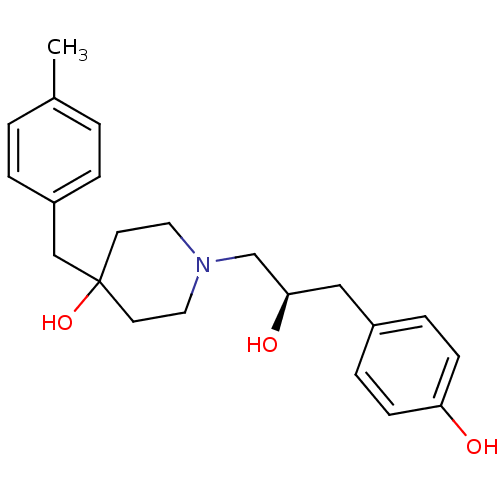
BDBM50103133 1-[(R)-2-Hydroxy-3-(4-hydroxy-phenyl)-propyl]-4-(4-methyl-benzyl)-piperidin-4-ol::CHEMBL70918
SMILES Cc1ccc(CC2(O)CCN(C[C@H](O)Cc3ccc(O)cc3)CC2)cc1
InChI Key InChIKey=MAQRRFANFOCRSD-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 3 hits for monomerid = 50103133
Found 3 hits for monomerid = 50103133
Affinity DataIC50: 19nMAssay Description:Evaluated for in vitro inhibition of recombinant rat NR1C/2B receptor expressed in xenopus oocytesMore data for this Ligand-Target Pair
Affinity DataKi: 7.30E+3nMAssay Description:In vitro binding affinity towards Alpha-1 adrenergic receptor by displacing [3H]prazosin radioligandMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Evaluated for in vitro inhibition of recombinant rat NR1C/2A receptor expressed in xenopus oocytesMore data for this Ligand-Target Pair