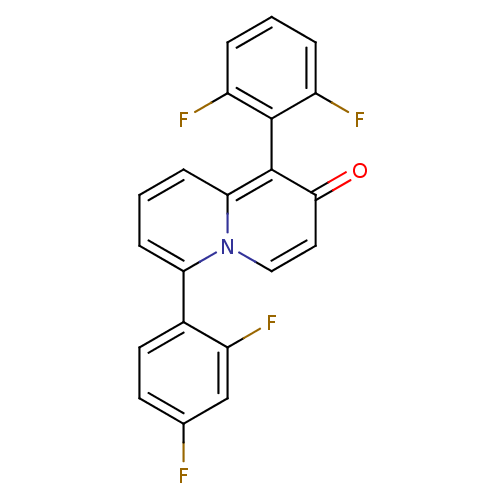
BDBM50317579 6-(2,4-difluorophenyl)-1-(2,6-difluorophenyl)-2H-quinolizin-2-one::CHEMBL1098524
SMILES Fc1ccc(c(F)c1)-c1cccc2c(-c3c(F)cccc3F)c(=O)ccn12
InChI Key InChIKey=FMRNKJBMXLVUBM-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 6 hits for monomerid = 50317579
Found 6 hits for monomerid = 50317579
Affinity DataIC50: 7.10nMAssay Description:Inhibition of mouse p38apha assessed as reduction in GST-ATF2 substrate phosphorylation by SPA assayMore data for this Ligand-Target Pair
Affinity DataIC50: 7.10nMAssay Description:Inhibition of mouse p38alpha after 3 hrs by SPA methodMore data for this Ligand-Target Pair
Affinity DataIC50: 9.20nMAssay Description:Inhibition of mouse p38beta assessed as reduction in GST-ATF2 substrate phosphorylation by SPA assayMore data for this Ligand-Target Pair
TargetNuclear receptor subfamily 1 group I member 2(Human)
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataEC50: 3.30E+3nMAssay Description:Activation of PXRMore data for this Ligand-Target Pair
TargetNuclear receptor subfamily 1 group I member 2(Human)
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataEC50: 3.30E+3nMAssay Description:Activation of PXRMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Human)
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataKi: >2.00E+4nMAssay Description:Displacement of labeled MK-499 from human ERG in HEK293 cellsMore data for this Ligand-Target Pair