BDBM50462002 CHEMBL4226994
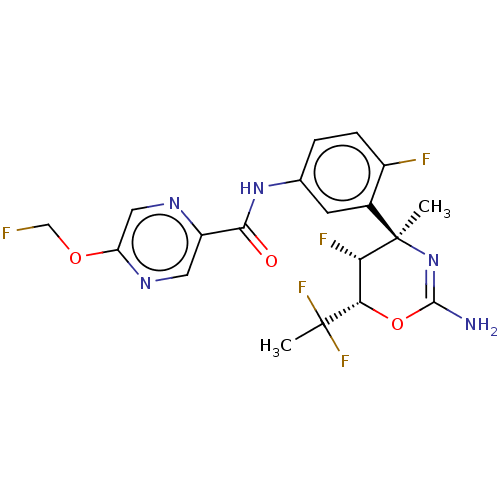
SMILES C[C@]1([C@H]([C@H](OC(=N1)N)C(C)(F)F)F)c2cc(ccc2F)NC(=O)c3cnc(cn3)OCF
InChI Key InChIKey=DKIWJWSKVCKJPV-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 5 hits for monomerid = 50462002
Found 5 hits for monomerid = 50462002
Affinity DataIC50: 1.90nMAssay Description:Inhibition of BACE1 in human SH-SY5Y cells harboring wild-type human beta-APP assessed as reduction in secreted amyloid beta (1 to 40) after 24 hrs b...More data for this Ligand-Target Pair
Affinity DataIC50: 8.70nMAssay Description:Inhibition of recombinant human BACE1 (43 to 454 residues) expressed in Escherichia coli BL21(DE3) using Biotin-epsilon-amino-n-capronic acid-SEVNLDA...More data for this Ligand-Target Pair
Affinity DataIC50: 44nMAssay Description:Inhibition of BACE2 (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 5.10E+3nMAssay Description:Inhibition of CYP2D6 in human liver microsomes using dextromethorphan as substrate after 15 mins in presence of NADPH by LC-MS/MS analysisMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+5nMAssay Description:Inhibition of Cathepsin D (unknown origin)More data for this Ligand-Target Pair
 3D Structure (crystal)
3D Structure (crystal)