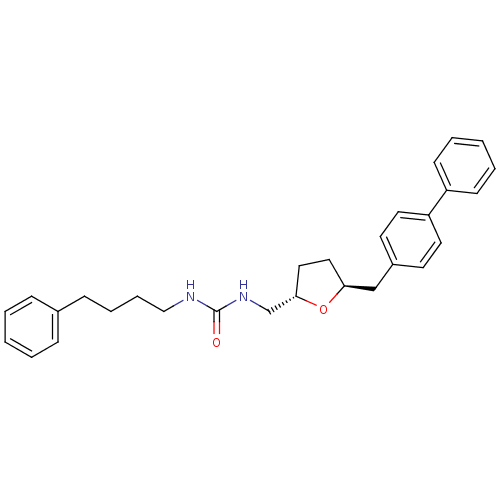
BDBM76251 1-(4-phenylbutyl)-3-[[(2S,5S)-5-[(4-phenylphenyl)methyl]-2-oxolanyl]methyl]urea::1-(4-phenylbutyl)-3-[[(2S,5S)-5-[(4-phenylphenyl)methyl]oxolan-2-yl]methyl]urea::1-[[(2S,5S)-5-(4-phenylbenzyl)tetrahydrofuran-2-yl]methyl]-3-(4-phenylbutyl)urea::MLS002603698::SMR001522428::cid_42627831
SMILES O=C(NCCCCc1ccccc1)NC[C@@H]1CC[C@@H](Cc2ccc(cc2)-c2ccccc2)O1
InChI Key InChIKey=ZMFMXLUFGRGZSQ-NSOVKSMOSA-N
Data 4 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 4 hits for monomerid = 76251
Found 4 hits for monomerid = 76251
TargetIntestinal-type alkaline phosphatase(Homo sapiens (Human))
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Affinity DataIC50: 2.10E+3nMAssay Description:Data Source: Sanford-Burnham Center for Chemical Genomics (SBCCG) Source Affiliation: Sanford-Burnham Medical Research Institute(SBMRI, San Diego, CA...More data for this Ligand-Target Pair
TargetIntestinal-type alkaline phosphatase 1(Rattus norvegicus (Rat))
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Affinity DataIC50: 2.60E+3nMAssay Description:Data Source: Sanford-Burnham Center for Chemical Genomics (SBCCG) Source Affiliation: Sanford-Burnham Medical Research Institute (SBMRI, San Diego, C...More data for this Ligand-Target Pair
TargetAlkaline phosphatase, tissue-nonspecific isozyme(Homo sapiens (Human))
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Affinity DataIC50: 1.56E+4nMAssay Description:Data Source: Sanford-Burnham Center for Chemical Genomics (SBCCG) Source Affiliation: Sanford-Burnham Medical Research Institute (SBMRI, San Diego, C...More data for this Ligand-Target Pair
TargetAlkaline phosphatase, germ cell type(Homo sapiens (Human))
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Affinity DataIC50: 6.58E+3nMAssay Description:Data Source: Sanford-Burnham Center for Chemical Genomics (SBCCG) Source Affiliation: Sanford-Burnham Medical Research Institute (SBMRI, San Diego, C...More data for this Ligand-Target Pair