Affinity DataIC50: 0.100nMAssay Description:The binding affinity of the present compounds to human 5-HT1A receptor, human D4 receptor, and human D2 receptor was measured in a manner mentioned b...More data for this Ligand-Target Pair
Affinity DataIC50: 0.110nMAssay Description:Inhibition of 5-HT1a (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 0.129nMAssay Description:Binding affinity towards 5-hydroxytryptamine 1A receptor by displacement of [3H]8-OH-DPAT.More data for this Ligand-Target Pair
Affinity DataIC50: 0.140nMAssay Description:Inhibition of 5-HT1a (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 0.200nMAssay Description:Binding affinity towards 5-hydroxytryptamine 1A receptor by displacement of [3H]8-OH-DPAT.More data for this Ligand-Target Pair
Affinity DataIC50: 0.200nMAssay Description:Affinity towards 5-hydroxytryptamine 1A receptor in membranes from bovine hippocampus using [3H]-OH-DPATMore data for this Ligand-Target Pair
Affinity DataIC50: 0.200nMAssay Description:Affinity towards 5-hydroxytryptamine 1A receptor in membranes from bovine hippocampus using [3H]-OH-DPATMore data for this Ligand-Target Pair
Affinity DataIC50: 0.200nMAssay Description:The binding affinity of the present compounds to human 5-HT1A receptor, human D4 receptor, and human D2 receptor was measured in a manner mentioned b...More data for this Ligand-Target Pair
Affinity DataIC50: 0.200nMAssay Description:Agonist activity at human 5HT1A expressed in CHO cell membranes assessed as increase in [35S]-GTPgammaS binding after 30 mins by liquid scintillation...More data for this Ligand-Target Pair
Affinity DataIC50: 0.200nMAssay Description:The compound was evaluated for its ability to displace [3H]-8-OH-DPAT from 5-hydroxytryptamine 1A receptor in cellular brain membranesMore data for this Ligand-Target Pair
Affinity DataIC50: 0.200nMAssay Description:Affinity towards 5-hydroxytryptamine 1A receptor in membranes from bovine hippocampus using [3H]-OH-DPATMore data for this Ligand-Target Pair
Affinity DataIC50: 0.280nMAssay Description:Displacement of [3H]OH-DPAT from human recombinant 5-HT1A receptor measured after 60 mins by scintillation counter methodMore data for this Ligand-Target Pair
Affinity DataIC50: 0.300nMAssay Description:The binding affinity of the present compounds to human 5-HT1A receptor, human D4 receptor, and human D2 receptor was measured in a manner mentioned b...More data for this Ligand-Target Pair
Affinity DataIC50: 0.300nMAssay Description:Affinity towards 5-hydroxytryptamine 1A receptor in membranes from bovine hippocampus using [3H]-OH-DPATMore data for this Ligand-Target Pair
Affinity DataIC50: 0.300nMAssay Description:The compound was evaluated for its ability to displace [3H]-8-OH-DPAT from 5-hydroxytryptamine 1A receptor in cellular brain membranesMore data for this Ligand-Target Pair
Affinity DataIC50: 0.300nMAssay Description:In Vitro Binding affinity againist 5-HT1A receptor by displacing [3H]8-OH-DPAT from pig cortexMore data for this Ligand-Target Pair
Affinity DataIC50: 0.398nMAssay Description:Ability to bind to 5-hydroxytryptamine 1A receptor from cloned human expressed in Ha7 cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 0.400nMAssay Description:The binding affinity of the present compounds to human 5-HT1A receptor, human D4 receptor, and human D2 receptor was measured in a manner mentioned b...More data for this Ligand-Target Pair
Affinity DataIC50: 0.400nMAssay Description:The binding affinity of the present compounds to human 5-HT1A receptor, human D4 receptor, and human D2 receptor was measured in a manner mentioned b...More data for this Ligand-Target Pair
Affinity DataIC50: 0.400nMAssay Description:The binding affinity of the present compounds to human 5-HT1A receptor, human D4 receptor, and human D2 receptor was measured in a manner mentioned b...More data for this Ligand-Target Pair
Affinity DataIC50: 0.430nMAssay Description:Displacement of [3H]8-OH-DPAT from recombinant human 5-HT1A receptor after 60 mins by scintillation counting analysisMore data for this Ligand-Target Pair
Affinity DataIC50: 0.5nMAssay Description:Inhibition of 5-HT1a (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 0.530nMAssay Description:Displacement of [3H]8-OHDPAT from human recombinant 5-HT1A receptor expressed in HEK293 cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 0.530nMAssay Description:Displacement of [3H]8-OH-DPAT from human recombinant 5HT1A receptor expressed in HEK293 cells measured after 60 mins by scintillation counting methodMore data for this Ligand-Target Pair
Affinity DataIC50: 0.600nMAssay Description:Affinity towards 5-hydroxytryptamine 1A receptor in membranes from bovine hippocampus using [3H]-OH-DPATMore data for this Ligand-Target Pair
Affinity DataIC50: 0.600nMAssay Description:Displacement of [3H]8-OH-DPAT from recombinant human 5-HT1A receptor after 60 mins by scintillation counting analysisMore data for this Ligand-Target Pair
Affinity DataIC50: 0.700nMAssay Description:Affinity towards 5-hydroxytryptamine 1A receptor in membranes from bovine hippocampus using [3H]-OH-DPATMore data for this Ligand-Target Pair
Affinity DataIC50: 0.700nMAssay Description:Affinity towards 5-hydroxytryptamine 1A receptor in membranes from bovine hippocampus using [3H]-OH-DPATMore data for this Ligand-Target Pair
Affinity DataIC50: 0.700nMAssay Description:Agonist activity at human 5HT1A expressed in CHO cell membranes assessed as increase in [35S]-GTPgammaS binding after 30 mins by liquid scintillation...More data for this Ligand-Target Pair
Affinity DataIC50: 0.700nMAssay Description:Affinity towards 5-hydroxytryptamine 1A receptor in membranes from bovine hippocampus using [3H]-OH-DPATMore data for this Ligand-Target Pair
Affinity DataIC50: 0.770nMAssay Description:Displacement of [3H]8-OH-DPAT from human 5HT1A receptor expressed in HEK293 EBNA cells after 2 hrs by liquid scintillation countingMore data for this Ligand-Target Pair
Affinity DataIC50: 0.770nMAssay Description:Displacement of [3H]8-OH-DPAT from human cloned 5HT1A receptor expressed in HEK293 EBNA cells after 1 hr by liquid scintillation countingMore data for this Ligand-Target Pair
Affinity DataIC50: 0.800nMAssay Description:Affinity towards 5-hydroxytryptamine 1A receptor in membranes from bovine hippocampus using [3H]-OH-DPATMore data for this Ligand-Target Pair
Affinity DataIC50: 0.800nMAssay Description:Affinity towards 5-hydroxytryptamine 1A receptor in membranes from bovine hippocampus using [3H]-OH-DPATMore data for this Ligand-Target Pair
Affinity DataIC50: 0.800nMAssay Description:Inhibition of 5HT1A receptorMore data for this Ligand-Target Pair
Affinity DataIC50: 0.800nMAssay Description:The binding affinity of the present compounds to human 5-HT1A receptor, human D4 receptor, and human D2 receptor was measured in a manner mentioned b...More data for this Ligand-Target Pair
Affinity DataIC50: 0.910nMAssay Description:Displacement of [3H]8-OH-DPAT from human 5HT1A receptor expressed in HEK293 EBNA cells after 2 hrs by liquid scintillation countingMore data for this Ligand-Target Pair
Affinity DataIC50: 0.910nMAssay Description:Displacement of [3H]8-OH-DPAT from human cloned 5HT1A receptor expressed in HEK293 EBNA cells after 1 hr by liquid scintillation countingMore data for this Ligand-Target Pair
Affinity DataKi: 0.650nM IC50: 0.970nMAssay Description:Receptor Source: Human recombinant expressed in HEK-293 cells Radioligand: [3H]-8-OH-DPAT (221 Ci/mmol) Control Compound: 8-OH-DPAT Incubati...More data for this Ligand-Target Pair
Affinity DataIC50: 0.970nMAssay Description:Serotonin, 5HT1A:Receptor Source: Human recombinant expressed in HEK-293 cellsRadioligand: [3H]-8-OH-DPAT (221 Ci/mmol)Control Compound: 8-OH-DPATInc...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:The compound was evaluated for its ability to displace [3H]-8-OH-DPAT from 5-hydroxytryptamine 1A receptor in cellular brain membranesMore data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:Agonist activity at human 5HT1A expressed in CHO cell membranes assessed as increase in [35S]-GTPgammaS binding after 30 mins by liquid scintillation...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:Displacement of [3H]8-OH-DPAT from human recombinant 5HT1A receptor expressed in HEK293 cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:Affinity towards 5-hydroxytryptamine 1A receptor in membranes from bovine hippocampus using [3H]-OH-DPATMore data for this Ligand-Target Pair
Affinity DataIC50: 1.10nMAssay Description:Antagonist activity at human recombinant 5-HT1A receptor expressed in CHOK1 cells incubated for 25 mins followed by addition of methiothepin by micro...More data for this Ligand-Target Pair
Affinity DataIC50: 1.10nMAssay Description:Antagonist activity at human recombinant 5-HT1A receptor expressed in CHOK1 cells incubated for 25 mins followed by addition of methiothepin by micro...More data for this Ligand-Target Pair
Affinity DataIC50: 1.10nMAssay Description:The binding affinity of the present compounds to human 5-HT1A receptor, human D4 receptor, and human D2 receptor was measured in a manner mentioned b...More data for this Ligand-Target Pair
Affinity DataIC50: 1.10nMAssay Description:The binding affinity of the present compounds to human 5-HT1A receptor, human D4 receptor, and human D2 receptor was measured in a manner mentioned b...More data for this Ligand-Target Pair
Affinity DataIC50: 1.19nMAssay Description:Displacement of [3H]8-OH-DPAT from human cloned 5HT1A receptor expressed in HEK293 EBNA cells after 1 hr by liquid scintillation countingMore data for this Ligand-Target Pair
Affinity DataIC50: 1.20nMAssay Description:In vitro inhibitory concentration required against [3H]8-OH-DPAT binding to cloned human 5-hydroxytryptamine 1A receptorMore data for this Ligand-Target Pair

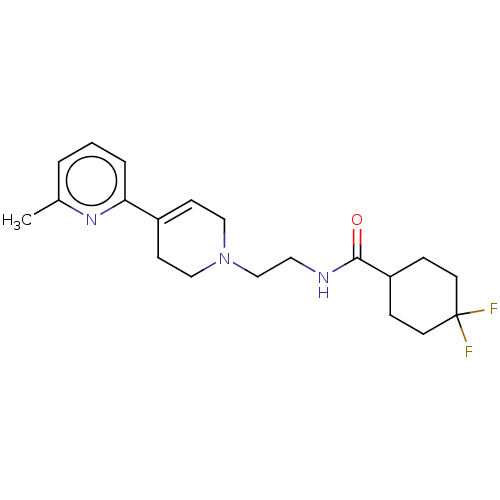
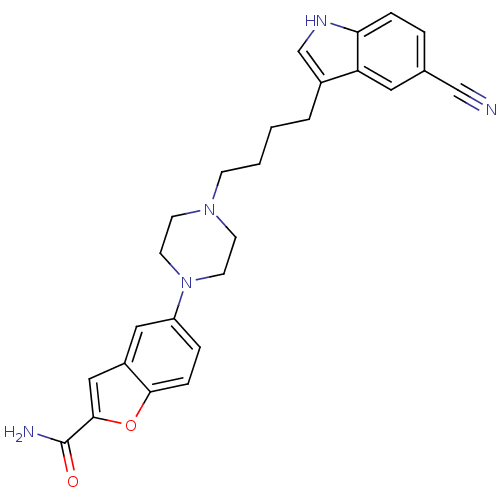
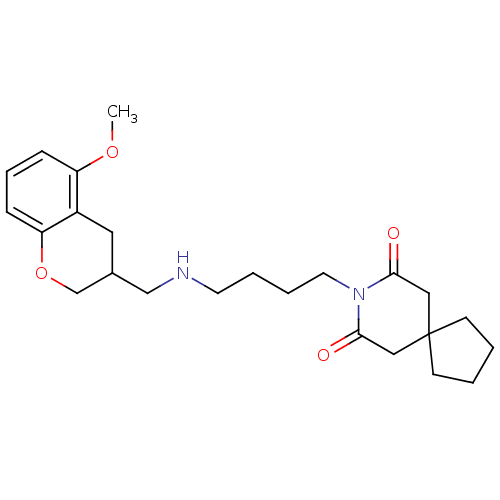
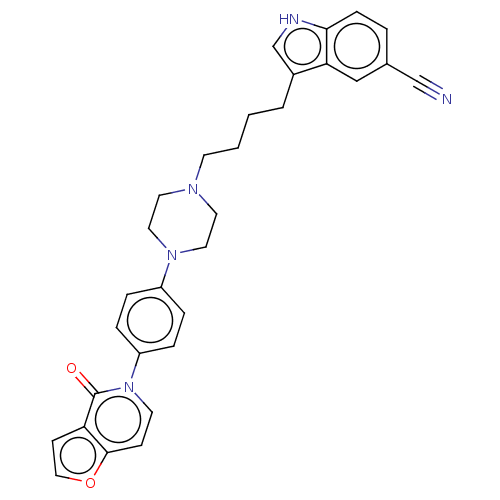
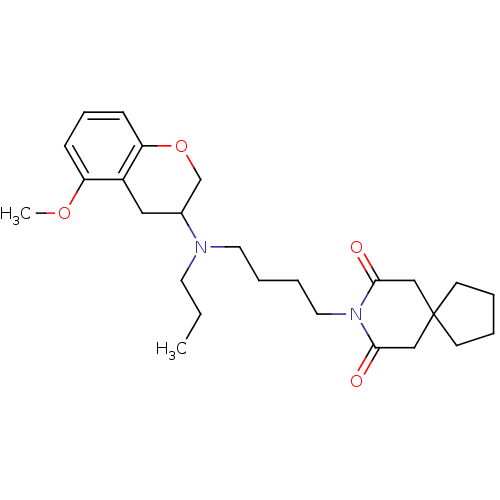

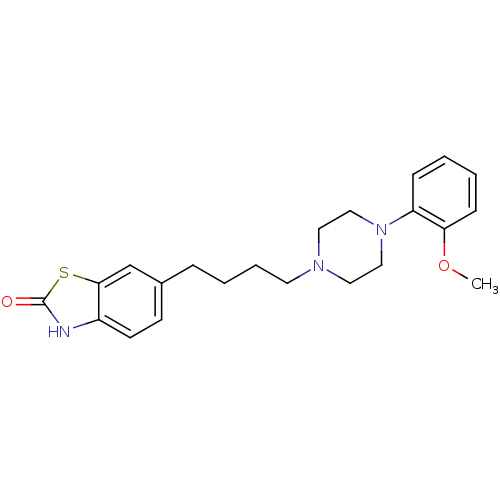
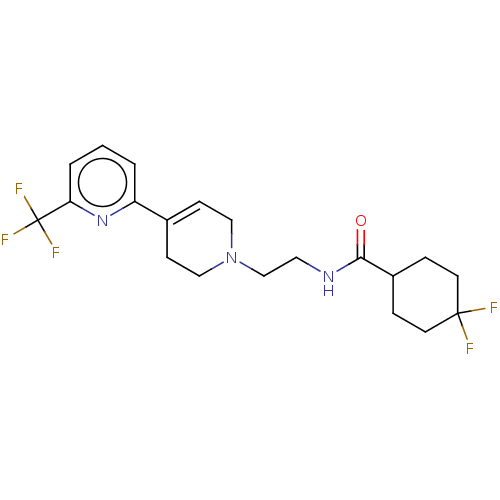

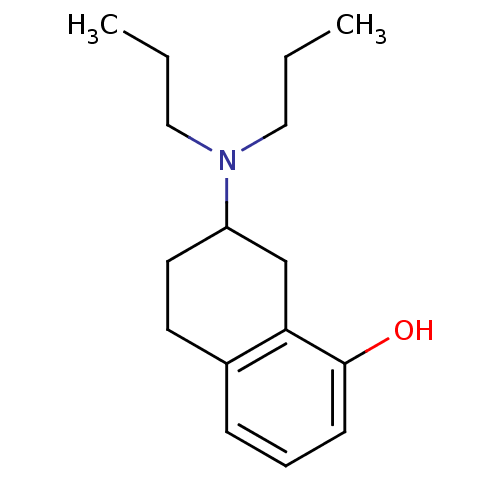
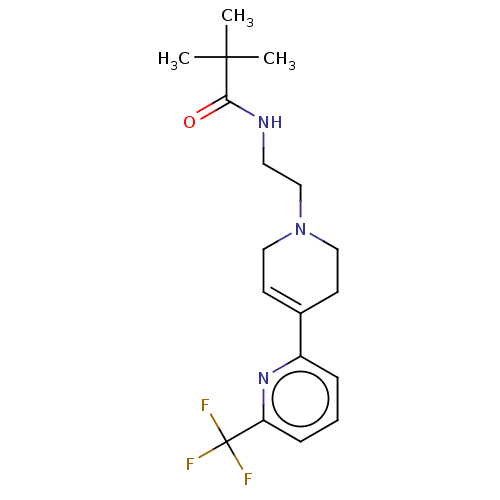

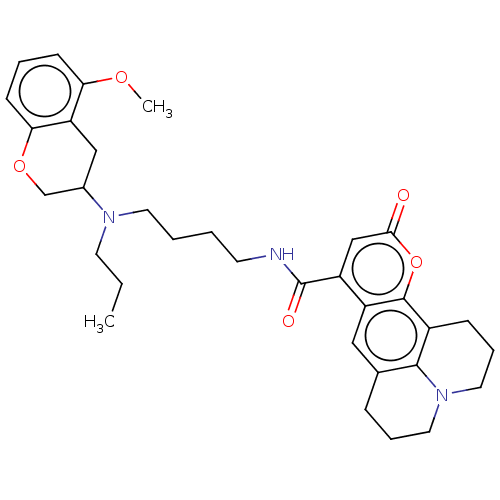
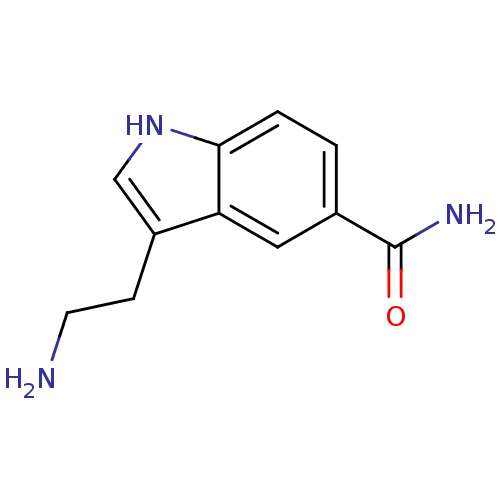

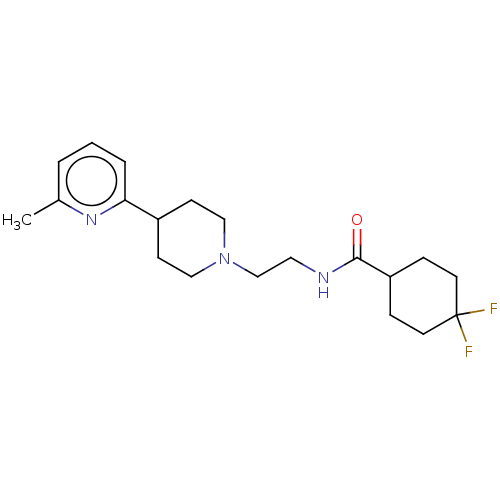
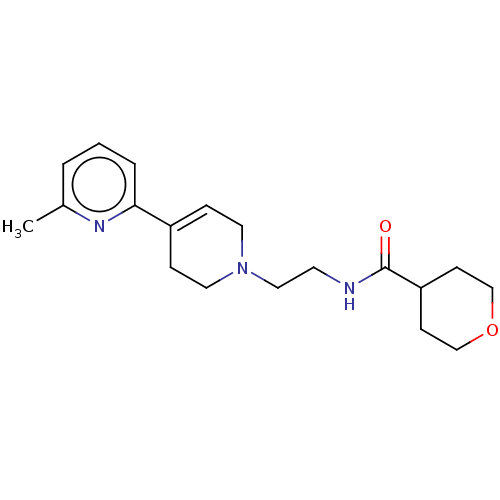
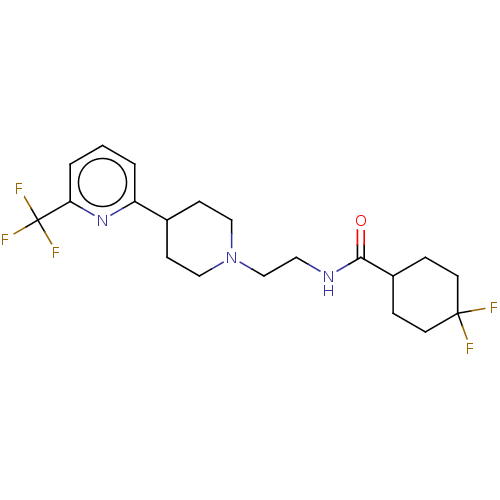
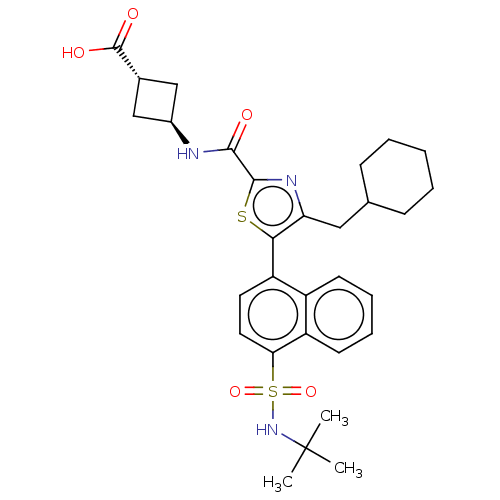
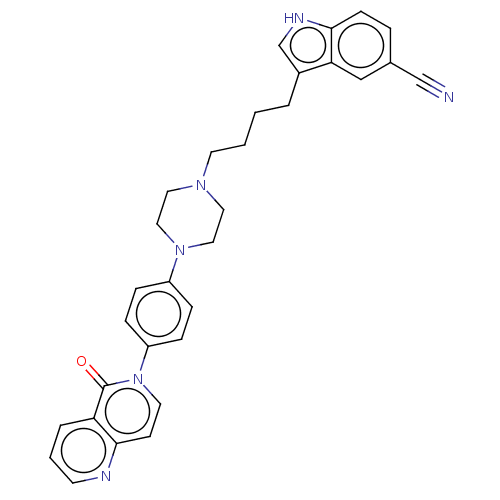

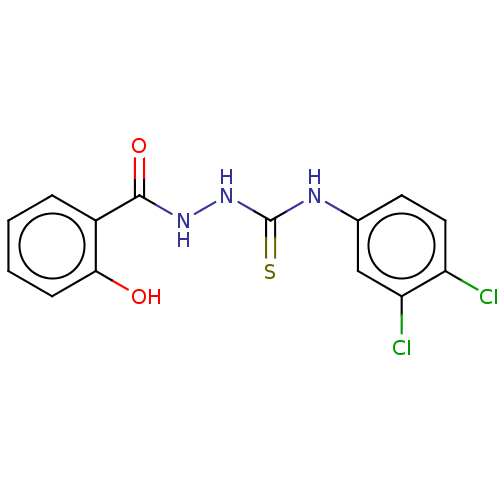

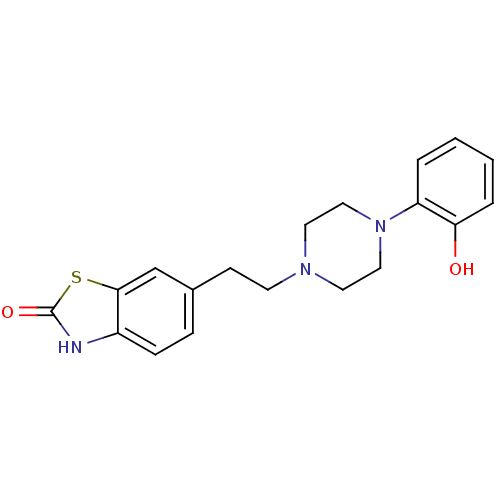

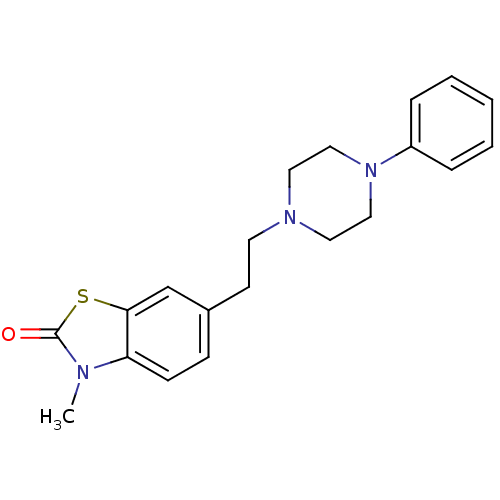
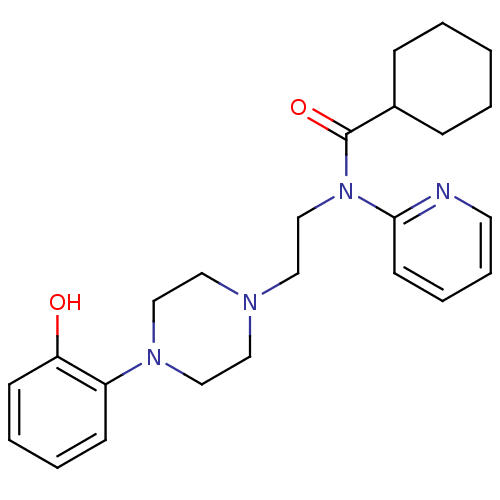
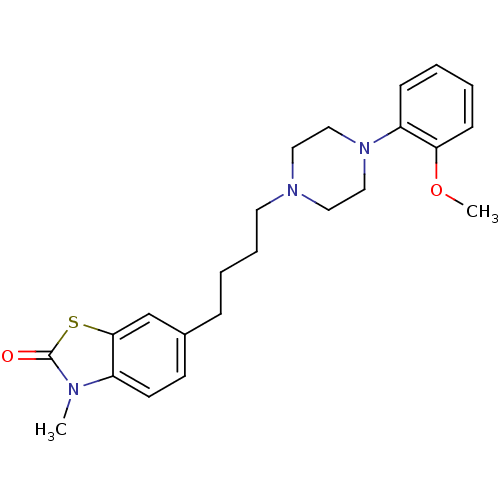



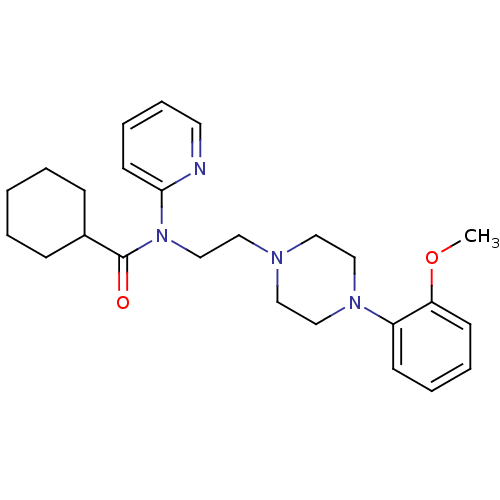
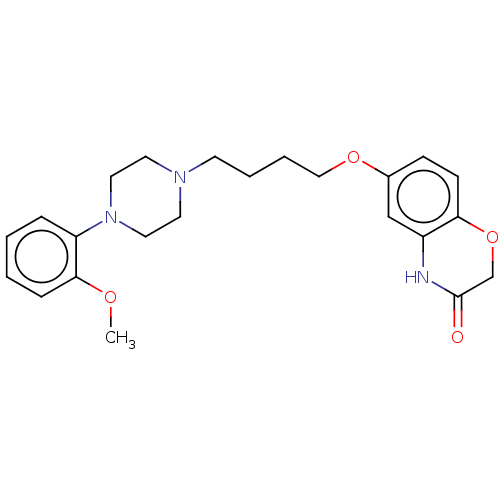

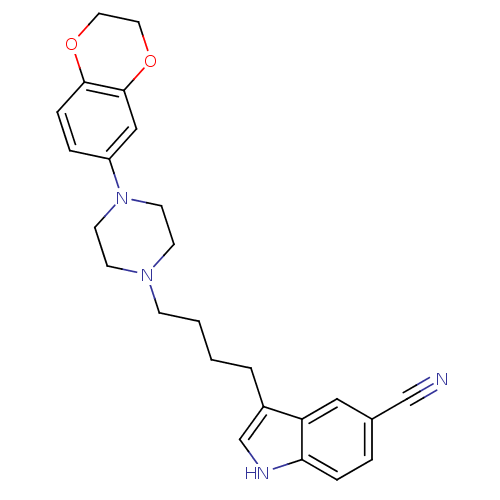
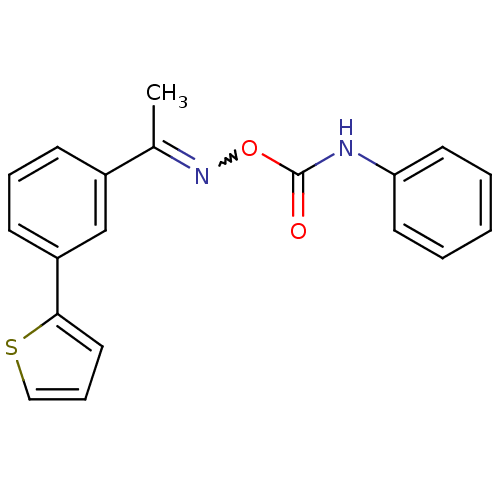
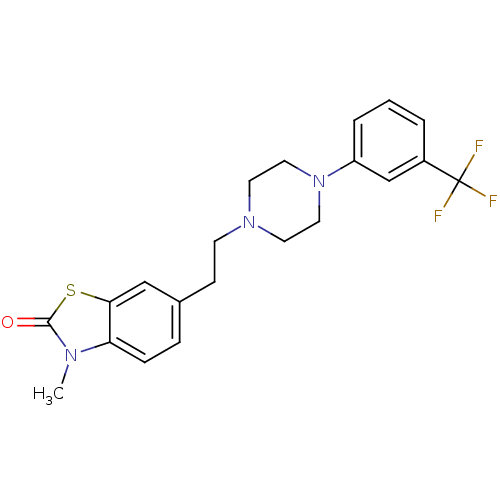

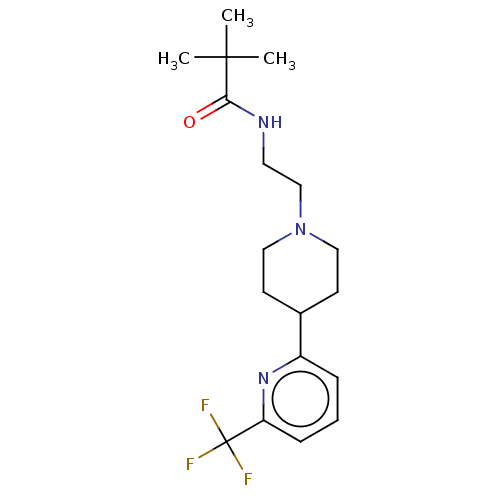
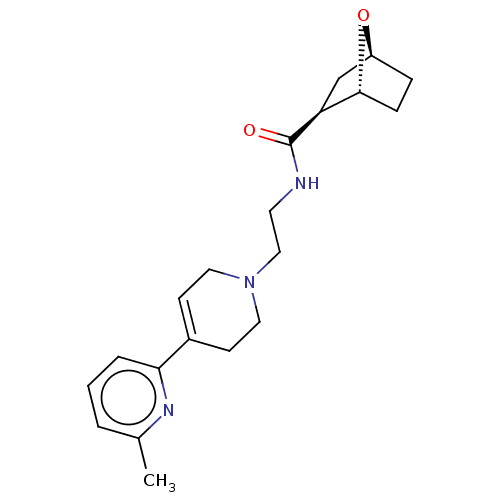

 3D Structure (crystal)
3D Structure (crystal)