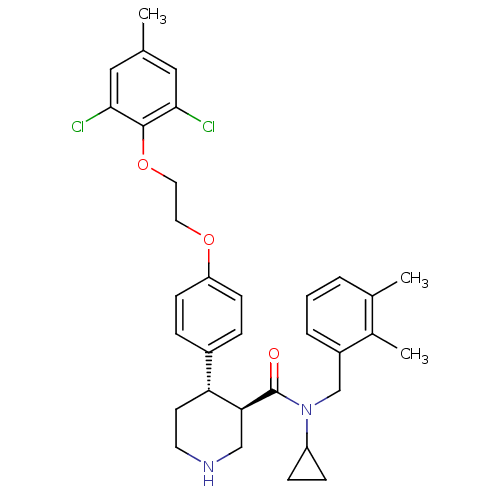
BDBM50328854 CHEMBL1270661::rac-N-cyclopropyl-4-(4-(2-(2,6-dichloro-4-methylphenoxy)ethoxy)phenyl)-N-(2,3-dimethylbenzyl)piperidine-3-carboxamide
SMILES Cc1cc(Cl)c(OCCOc2ccc(cc2)[C@H]2CCNC[C@@H]2C(=O)N(Cc2cccc(C)c2C)C2CC2)c(Cl)c1
InChI Key InChIKey=PEFPYUBFXFOFOB-UHFFFAOYSA-N
Data 5 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 5 hits for monomerid = 50328854
Found 5 hits for monomerid = 50328854
Affinity DataIC50: 0.310nMAssay Description:Inhibition of renin in bufferMore data for this Ligand-Target Pair
Affinity DataIC50: 13nMAssay Description:Inhibition of renin in plasmaMore data for this Ligand-Target Pair
Affinity DataIC50: 7.00E+3nMAssay Description:Inhibition of CYP3A4More data for this Ligand-Target Pair
Affinity DataIC50: 1.10E+4nMAssay Description:Inhibition of CYP3A4 using testosterone as substrateMore data for this Ligand-Target Pair
Affinity DataIC50: 1.10E+4nMAssay Description:Inhibition of CYP3A4 using testosterone as substrateMore data for this Ligand-Target Pair