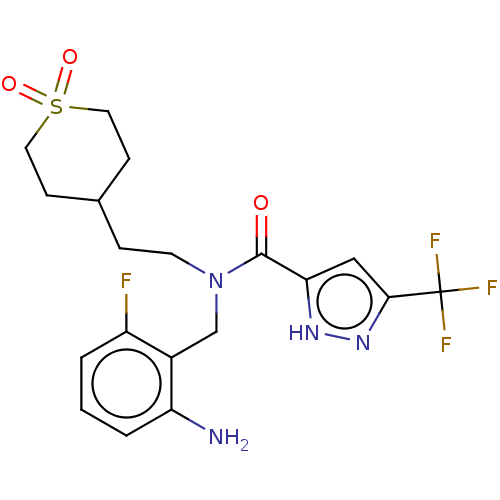
BDBM50022801 CHEMBL5274026
SMILES Nc1cccc(F)c1CN(CCC1CCS(=O)(=O)CC1)C(=O)c1cc(n[nH]1)C(F)(F)F
InChI Key InChIKey=GASMZUDGPKMAEI-UHFFFAOYSA-N
Data 1 EC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 2 hits for monomerid = 50022801
Found 2 hits for monomerid = 50022801
TargetPyruvate kinase PKM(Human)
National Institute of Pharmaceutical Education and Research Ahmedabad
Curated by ChEMBL
National Institute of Pharmaceutical Education and Research Ahmedabad
Curated by ChEMBL
Affinity DataEC50: 7.10nMAssay Description:Activation of PKM2 (unknown origin) in presence of sorafenibMore data for this Ligand-Target Pair
TargetPyruvate kinase PKM(Human)
National Institute of Pharmaceutical Education and Research Ahmedabad
Curated by ChEMBL
National Institute of Pharmaceutical Education and Research Ahmedabad
Curated by ChEMBL
Affinity DataEC50: 50nMAssay Description:Activation of PKM2 (unknown origin)More data for this Ligand-Target Pair