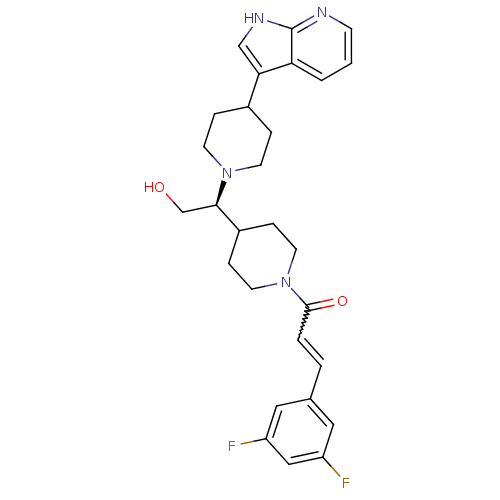
BDBM50246354 (S)-1-(4-(1-(4-(1H-pyrrolo[2,3-b]pyridin-3-yl)piperidin-1-yl)-2-hydroxyethyl)piperidin-1-yl)-3-(3,5-difluorophenyl)prop-2-en-1-one::CHEMBL455683
SMILES OC[C@H](C1CCN(CC1)C(=O)C=Cc1cc(F)cc(F)c1)N1CCC(CC1)c1c[nH]c2ncccc12
InChI Key InChIKey=JWWBRTLPEUWQCU-UHFFFAOYSA-N
Data 7 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 7 hits for monomerid = 50246354
Found 7 hits for monomerid = 50246354
TargetC-C chemokine receptor type 2(Human)
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataIC50: 4nMAssay Description:Antagonist activity at human CCR2More data for this Ligand-Target Pair
TargetCytochrome P450 2D6(Human)
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataIC50: 1.33E+4nMAssay Description:Inhibition of human CYP2D6More data for this Ligand-Target Pair
TargetCytochrome P450 3A4(Human)
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataIC50: 2.06E+4nMAssay Description:Inhibition of human CYP3A4 using dibenzylfluorescein as substrateMore data for this Ligand-Target Pair
TargetCytochrome P450 3A4(Human)
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataIC50: 2.52E+4nMAssay Description:Inhibition of human CYP3A4 using 7-benzyloxy-4-trifluoromethylcoumarin as substrateMore data for this Ligand-Target Pair
TargetCytochrome P450 2C19(Human)
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataIC50: 2.71E+4nMAssay Description:Inhibition of human CYP2C19More data for this Ligand-Target Pair
TargetCytochrome P450 1A2(Human)
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataIC50: 4.00E+4nMAssay Description:Inhibition of human CYP1A2More data for this Ligand-Target Pair
TargetCytochrome P450 3A4(Human)
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataIC50: 4.00E+4nMAssay Description:Inhibition of human CYP3A4 using 7-benzyloxyquinoline as substrateMore data for this Ligand-Target Pair