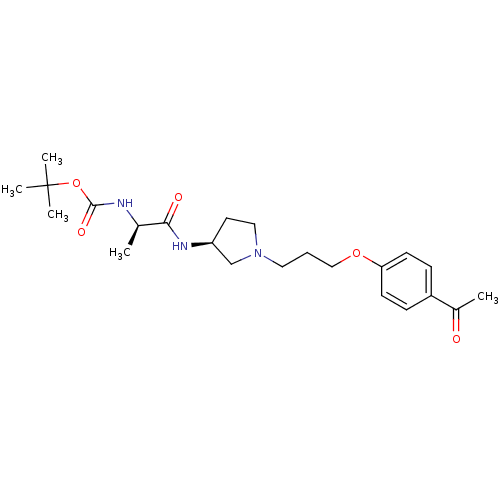
BDBM50119726 (1-{(S)-1-[3-(4-Acetyl-phenoxy)-propyl]-pyrrolidin-3-ylcarbamoyl}-ethyl)-carbamic acid tert-butyl ester::CHEMBL103828
SMILES C[C@@H](NC(=O)OC(C)(C)C)C(=O)N[C@H]1CCN(CCCOc2ccc(cc2)C(C)=O)C1
InChI Key InChIKey=JWVSNNPXFZNTNU-UHFFFAOYSA-N
Data 3 KI
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 3 hits for monomerid = 50119726
Found 3 hits for monomerid = 50119726
Affinity DataKi: 307nMAssay Description:Binding affinity towards rats Histamine type 3 (H3) receptorMore data for this Ligand-Target Pair
Affinity DataKi: 4.00E+4nMAssay Description:Binding affinity towards human Histamine H2 receptor (For compound 11)More data for this Ligand-Target Pair
Affinity DataKi: 5.60E+4nMAssay Description:Binding affinity to the human Histamine H1 receptorMore data for this Ligand-Target Pair