Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Integrase
Ligand
BDBM50527151
Substrate
n/a
Meas. Tech.
ChEMBL_1902393 (CHEMBL4404615)
Ki
>40000±n/a nM
Citation
 Li, G; Meanwell, NA; Krystal, MR; Langley, DR; Naidu, BN; Sivaprakasam, P; Lewis, H; Kish, K; Khan, JA; Ng, A; Trainor, GL; Cianci, C; Dicker, IB; Walker, MA; Lin, Z; Protack, T; Discotto, L; Jenkins, S; Gerritz, SW; Pendri, A Discovery and Optimization of Novel Pyrazolopyrimidines as Potent and Orally Bioavailable Allosteric HIV-1 Integrase Inhibitors. J Med Chem 63:2620-2637 (2020) [PubMed] Article
Li, G; Meanwell, NA; Krystal, MR; Langley, DR; Naidu, BN; Sivaprakasam, P; Lewis, H; Kish, K; Khan, JA; Ng, A; Trainor, GL; Cianci, C; Dicker, IB; Walker, MA; Lin, Z; Protack, T; Discotto, L; Jenkins, S; Gerritz, SW; Pendri, A Discovery and Optimization of Novel Pyrazolopyrimidines as Potent and Orally Bioavailable Allosteric HIV-1 Integrase Inhibitors. J Med Chem 63:2620-2637 (2020) [PubMed] Article More Info.:
Target
Name:
Integrase
Synonyms:
pol
Type:
PROTEIN
Mol. Mass.:
32203.43
Organism:
Human immunodeficiency virus 1
Description:
ChEMBL_106649
Residue:
288
Sequence:
FLDGIDKAQEEHEKYHSNWRAMASDFNLPPVVAKEIVASCDKCQLKGEAMHGQVDCSPGIWQLDCTHLEGKVILVAVHVASGYIEAEVIPAETGQETAYFLLKLAGRWPVKTVHTDNGSNFTSTTVKAACWWAGIKQEFGIPYNPQSQGVIESMNKELKKIIGQVRDQAEHLKTAVQMAVFIHNFKRKGGIGGYSAGERIVDIIATDIQTKELQKQITKIQNFRVYYRDSRDPVWKGPAKLLWKGEGAVVIQDNSDIKVVPRRKAKIIRDYGKQMAGDDCVASRQDED
Inhibitor
Name:
BDBM50527151
Synonyms:
CHEMBL4440179
Type:
Small organic molecule
Emp. Form.:
C26H27N3O4
Mol. Mass.:
445.5103
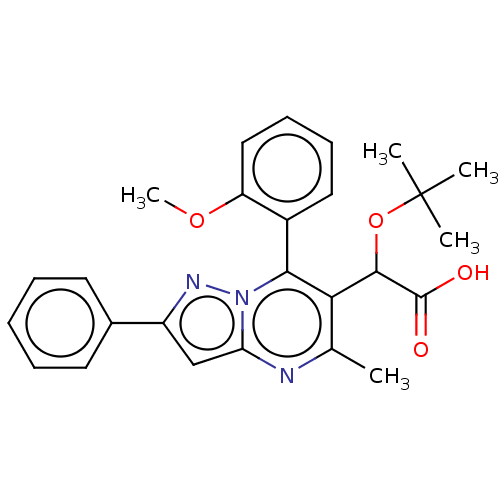
SMILES:
COc1ccccc1-c1c(C(OC(C)(C)C)C(O)=O)c(C)nc2cc(nn12)-c1ccccc1 |(44.98,-21.36,;46.31,-22.12,;47.63,-21.34,;47.62,-19.8,;48.95,-19.02,;50.29,-19.78,;50.3,-21.32,;48.97,-22.1,;48.98,-23.63,;50.31,-24.39,;51.64,-23.61,;51.63,-22.07,;52.96,-21.3,;52.96,-19.76,;54.3,-22.06,;54.29,-20.51,;52.98,-24.38,;54.31,-23.6,;52.98,-25.92,;50.32,-25.94,;51.65,-26.71,;48.98,-26.71,;47.64,-25.95,;46.18,-26.42,;45.27,-25.17,;46.18,-23.93,;47.65,-24.41,;43.73,-25.17,;42.97,-23.84,;41.43,-23.83,;40.65,-25.17,;41.43,-26.51,;42.97,-26.5,)|