylcholine receptor subunit alpha-4/beta-2(Homo sapiens (Human))
University of Bristol
US Patent
University of Bristol
US Patent
Affinity DataKi: 0.880nMAssay Description:The binding of a group of compounds mentioned above were tested for their affinity at different nAChR subtypes, specifically the α4β2, the ...More data for this Ligand-Target Pair
Affinity DataKi: 0.950nMAssay Description:Binding affinity to serotonin transporter, using [3H]-citalopram as radioligandMore data for this Ligand-Target Pair
Affinity DataKi: 0.960nMAssay Description:Displacement of [3H]citalopram from SERTMore data for this Ligand-Target Pair
Affinity DataKi: 0.990nMAssay Description:Displacement of [3H]citalopram from SERTMore data for this Ligand-Target Pair
Affinity DataKi: 1.10nMAssay Description:Displacement of [3H]citalopram from SERTMore data for this Ligand-Target Pair
Affinity DataKi: 1.10nMAssay Description:Binding affinity to serotonin transporter, using [3H]-citalopram as radioligandMore data for this Ligand-Target Pair
Affinity DataKi: 1.10nMAssay Description:Displacement of [3H]citalopram from SERTMore data for this Ligand-Target Pair
Affinity DataKi: 1.20nMAssay Description:Displacement of [3H]citalopram from SERTMore data for this Ligand-Target Pair
TargetNeuronal acetylcholine receptor subunit alpha-4/beta-2(Homo sapiens (Human))
University of Bristol
US Patent
University of Bristol
US Patent
Affinity DataKi: 1.24nMAssay Description:The binding of a group of compounds mentioned above were tested for their affinity at different nAChR subtypes, specifically the α4β2, the ...More data for this Ligand-Target Pair
TargetNeuronal acetylcholine receptor subunit alpha-4/beta-2(Homo sapiens (Human))
University of Bristol
US Patent
University of Bristol
US Patent
Affinity DataKi: 1.27nMAssay Description:The binding of a group of compounds mentioned above were tested for their affinity at different nAChR subtypes, specifically the α4β2, the ...More data for this Ligand-Target Pair
Affinity DataKi: 1.30nMAssay Description:Displacement of [3H]citalopram from SERTMore data for this Ligand-Target Pair
Affinity DataKi: 1.40nMAssay Description:Binding affinity to serotonin transporter, using [3H]-citalopram as radioligandMore data for this Ligand-Target Pair
Affinity DataKi: 1.40nMAssay Description:Displacement of [3H]citalopram from SERTMore data for this Ligand-Target Pair
Affinity DataKi: 1.40nMAssay Description:Displacement of [3H]citalopram from SERTMore data for this Ligand-Target Pair
Affinity DataKi: 1.40nMAssay Description:Displacement of [3H]nisoxetine from NETMore data for this Ligand-Target Pair
Affinity DataKi: 1.5nMAssay Description:Displacement of [3H]citalopram from SERTMore data for this Ligand-Target Pair
Affinity DataKi: 1.5nMAssay Description:Displacement of [3H]citalopram from SERTMore data for this Ligand-Target Pair
Affinity DataKi: 1.5nMAssay Description:Displacement of [3H]citalopram from SERTMore data for this Ligand-Target Pair
Affinity DataKi: 1.60nMAssay Description:Displacement of [3H]citalopram from SERTMore data for this Ligand-Target Pair
TargetNeuronal acetylcholine receptor subunit alpha-4/beta-2(Homo sapiens (Human))
University of Bristol
US Patent
University of Bristol
US Patent
Affinity DataKi: 1.77nMAssay Description:The binding of a group of compounds mentioned above were tested for their affinity at different nAChR subtypes, specifically the α4β2, the ...More data for this Ligand-Target Pair
Affinity DataKi: 1.80nMAssay Description:Displacement of [3H]nisoxetine from NETMore data for this Ligand-Target Pair
TargetNeuronal acetylcholine receptor subunit alpha-4/beta-2(Homo sapiens (Human))
University of Bristol
US Patent
University of Bristol
US Patent
Affinity DataKi: 1.84nMAssay Description:The binding of a group of compounds mentioned above were tested for their affinity at different nAChR subtypes, specifically the α4β2, the ...More data for this Ligand-Target Pair
Affinity DataKi: 1.90nMAssay Description:Displacement of [3H]citalopram from SERTMore data for this Ligand-Target Pair
Affinity DataKi: 2nMAssay Description:Binding affinity to serotonin transporter, using [3H]-citalopram as radioligandMore data for this Ligand-Target Pair
Affinity DataKi: 2nMAssay Description:Displacement of [3H]nisoxetine from NETMore data for this Ligand-Target Pair
Affinity DataKi: 2.10nMAssay Description:Binding affinity to serotonin transporter, using [3H]-citalopram as radioligandMore data for this Ligand-Target Pair
Affinity DataKi: 2.10nMAssay Description:Displacement of [3H]citalopram from SERTMore data for this Ligand-Target Pair
Affinity DataKi: 2.20nMAssay Description:Displacement of [3H]nisoxetine from NETMore data for this Ligand-Target Pair
Affinity DataKi: 2.20nMAssay Description:Displacement of [3H]citalopram from SERTMore data for this Ligand-Target Pair