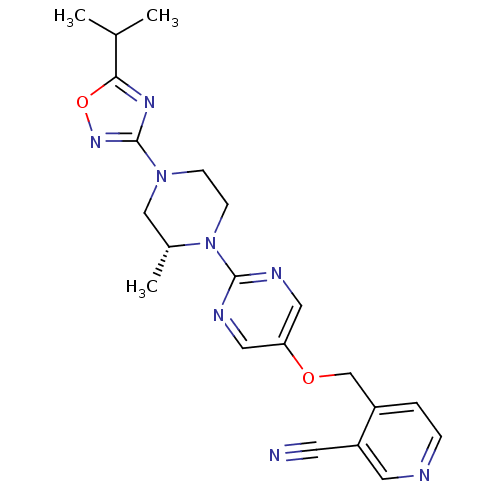
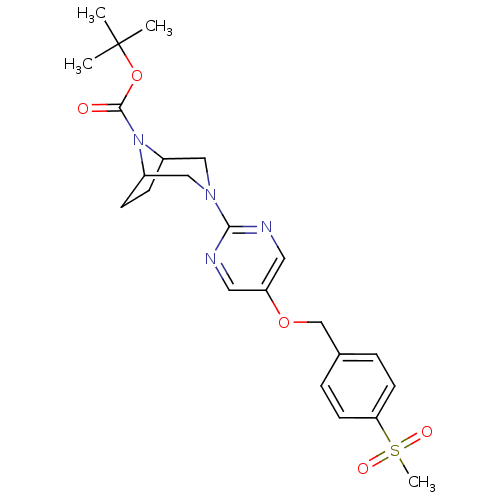
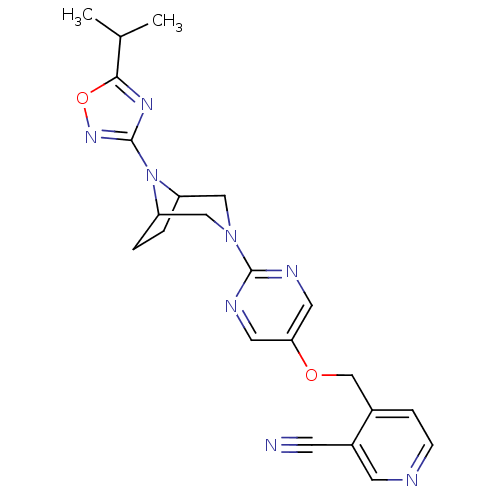
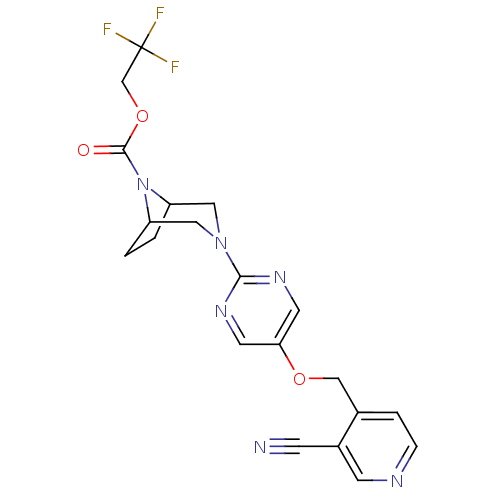
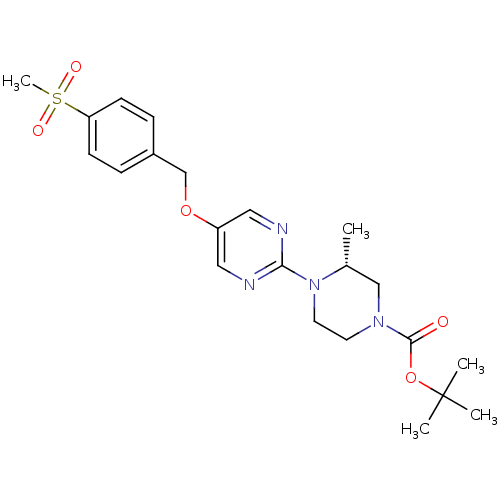
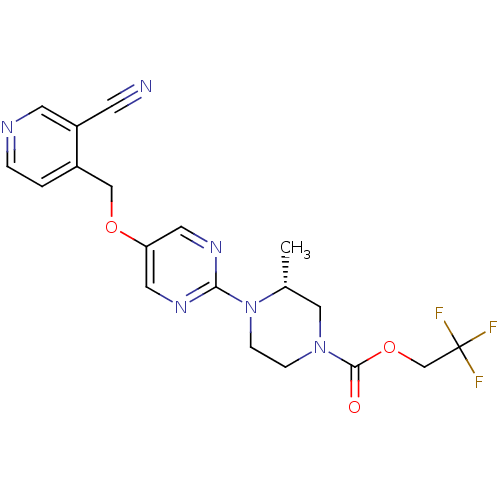
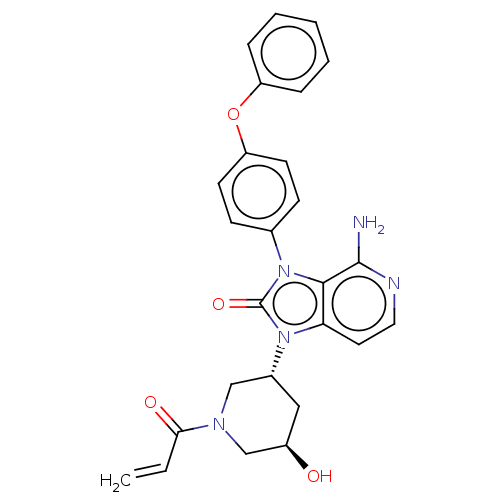
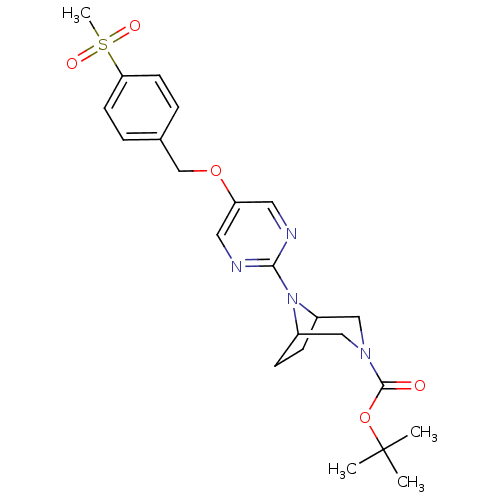
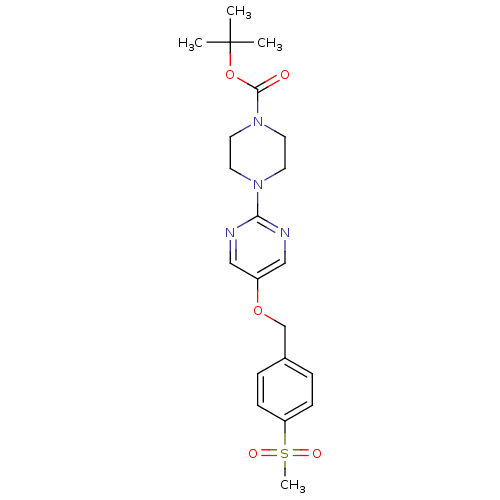
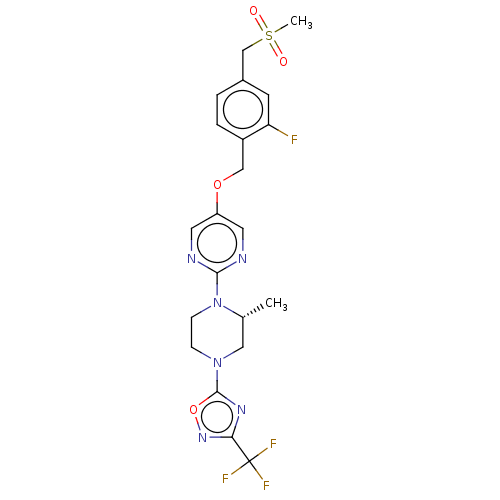
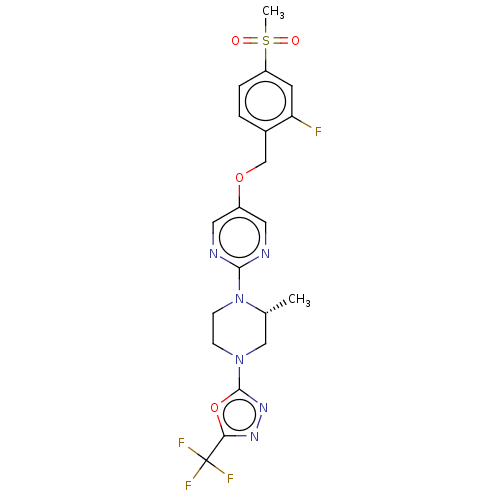
Affinity DataIC50: 1nMAssay Description:Displacement of [3H]-N-(2-fluoro-4-methylsulfonyl-phenyl)-6-[4-(3-isopropyl-1,2,4-oxadiazol-5-yl)-1-piperidyl]-5-nitro-pyrimidin-4-amine from human G...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:Displacement of [3H]-N-(2-fluoro-4-methylsulfonyl-phenyl)-6-[4-(3-isopropyl-1,2,4-oxadiazol-5-yl)-1-piperidyl]-5-nitro-pyrimidin-4-amine from human G...More data for this Ligand-Target Pair
Affinity DataIC50: 3nMAssay Description:Displacement of [3H]-N-(2-fluoro-4-methylsulfonyl-phenyl)-6-[4-(3-isopropyl-1,2,4-oxadiazol-5-yl)-1-piperidyl]-5-nitro-pyrimidin-4-amine from human G...More data for this Ligand-Target Pair
Affinity DataIC50: 3nMAssay Description:Displacement of [3H]-N-(2-fluoro-4-methylsulfonyl-phenyl)-6-[4-(3-isopropyl-1,2,4-oxadiazol-5-yl)-1-piperidyl]-5-nitro-pyrimidin-4-amine from human G...More data for this Ligand-Target Pair
Affinity DataIC50: 4nMAssay Description:Displacement of [3H]-N-(2-fluoro-4-methylsulfonyl-phenyl)-6-[4-(3-isopropyl-1,2,4-oxadiazol-5-yl)-1-piperidyl]-5-nitro-pyrimidin-4-amine from human G...More data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:Displacement of [3H]-N-(2-fluoro-4-methylsulfonyl-phenyl)-6-[4-(3-isopropyl-1,2,4-oxadiazol-5-yl)-1-piperidyl]-5-nitro-pyrimidin-4-amine from mouse G...More data for this Ligand-Target Pair
Affinity DataIC50: 6nMAssay Description:Displacement of [3H]-N-(2-fluoro-4-methylsulfonyl-phenyl)-6-[4-(3-isopropyl-1,2,4-oxadiazol-5-yl)-1-piperidyl]-5-nitro-pyrimidin-4-amine from human G...More data for this Ligand-Target Pair
Affinity DataIC50: 8nMAssay Description:Displacement of [3H]-N-(2-fluoro-4-methylsulfonyl-phenyl)-6-[4-(3-isopropyl-1,2,4-oxadiazol-5-yl)-1-piperidyl]-5-nitro-pyrimidin-4-amine from human G...More data for this Ligand-Target Pair
Affinity DataIC50: 9nMAssay Description:Displacement of [3H]-N-(2-fluoro-4-methylsulfonyl-phenyl)-6-[4-(3-isopropyl-1,2,4-oxadiazol-5-yl)-1-piperidyl]-5-nitro-pyrimidin-4-amine from mouse G...More data for this Ligand-Target Pair
Affinity DataIC50: 9nMAssay Description:Displacement of [3H]-N-(2-fluoro-4-methylsulfonyl-phenyl)-6-[4-(3-isopropyl-1,2,4-oxadiazol-5-yl)-1-piperidyl]-5-nitro-pyrimidin-4-amine from human G...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:Displacement of [3H]-N-(2-fluoro-4-methylsulfonyl-phenyl)-6-[4-(3-isopropyl-1,2,4-oxadiazol-5-yl)-1-piperidyl]-5-nitro-pyrimidin-4-amine from human G...More data for this Ligand-Target Pair
Affinity DataIC50: 12nMAssay Description:Displacement of [3H]-N-(2-fluoro-4-methylsulfonyl-phenyl)-6-[4-(3-isopropyl-1,2,4-oxadiazol-5-yl)-1-piperidyl]-5-nitro-pyrimidin-4-amine from human G...More data for this Ligand-Target Pair
Affinity DataIC50: 17nMAssay Description:Displacement of [3H]-N-(2-fluoro-4-methylsulfonyl-phenyl)-6-[4-(3-isopropyl-1,2,4-oxadiazol-5-yl)-1-piperidyl]-5-nitro-pyrimidin-4-amine from human G...More data for this Ligand-Target Pair
Affinity DataIC50: 18nMAssay Description:Displacement of [3H]-N-(2-fluoro-4-methylsulfonyl-phenyl)-6-[4-(3-isopropyl-1,2,4-oxadiazol-5-yl)-1-piperidyl]-5-nitro-pyrimidin-4-amine from human G...More data for this Ligand-Target Pair
Affinity DataIC50: 25nMAssay Description:Displacement of [3H]-N-(2-fluoro-4-methylsulfonyl-phenyl)-6-[4-(3-isopropyl-1,2,4-oxadiazol-5-yl)-1-piperidyl]-5-nitro-pyrimidin-4-amine from human G...More data for this Ligand-Target Pair
Affinity DataIC50: 31nMAssay Description:Displacement of [3H]-N-(2-fluoro-4-methylsulfonyl-phenyl)-6-[4-(3-isopropyl-1,2,4-oxadiazol-5-yl)-1-piperidyl]-5-nitro-pyrimidin-4-amine from human G...More data for this Ligand-Target Pair
Affinity DataIC50: 34nMAssay Description:Compounds of Formula Ib, Formula II, and Formula III were tested against BTK(h) as described herein. All compounds tested were prepared to a working ...More data for this Ligand-Target Pair
Target InfoPDBMMDBNCI pathwayReactome pathwayKEGG
UniProtKB/SwissProt
B.MOADDrugBankantibodypediaGoogleScholar
UniProtKB/SwissProt
B.MOADDrugBankantibodypediaGoogleScholar
Ligand Info
Affinity DataIC50: 65nMAssay Description:Displacement of [3H]-N-(2-fluoro-4-methylsulfonyl-phenyl)-6-[4-(3-isopropyl-1,2,4-oxadiazol-5-yl)-1-piperidyl]-5-nitro-pyrimidin-4-amine from mouse G...More data for this Ligand-Target Pair
Affinity DataIC50: 72nMAssay Description:Displacement of [3H]-N-(2-fluoro-4-methylsulfonyl-phenyl)-6-[4-(3-isopropyl-1,2,4-oxadiazol-5-yl)-1-piperidyl]-5-nitro-pyrimidin-4-amine from mouse G...More data for this Ligand-Target Pair
Affinity DataIC50: 92nMAssay Description:Displacement of [3H]-N-(2-fluoro-4-methylsulfonyl-phenyl)-6-[4-(3-isopropyl-1,2,4-oxadiazol-5-yl)-1-piperidyl]-5-nitro-pyrimidin-4-amine from human G...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:Displacement of [3H]-N-(2-fluoro-4-methylsulfonyl-phenyl)-6-[4-(3-isopropyl-1,2,4-oxadiazol-5-yl)-1-piperidyl]-5-nitro-pyrimidin-4-amine from mouse G...More data for this Ligand-Target Pair
Affinity DataIC50: 104nMAssay Description:Displacement of [3H]-N-(2-fluoro-4-methylsulfonyl-phenyl)-6-[4-(3-isopropyl-1,2,4-oxadiazol-5-yl)-1-piperidyl]-5-nitro-pyrimidin-4-amine from mouse G...More data for this Ligand-Target Pair
Affinity DataIC50: 122nMAssay Description:Displacement of [3H]-N-(2-fluoro-4-methylsulfonyl-phenyl)-6-[4-(3-isopropyl-1,2,4-oxadiazol-5-yl)-1-piperidyl]-5-nitro-pyrimidin-4-amine from mouse G...More data for this Ligand-Target Pair
Affinity DataIC50: 129nMAssay Description:Displacement of [3H]-N-(2-fluoro-4-methylsulfonyl-phenyl)-6-[4-(3-isopropyl-1,2,4-oxadiazol-5-yl)-1-piperidyl]-5-nitro-pyrimidin-4-amine from mouse G...More data for this Ligand-Target Pair
Affinity DataIC50: 279nMAssay Description:Compounds of Formula Ib, Formula II, and Formula III were tested against BTK(h) as described herein. All compounds tested were prepared to a working ...More data for this Ligand-Target Pair
Target InfoPDBMMDBNCI pathwayReactome pathwayKEGG
UniProtKB/SwissProt
B.MOADDrugBankantibodypediaGoogleScholar
UniProtKB/SwissProt
B.MOADDrugBankantibodypediaGoogleScholar
Ligand Info
Affinity DataIC50: 351nMAssay Description:Displacement of [3H]-N-(2-fluoro-4-methylsulfonyl-phenyl)-6-[4-(3-isopropyl-1,2,4-oxadiazol-5-yl)-1-piperidyl]-5-nitro-pyrimidin-4-amine from human G...More data for this Ligand-Target Pair
Affinity DataIC50: 507nMAssay Description:Displacement of [3H]-N-(2-fluoro-4-methylsulfonyl-phenyl)-6-[4-(3-isopropyl-1,2,4-oxadiazol-5-yl)-1-piperidyl]-5-nitro-pyrimidin-4-amine from mouse G...More data for this Ligand-Target Pair
Affinity DataIC50: 543nMAssay Description:Displacement of [3H]-N-(2-fluoro-4-methylsulfonyl-phenyl)-6-[4-(3-isopropyl-1,2,4-oxadiazol-5-yl)-1-piperidyl]-5-nitro-pyrimidin-4-amine from mouse G...More data for this Ligand-Target Pair
Affinity DataIC50: 568nMAssay Description:Displacement of [3H]-N-(2-fluoro-4-methylsulfonyl-phenyl)-6-[4-(3-isopropyl-1,2,4-oxadiazol-5-yl)-1-piperidyl]-5-nitro-pyrimidin-4-amine from mouse G...More data for this Ligand-Target Pair
Affinity DataIC50: 694nMAssay Description:Displacement of [3H]-N-(2-fluoro-4-methylsulfonyl-phenyl)-6-[4-(3-isopropyl-1,2,4-oxadiazol-5-yl)-1-piperidyl]-5-nitro-pyrimidin-4-amine from mouse G...More data for this Ligand-Target Pair
Affinity DataIC50: 758nMAssay Description:Displacement of [3H]-N-(2-fluoro-4-methylsulfonyl-phenyl)-6-[4-(3-isopropyl-1,2,4-oxadiazol-5-yl)-1-piperidyl]-5-nitro-pyrimidin-4-amine from mouse G...More data for this Ligand-Target Pair
Affinity DataIC50: 1.38E+3nMAssay Description:Compounds of Formula Ib, Formula II, and Formula III were tested against BTK(h) as described herein. All compounds tested were prepared to a working ...More data for this Ligand-Target Pair
Target InfoPDBMMDBNCI pathwayReactome pathwayKEGG
UniProtKB/SwissProt
B.MOADDrugBankantibodypediaGoogleScholar
UniProtKB/SwissProt
B.MOADDrugBankantibodypediaGoogleScholar
Ligand Info
Affinity DataIC50: 1.83E+3nMAssay Description:Displacement of [3H]-N-(2-fluoro-4-methylsulfonyl-phenyl)-6-[4-(3-isopropyl-1,2,4-oxadiazol-5-yl)-1-piperidyl]-5-nitro-pyrimidin-4-amine from mouse G...More data for this Ligand-Target Pair
Affinity DataIC50: 2.10E+3nMAssay Description:Inhibition of EGFR (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 2.72E+3nMAssay Description:Displacement of [3H]-N-(2-fluoro-4-methylsulfonyl-phenyl)-6-[4-(3-isopropyl-1,2,4-oxadiazol-5-yl)-1-piperidyl]-5-nitro-pyrimidin-4-amine from mouse G...More data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
Astrazeneca
Curated by ChEMBL
Astrazeneca
Curated by ChEMBL
Affinity DataIC50: 3.60E+3nMAssay Description:Inhibition of human ERG by patch clamp based electrophysiology methodMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
Astrazeneca
Curated by ChEMBL
Astrazeneca
Curated by ChEMBL
Affinity DataIC50: 3.60E+3nMAssay Description:Inhibition of human ERG by patch clamp based electrophysiology methodMore data for this Ligand-Target Pair
Affinity DataIC50: 4.99E+3nMAssay Description:Displacement of [3H]-N-(2-fluoro-4-methylsulfonyl-phenyl)-6-[4-(3-isopropyl-1,2,4-oxadiazol-5-yl)-1-piperidyl]-5-nitro-pyrimidin-4-amine from mouse G...More data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
Astrazeneca
Curated by ChEMBL
Astrazeneca
Curated by ChEMBL
Affinity DataIC50: 5.70E+3nMAssay Description:Inhibition of human ERG by patch clamp based electrophysiology methodMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
Astrazeneca
Curated by ChEMBL
Astrazeneca
Curated by ChEMBL
Affinity DataIC50: 6.40E+3nMAssay Description:Inhibition of human ERG by patch clamp based electrophysiology methodMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
Astrazeneca
Curated by ChEMBL
Astrazeneca
Curated by ChEMBL
Affinity DataIC50: 6.90E+3nMAssay Description:Inhibition of human ERG by patch clamp based electrophysiology methodMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
Astrazeneca
Curated by ChEMBL
Astrazeneca
Curated by ChEMBL
Affinity DataIC50: 7.40E+3nMAssay Description:Inhibition of human ERG by patch clamp based electrophysiology methodMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
Astrazeneca
Curated by ChEMBL
Astrazeneca
Curated by ChEMBL
Affinity DataIC50: 9.40E+3nMAssay Description:Inhibition of human ERG by patch clamp based electrophysiology methodMore data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+4nMAssay Description:Inhibition of CYP3A4 (unknown origin) by high throughput fluorescence assayMore data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+4nMAssay Description:Inhibition of CYP2D6 (unknown origin) by high throughput fluorescence assayMore data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+4nMAssay Description:Inhibition of CYP2C19 (unknown origin) by high throughput fluorescence assayMore data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+4nMAssay Description:Inhibition of CYP2C9 (unknown origin) by high throughput fluorescence assayMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
Astrazeneca
Curated by ChEMBL
Astrazeneca
Curated by ChEMBL
Affinity DataIC50: 1.00E+4nMAssay Description:Inhibition of human hERGMore data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+4nMAssay Description:Inhibition of CYP1A2 (unknown origin) by high throughput fluorescence assayMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
Astrazeneca
Curated by ChEMBL
Astrazeneca
Curated by ChEMBL
Affinity DataIC50: 1.10E+4nMAssay Description:Inhibition of human ERG by patch clamp based electrophysiology methodMore data for this Ligand-Target Pair