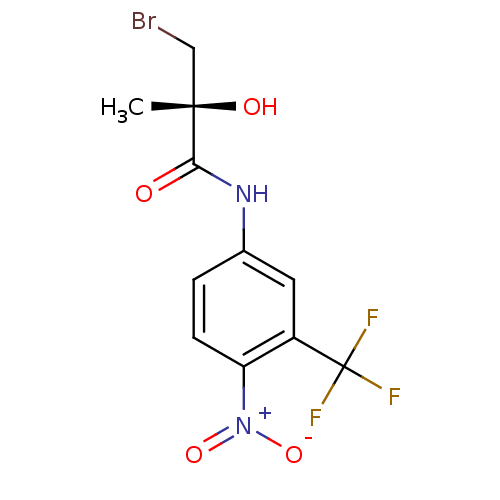
BDBM18699 (2R)-3-bromo-2-hydroxy-2-methyl-N-[4-nitro-3-(trifluoromethyl)phenyl]propanamide::CHEMBL296323::R-3
SMILES C[C@](O)(CBr)C(=O)Nc1ccc(c(c1)C(F)(F)F)[N+]([O-])=O
InChI Key InChIKey=QDSWNDMHSBZXKX-JTQLQIEISA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 4 hits for monomerid = 18699
Found 4 hits for monomerid = 18699
Affinity DataKi: 0.300nM ΔG°: -12.1kcal/mole EC50: 500nMpH: 7.4 T: 2°CAssay Description:The Ki values were determined by the application of the Cheng-Prusoff equation: Ki = (IC50 x Kd)/(Kd+[L]) where [L] is the concentration of [3H]MIB (...More data for this Ligand-Target Pair
TargetAndrogen receptor(Homo sapiens (Human))
The University of Tennessee Health Science Center
Curated by ChEMBL
The University of Tennessee Health Science Center
Curated by ChEMBL
Affinity DataKi: 0.300nMAssay Description:Binding affinity against human androgen receptor (hAR) in competitive binding assayMore data for this Ligand-Target Pair
TargetAndrogen receptor(Homo sapiens (Human))
The University of Tennessee Health Science Center
Curated by ChEMBL
The University of Tennessee Health Science Center
Curated by ChEMBL
Affinity DataKi: 0.300nMAssay Description:Agonist activity at androgen receptor (unknown origin)More data for this Ligand-Target Pair
Affinity DataKi: 0.302nMAssay Description:Inhibition of [3H]mibolerone binding to cytosolic androgen receptor of rat ventral prostateMore data for this Ligand-Target Pair
 3D Structure (crystal)
3D Structure (crystal)