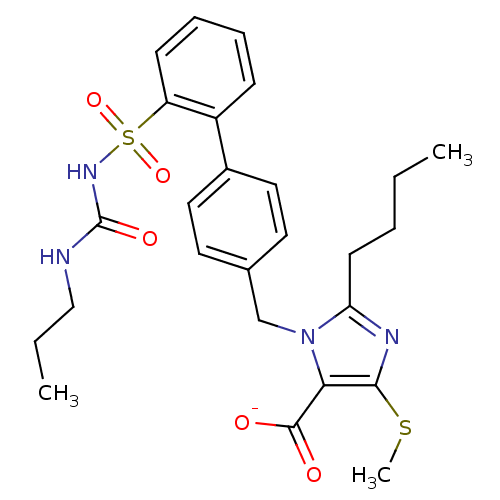
BDBM50031530 2-Butyl-4-(methylthio)-1-[[2'-[[[(propylamino)carbonyl]amino]sulfonyl(1,1'-biphenyl)-4-yl]-methyl]-1H-imidazol-5-carboxylic acid Dipotassium salt::5-alpha-hydroxyacid imidazolyl biphenyl sulfonyl urea analogue::CHEMBL80370::potassium 2-butyl-4-(methylsulfanyl)-1-{[4-(2-{[(propylcarbamoyl)amino]sulfonyl}phenyl)phenyl]methyl}-1H-imidazole-5-carboxylate
SMILES CCCCc1nc(SC)c(C([O-])=O)n1Cc1ccc(cc1)-c1ccccc1S(=O)(=O)NC(=O)NCCC
InChI Key InChIKey=DKCCTYVLJMSFOG-UHFFFAOYSA-M
Data 4 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 4 hits for monomerid = 50031530
Found 4 hits for monomerid = 50031530
TargetType-1 angiotensin II receptor A/B(Rat)
Hoechst Roussel Pgu Cardiovascular Agents
Curated by ChEMBL
Hoechst Roussel Pgu Cardiovascular Agents
Curated by ChEMBL
Affinity DataIC50: 0.480nMAssay Description:Specific binding inhibition of [125I]AII to Angiotensin II receptor, type 1 in rat liver membraneMore data for this Ligand-Target Pair
TargetType-1 angiotensin II receptor A/B(Rat)
Hoechst Roussel Pgu Cardiovascular Agents
Curated by ChEMBL
Hoechst Roussel Pgu Cardiovascular Agents
Curated by ChEMBL
Affinity DataIC50: 0.5nMAssay Description:Concentration required for 50% inhibition of [125I]- AII binding to rat liver membrane preparation (AT1)More data for this Ligand-Target Pair
TargetType-1 angiotensin II receptor A/B(Rat)
Hoechst Roussel Pgu Cardiovascular Agents
Curated by ChEMBL
Hoechst Roussel Pgu Cardiovascular Agents
Curated by ChEMBL
Affinity DataIC50: 0.5nMAssay Description:In vitro binding affinity at Angiotensin II Type 1 receptor in rat liver membrane by [125I]AII displacement.More data for this Ligand-Target Pair
Affinity DataIC50: 920nMAssay Description:In vitro binding affinity at Angiotensin II Type 2 receptor in rabbit uterus membrane by [125I]AII displacement.More data for this Ligand-Target Pair