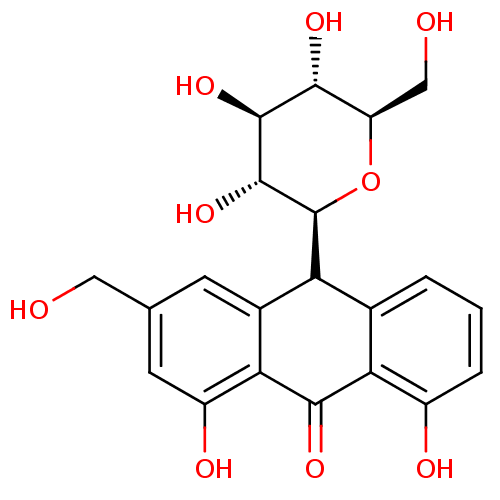
BDBM50269016 CHEMBL497001::aloin
SMILES OC[C@H]1O[C@H]([C@H](O)[C@@H](O)[C@@H]1O)C1c2cccc(O)c2C(=O)c2c(O)cc(CO)cc12
InChI Key InChIKey=AFHJQYHRLPMKHU-CGISPIQUSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 6 hits for monomerid = 50269016
Found 6 hits for monomerid = 50269016
Affinity DataKi: >1.00E+4nMAssay Description:Displacement of [3H]PSB0413 from human platelet P2Y12 receptorMore data for this Ligand-Target Pair
TargetBifunctional epoxide hydrolase 2(Homo sapiens (Human))
Chungnam National University
Curated by ChEMBL
Chungnam National University
Curated by ChEMBL
Affinity DataKi: 5.73E+4nMAssay Description:Mixed-type inhibition of sEH (unknown origin) using PHOME as substrate assessed as formation of 6-methoxy-2-naphthaldehyde measured during 30 mins by...More data for this Ligand-Target Pair
Affinity DataIC50: >3.00E+4nMAssay Description:5-LO inhibitory activity was determined by inhibition of LTB4 biosynthesis in bovine polymorphonuclear leukocytes (PMNL)More data for this Ligand-Target Pair
TargetThioredoxin reductase 2, mitochondrial(Rattus norvegicus)
University Of Padova
Curated by ChEMBL
University Of Padova
Curated by ChEMBL
Affinity DataIC50: >2.00E+5nMAssay Description:Inhibition of rat liver mitochondrial TrxR2 by spectrophotometryMore data for this Ligand-Target Pair
TargetBifunctional epoxide hydrolase 2(Homo sapiens (Human))
Chungnam National University
Curated by ChEMBL
Chungnam National University
Curated by ChEMBL
Affinity DataIC50: 3.74E+4nMAssay Description:Inhibition of sEH (unknown origin) using PHOME as substrate assessed as formation of 6-methoxy-2-naphthaldehyde measured during 1 hr by fluorescence ...More data for this Ligand-Target Pair
Affinity DataIC50: >2.00E+5nMAssay Description:Inhibition of rat liver cytosolic TrxR1 by spectrophotometryMore data for this Ligand-Target Pair