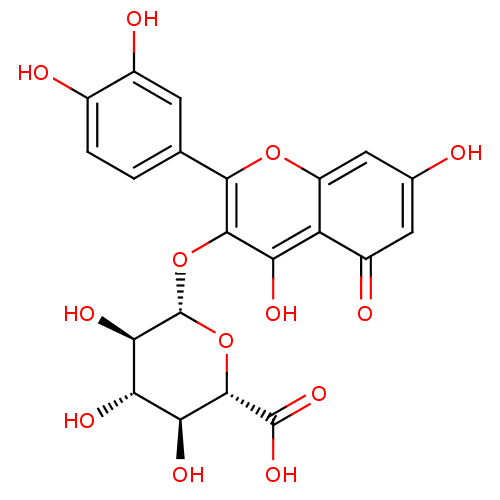
BDBM50242280 (2S,3S,4S,5R,6S)-6-(2-(3,4-dihydroxyphenyl)-5,7-dihydroxy-4-oxo-4H-chromen-3-yloxy)-3,4,5-trihydroxytetrahydro-2H-pyran-2-carboxylic acid::CHEMBL520546::Quercetin 3-O-beta-D-glucuronide::Quercetin-3-O-beta-D-glucuronopyranoside::cid_12004528::quercetin glucuronide
SMILES O[C@H]1[C@H](Oc2c(O)c3c(cc(O)cc3=O)oc2-c2ccc(O)c(O)c2)O[C@@H]([C@@H](O)[C@@H]1O)C(O)=O
InChI Key InChIKey=HUBZKKHGBCSTRB-ZUGPOPFOSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 8 hits for monomerid = 50242280
Found 8 hits for monomerid = 50242280
Affinity DataIC50: 4.82E+4nMAssay Description:Southern Research Molecular Libraries Screening Center (SRMLSC) Southern Research Institute (Birmingham, Alabama) NIH Molecular Libraries Screening...More data for this Ligand-Target Pair
TargetIntestinal-type alkaline phosphatase(Homo sapiens (Human))
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Affinity DataIC50: 3.15E+3nMAssay Description:Data Source: Sanford-Burnham Center for Chemical Genomics (SBCCG) Source Affiliation: Sanford-Burnham Medical Research Institute (SBMRI, San Diego, C...More data for this Ligand-Target Pair
TargetIntestinal-type alkaline phosphatase 1(Rattus norvegicus (Rat))
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Affinity DataIC50: 3.29E+3nMAssay Description:Data Source: Sanford-Burnham Center for Chemical Genomics (SBCCG) Source Affiliation: Sanford-Burnham Medical Research Institute (SBMRI, San Diego, C...More data for this Ligand-Target Pair
TargetAngiotensin-converting enzyme(Oryctolagus cuniculus)
India Institute Of Chemical Technology
Curated by ChEMBL
India Institute Of Chemical Technology
Curated by ChEMBL
Affinity DataIC50: 2.00E+5nMAssay Description:Inhibition of rabbit lung ACE assessed as reduction in hippuryl-histidyl-leucine substrate by colorimetric assayMore data for this Ligand-Target Pair
TargetAlkaline phosphatase, germ cell type(Homo sapiens (Human))
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Affinity DataEC50: >1.00E+5nMAssay Description:Data Source: Sanford-Burnham Center for Chemical Genomics (SBCCG) Source Affiliation: Sanford-Burnham Medical Research Institute (SBMRI, San Diego, C...More data for this Ligand-Target Pair
TargetIntestinal-type alkaline phosphatase(Homo sapiens (Human))
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Affinity DataIC50: 891nMAssay Description:Data Source: Sanford-Burnham Center for Chemical Genomics (SBCCG) Source Affiliation: Sanford-Burnham Medical Research Institute(SBMRI, San Diego, CA...More data for this Ligand-Target Pair
TargetAngiotensin-converting enzyme(Oryctolagus cuniculus)
India Institute Of Chemical Technology
Curated by ChEMBL
India Institute Of Chemical Technology
Curated by ChEMBL
Affinity DataIC50: 2.00E+5nMAssay Description:Inhibition of ACE in rabbit lung assessed as decrease in dansylglycine concentration after 5 mins by HPLC analysisMore data for this Ligand-Target Pair
TargetAlkaline phosphatase, tissue-nonspecific isozyme(Homo sapiens (Human))
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Affinity DataIC50: >1.00E+5nMAssay Description:Data Source: Sanford-Burnham Center for Chemical Genomics (SBCCG) Source Affiliation: Sanford-Burnham Medical Research Institute(SBMRI, San Diego, CA...More data for this Ligand-Target Pair