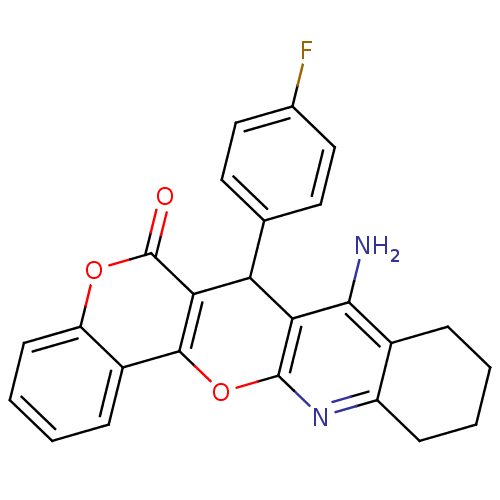
BDBM50440441 CHEMBL2425846
SMILES Nc1c2CCCCc2nc2Oc3c(C(c4ccc(F)cc4)c12)c(=O)oc1ccccc31
InChI Key InChIKey=DHAXWBWQDZCBTC-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 3 hits for monomerid = 50440441
Found 3 hits for monomerid = 50440441
Affinity DataIC50: 5nMAssay Description:Inhibition of electric eel AChE using acetylthiocholine iodide as substrate preincubated for 10 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataKi: 22nMAssay Description:Mixed-type inhibition of electric eel AChE using acetylthiocholine iodide as substrate by Lineweaver-Burk plot analysisMore data for this Ligand-Target Pair
Affinity DataIC50: 1.57E+4nMAssay Description:Inhibition of equine serum BChE using butyrylthiocholine iodide as substrate preincubated for 10 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair