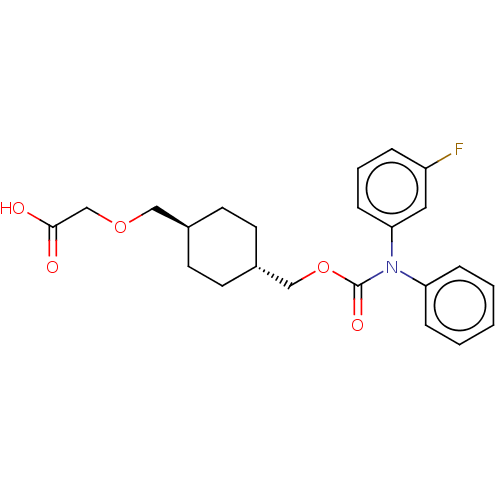
BDBM50235370 CHEMBL3933704
SMILES OC(=O)COC[C@H]1CC[C@H](COC(=O)N(c2ccccc2)c2cccc(F)c2)CC1
InChI Key InChIKey=UMQLASXGJFGXFL-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 17 hits for monomerid = 50235370
Found 17 hits for monomerid = 50235370
Affinity DataKi: 7nMAssay Description:Displacement of [3H]-iloprost from recombinant human IP receptor expressed in CHO-K1 cell membranes incubated for 1 hr by top count scintillation cou...More data for this Ligand-Target Pair
Affinity DataEC50: 8.70nMAssay Description:Agonist activity at recombinant human IP receptor expressed in CHO-K1 cells assessed as increase in intracellular cAMP level after 1 hr incubation by...More data for this Ligand-Target Pair
Affinity DataIC50: 18nMAssay Description:Agonist activity at IP receptor in human primary platelets assessed as inhibition of ADP-induced platelet aggregationMore data for this Ligand-Target Pair
Affinity DataKi: 51nMAssay Description:Displacement of [3H]-iloprost from recombinant rat IP receptor expressed in CHO-K1 cell membranes incubated for 1 hr by top count scintillation count...More data for this Ligand-Target Pair
Affinity DataKi: 210nMAssay Description:Displacement of [3H]-PGE2 from recombinant human EP3v6 receptor expressed in HEK293 cell membranes incubated for 1 hr by top count scintillation coun...More data for this Ligand-Target Pair
Affinity DataEC50: 284nMAssay Description:Agonist activity at recombinant rat IP receptor expressed in CHO-K1 cells assessed as increase in intracellular cAMP level after 1 hr incubation by H...More data for this Ligand-Target Pair
Affinity DataKi: 570nMAssay Description:Displacement of [3H]-PGE2 from recombinant human EP2 receptor expressed in HEK293 cell membranes incubated for 1 hrMore data for this Ligand-Target Pair
Affinity DataKi: 1.20E+3nMAssay Description:Displacement of [3H]-PGE2 from recombinant human EP4 receptor expressed in rat chem-1 cell membranes incubated for 1 hrMore data for this Ligand-Target Pair
Affinity DataKi: 1.85E+3nMAssay Description:Displacement of [3H]-PGD2 from recombinant human DP1 receptorMore data for this Ligand-Target Pair
Affinity DataEC50: 2.76E+3nMAssay Description:Agonist activity at recombinant human DP1 receptor expressed in CHO-K1 cells assessed as increase in intracellular cAMP level after 1 hr incubation b...More data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Human)
Arena Pharmaceuticals
Curated by ChEMBL
Arena Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 3.00E+4nMAssay Description:Inhibition of human ERG by patch clamp assayMore data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+4nMAssay Description:Inhibition of CYP2C8 (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+4nMAssay Description:Inhibition of CYP2C9 (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+4nMAssay Description:Inhibition of CYP3A4 (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+4nMAssay Description:Inhibition of CYP2D6 (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+4nMAssay Description:Inhibition of CYP1A2 (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+4nMAssay Description:Inhibition of CYP2C19 (unknown origin)More data for this Ligand-Target Pair