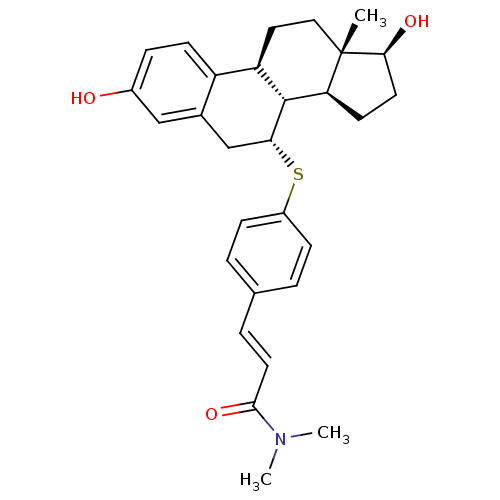
BDBM50366662 CHEMBL1627356
SMILES CN(C)C(=O)\C=C\c1ccc(S[C@@H]2Cc3cc(O)ccc3[C@H]3CC[C@]4(C)[C@@H](O)CC[C@H]4[C@H]23)cc1
InChI Key InChIKey=FROPUNBJULYSGP-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 3 hits for monomerid = 50366662
Found 3 hits for monomerid = 50366662
Affinity DataIC50: 59nMAssay Description:Displacement of [3H]17-beta-estradiol from human Estrogen receptor betaMore data for this Ligand-Target Pair
Affinity DataIC50: 94nMAssay Description:Agonist effect on transcriptional activation in MCF-7 cells expressing estrogen receptor alphaMore data for this Ligand-Target Pair
Affinity DataEC50: 173nMAssay Description:Agonist effect on transcriptional activation in MCF-7 cells expressing estrogen receptor alphaMore data for this Ligand-Target Pair