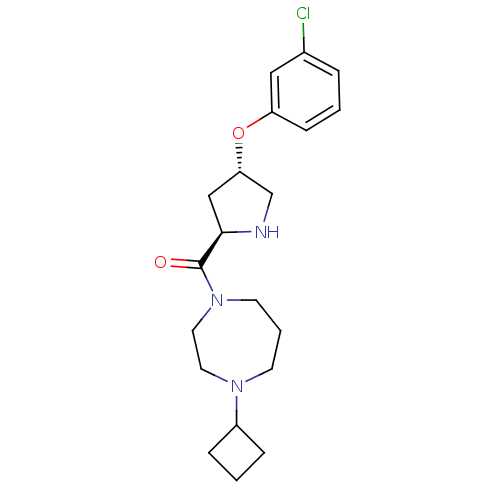
BDBM50317702 ((2R,4S)-4-(3-chlorophenoxy)pyrrolidin-2-yl)(4-cyclobutyl-1,4-diazepan-1-yl)methanone::CHEMBL1095877
SMILES Clc1cccc(O[C@@H]2CN[C@H](C2)C(=O)N2CCCN(CC2)C2CCC2)c1
InChI Key InChIKey=CYVDMGAAGFJWCN-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 4 hits for monomerid = 50317702
Found 4 hits for monomerid = 50317702
TargetHistamine H3 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 2.5nMAssay Description:Binding affinity to human histamine H3 receptorMore data for this Ligand-Target Pair
TargetHistamine H3 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 2.5nMAssay Description:Displacement of [125I]Iodoproxyfan from human recombinant histamine H3 receptor by Competitive binding assayMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataIC50: 7.20E+3nMAssay Description:Displacement of labeled dofetilide human ERGMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataIC50: 7.20E+3nMAssay Description:Displacement of [3H]dofetilide from human recombinant ERG by Competitive binding assayMore data for this Ligand-Target Pair