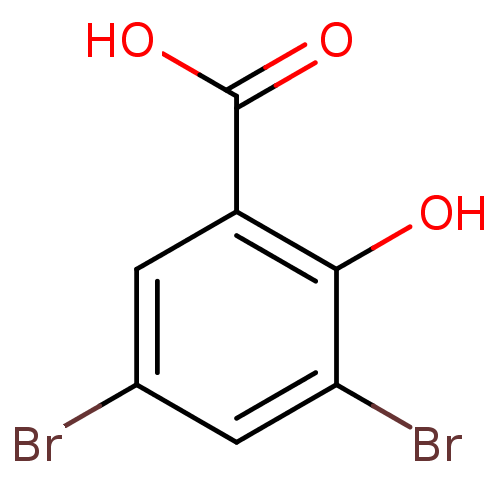
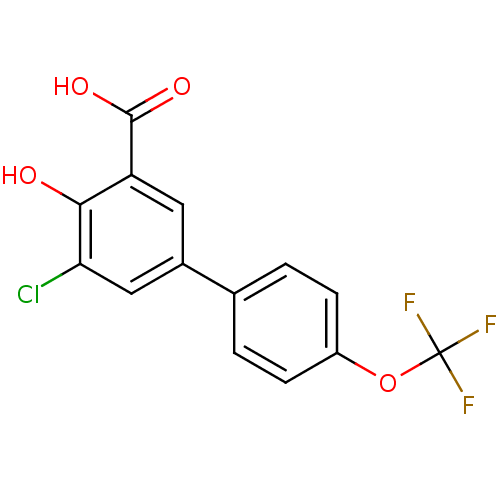
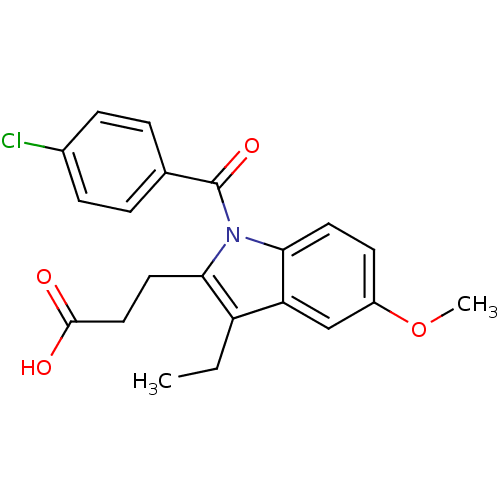
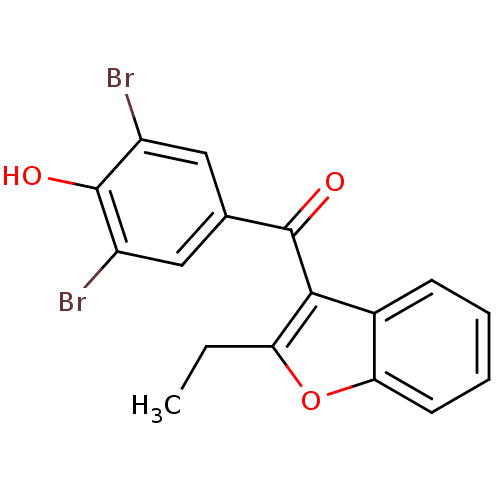
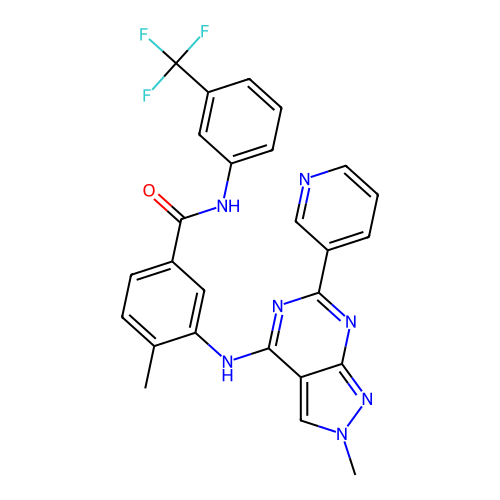
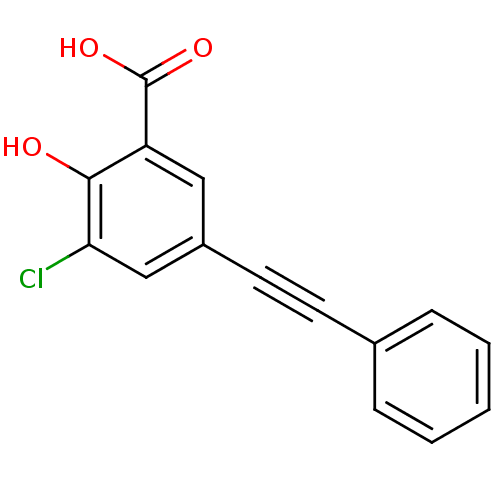
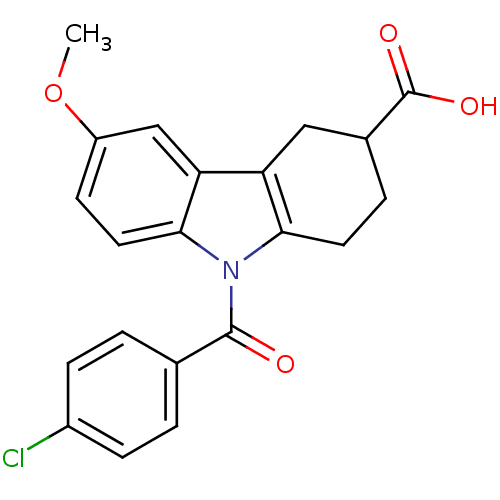
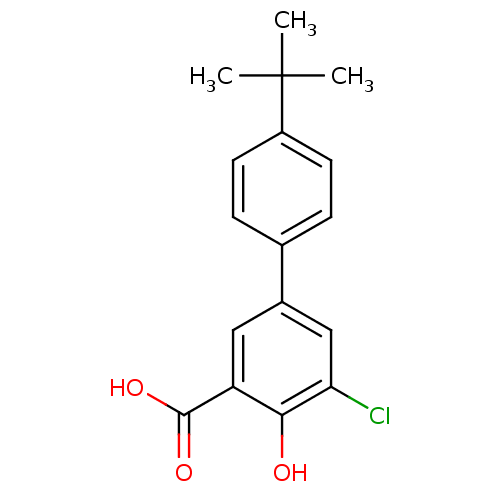
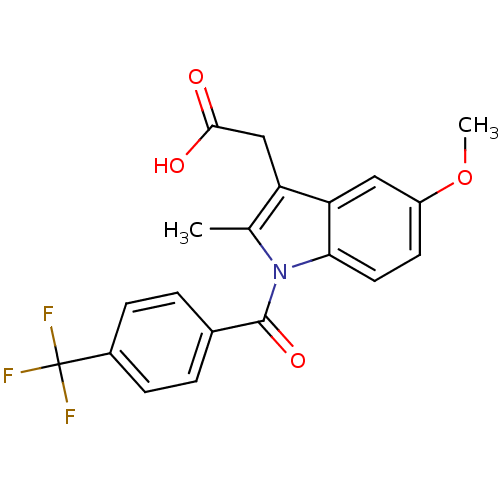
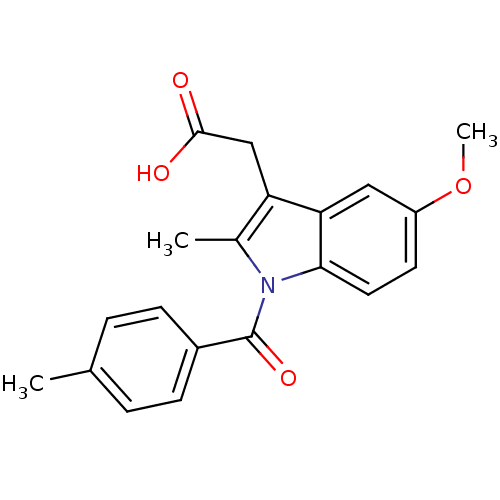
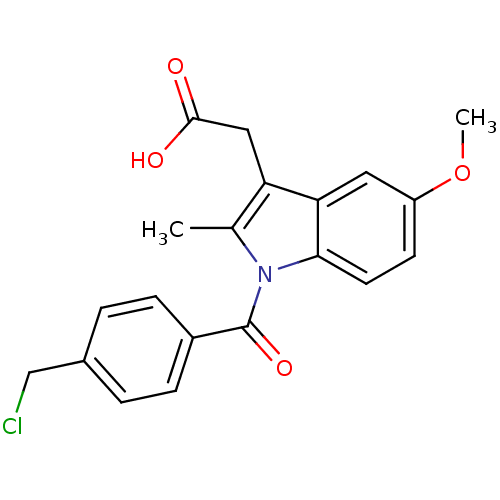
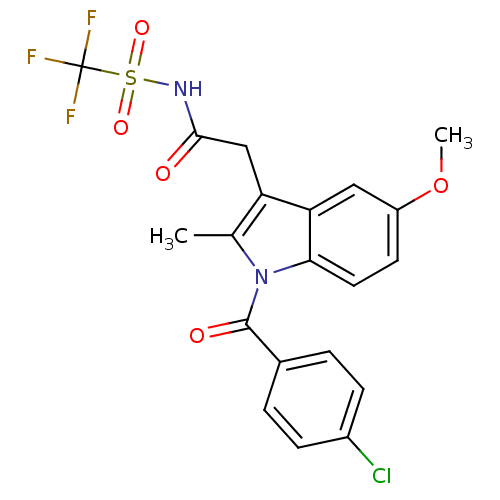
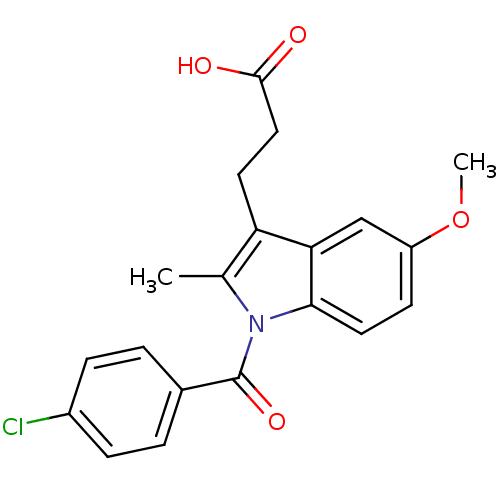
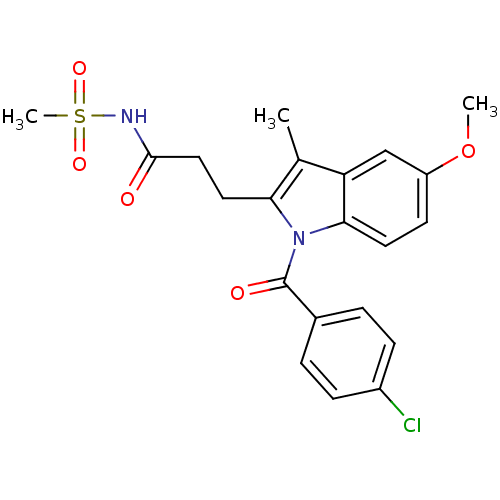
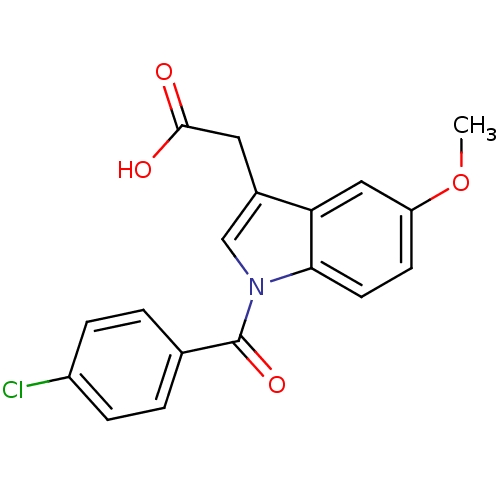
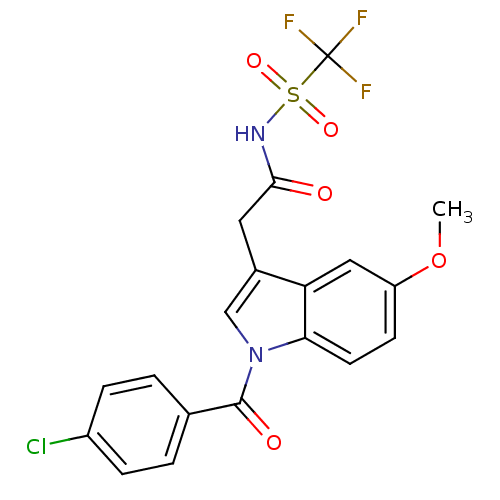
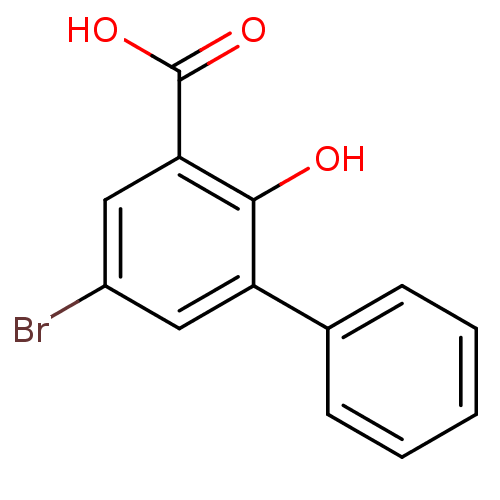
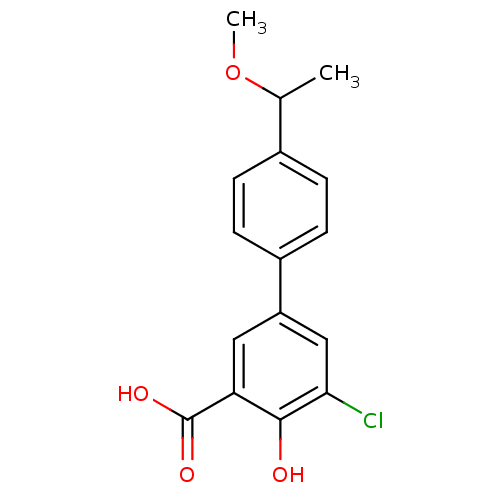
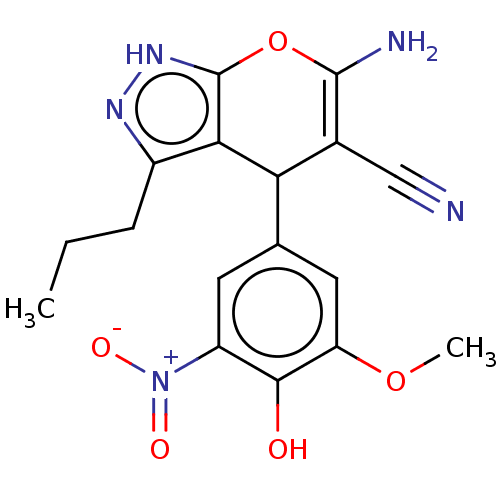
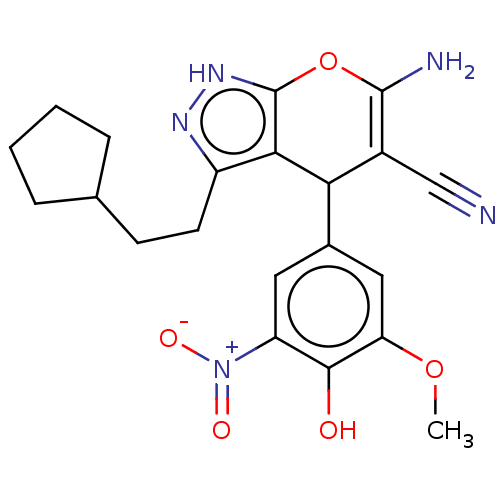
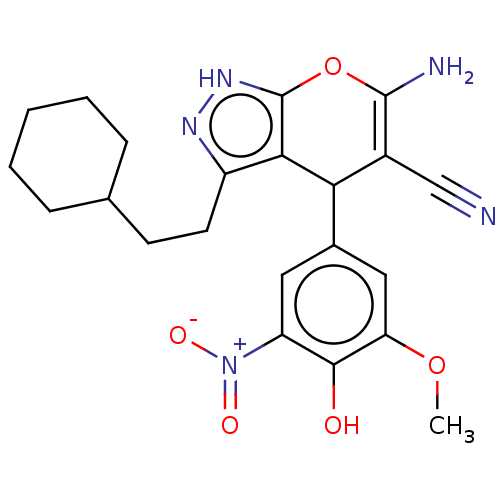
TargetAldo-keto reductase family 1 member C1(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataKi: 0.850nMAssay Description:Competitive inhibition of human recombinant AKR1C1 Phe311Leu mutant by fluorescence assayMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C1(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataKi: 0.860nMAssay Description:Inhibition of human wild type AKR1C1 dehydrogenase activity by fluorometric assayMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C1(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataKi: 1.20nMAssay Description:Competitive inhibition of human recombinant AKR1C1 Leu308Ala mutant by fluorescence assayMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C1(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataKi: 1.30nMAssay Description:Inhibition of human wild type AKR1C1 dehydrogenase activity by fluorometric assayMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C1(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataKi: 1.30nMAssay Description:Inhibition of human wild type AKR1C1 dehydrogenase activity by fluorometric assayMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C1(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataKi: 1.5nMAssay Description:Competitive inhibition of human recombinant AKR1C1 Phe311Ala mutant by fluorescence assayMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C1(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataKi: 1.70nMAssay Description:Competitive inhibition of human recombinant AKR1C1 Leu308Val mutant by fluorescence assayMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C1(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataKi: 2nMAssay Description:Inhibition of human wild type AKR1C1 dehydrogenase activity by fluorometric assayMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C1(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataKi: 2.10nMAssay Description:Inhibition of human wild type AKR1C1 dehydrogenase activity by fluorometric assayMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C1(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataKi: 2.60nMAssay Description:Inhibition of human wild type AKR1C1 dehydrogenase activity by fluorometric assayMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C1(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataKi: 4nMAssay Description:Inhibition of human recombinant 20-alpha HSD expressed in Escherichia coli JM109More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C1(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataKi: 4.10nMAssay Description:Inhibition of human recombinant AKR1C1 assessed as 1-acenaphthenol oxidation at 400 uM by spectrophotometryMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C1(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataKi: 4.10nMAssay Description:Inhibition of human wild type AKR1C1 dehydrogenase activity by fluorometric assayMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C1(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataKi: 4.80nMAssay Description:Competitive inhibition of human recombinant AKR1C1 Leu54Val mutant by fluorescence assayMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C1(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataKi: 5.90nM ΔG°: -47.0kJ/molepH: 7.4 T: 2°CAssay Description:The activity was assayed by measuring the rate of change in NADPH fluorescence (at 455 nm with an excitation wavelength of 340 nm) at 298 K. When the...More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C1(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataKi: 5.90nMAssay Description:Inhibition of human wild type AKR1C1 dehydrogenase activity by fluorometric assayMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C1(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataIC50: 6nMAssay Description:Inhibition of 20-alpha HSD (unknown origin)More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C1(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataKi: 6.60nMAssay Description:Competitive inhibition of human recombinant AKR1C1 Phe311Ala mutant by fluorescence assayMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C1(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataKi: 7nMAssay Description:Competitive inhibition of human recombinant AKR1C1 Phe311Leu mutant by fluorescence assayMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C1(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataKi: 9nMAssay Description:Inhibition of human recombinant 20-alpha HSD expressed in Escherichia coli JM109More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C1(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataIC50: 17.7nMAssay Description:Inhibitors were initially screened for an ability to block the NADP+ dependent oxidation of the artificial substrate S-tetralol catalyzed by AKR1C3. ...More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C1(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataKi: 29nMAssay Description:Inhibition of human wild type AKR1C1 dehydrogenase activity by fluorometric assayMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C1(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataIC50: 30.7nMAssay Description:Inhibitors were initially screened for an ability to block the NADP+ dependent oxidation of the artificial substrate S-tetralol catalyzed by AKR1C3. ...More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C1(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataIC50: 33nMAssay Description:Inhibition of 20-alpha HSD (unknown origin)More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C1(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataIC50: 48nMAssay Description:Inhibition of 20-alpha HSD (unknown origin)More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C1(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataKd: 52nMAssay Description:Binding affinity to human AKR1C1 incubated for 45 mins by Kinobead based pull down assayMore data for this Ligand-Target Pair
Affinity DataKi: 58nM ΔG°: -41.3kJ/molepH: 7.4 T: 2°CAssay Description:The activity was assayed by measuring the rate of change in NADPH fluorescence (at 455 nm with an excitation wavelength of 340 nm) at 298 K. When the...More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C1(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataKi: 64nMAssay Description:Inhibition of human wild type AKR1C1 dehydrogenase activity by fluorometric assayMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C1(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataKi: 70nMAssay Description:Competitive inhibition of human recombinant AKR1C1 Leu306Ala mutant by fluorescence assayMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C1(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataIC50: 76.3nMAssay Description:Inhibitors were initially screened for an ability to block the NADP+ dependent oxidation of the artificial substrate S-tetralol catalyzed by AKR1C3. ...More data for this Ligand-Target Pair
Affinity DataKi: 85nM ΔG°: -40.4kJ/molepH: 7.4 T: 2°CAssay Description:The activity was assayed by measuring the rate of change in NADPH fluorescence (at 455 nm with an excitation wavelength of 340 nm) at 298 K. When the...More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C1(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataKi: 96nMAssay Description:Inhibition of human wild type AKR1C1 dehydrogenase activity by fluorometric assayMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C1(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataIC50: 100nMAssay Description:Inhibitors were initially screened for an ability to block the NADP+ dependent oxidation of the artificial substrate S-tetralol catalyzed by AKR1C3. ...More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C1(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataIC50: 100nMAssay Description:Inhibition of human AKR1C1-mediated progesterone metabolism expressed in bovine aortic endothelial cells assessed as formation of 20alpha-hydroxyprog...More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C1(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataIC50: 100nMAssay Description:Inhibitors were initially screened for an ability to block the NADP+ dependent oxidation of the artificial substrate S-tetralol catalyzed by AKR1C3. ...More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C1(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataIC50: 100nMAssay Description:Inhibitors were initially screened for an ability to block the NADP+ dependent oxidation of the artificial substrate S-tetralol catalyzed by AKR1C3. ...More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C1(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataIC50: 100nMAssay Description:Inhibitors were initially screened for an ability to block the NADP+ dependent oxidation of the artificial substrate S-tetralol catalyzed by AKR1C3. ...More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C1(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataIC50: 100nMAssay Description:Inhibitors were initially screened for an ability to block the NADP+ dependent oxidation of the artificial substrate S-tetralol catalyzed by AKR1C3. ...More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C1(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataIC50: 100nMAssay Description:Inhibitors were initially screened for an ability to block the NADP+ dependent oxidation of the artificial substrate S-tetralol catalyzed by AKR1C3. ...More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C1(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataIC50: 100nMAssay Description:Inhibitors were initially screened for an ability to block the NADP+ dependent oxidation of the artificial substrate S-tetralol catalyzed by AKR1C3. ...More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C1(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataIC50: 100nMAssay Description:Inhibitors were initially screened for an ability to block the NADP+ dependent oxidation of the artificial substrate S-tetralol catalyzed by AKR1C3. ...More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C1(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataKi: 140nMAssay Description:Inhibition of human recombinant 20-alpha HSD expressed in Escherichia coli JM109More data for this Ligand-Target Pair
Affinity DataKi: 270nM ΔG°: -37.5kJ/molepH: 7.4 T: 2°CAssay Description:The activity was assayed by measuring the rate of change in NADPH fluorescence (at 455 nm with an excitation wavelength of 340 nm) at 298 K. When the...More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C1(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataIC50: 300nMAssay Description:Inhibition of human AKR1C1-mediated progesterone metabolism expressed in bovine aortic endothelial cells assessed as formation of 20alpha-hydroxyprog...More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C1(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataKi: 340nMAssay Description:Inhibition of human wild type AKR1C1 dehydrogenase activity by fluorometric assayMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C1(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataIC50: 411nMAssay Description:Inhibition of recombinant human AKR1C1 expressed in Escherichia coli BL21 cells in presence of 9,10-phenanthrenequinone and NADPH by fluorescence ass...More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C1(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataIC50: 433nMAssay Description:Inhibition of recombinant human AKR1C1 expressed in Escherichia coli BL21 cells in presence of 9,10-phenanthrenequinone and NADPH by fluorescence ass...More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C1(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataIC50: 443nMAssay Description:Inhibition of recombinant human AKR1C1 expressed in Escherichia coli BL21 cells in presence of 9,10-phenanthrenequinone and NADPH by fluorescence ass...More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C1(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataIC50: 460nMAssay Description:Inhibition of human AKR1C1-mediated progesterone metabolism expressed in bovine aortic endothelial cells assessed as formation of 20alpha-hydroxyprog...More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C1(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataIC50: 460nMAssay Description:Inhibition of human recombinant 20-alpha HSD expressed in BAEC assessed as inhibition of progesterone metabolism treated 2 hrs before progesterone ch...More data for this Ligand-Target Pair

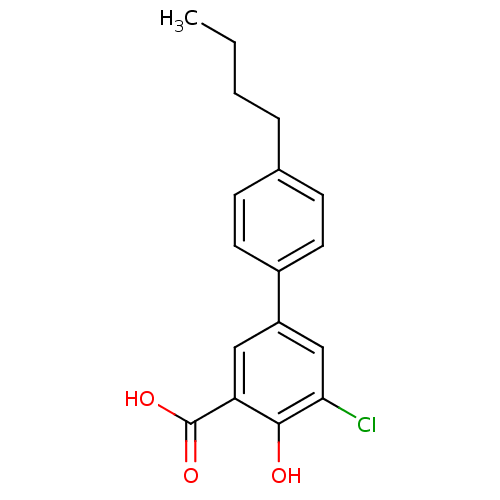
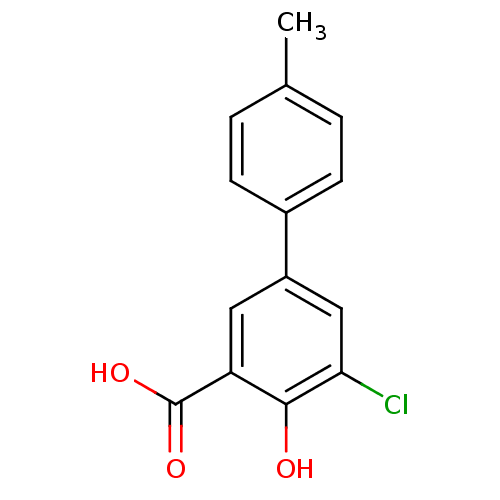
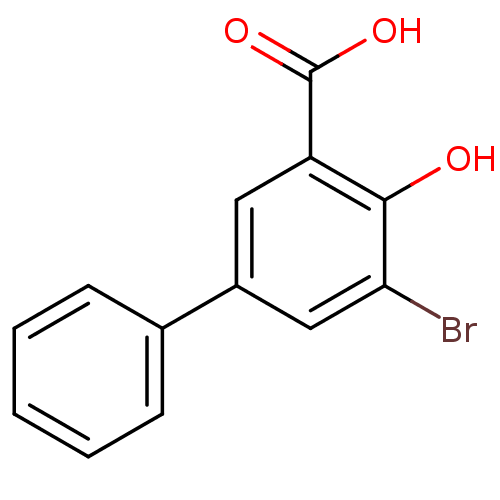
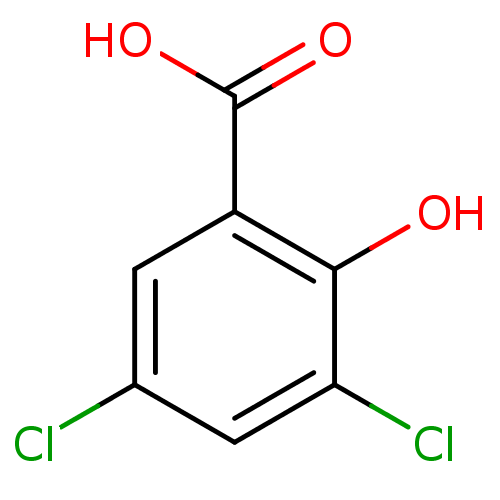
 3D Structure (crystal)
3D Structure (crystal)